Last updated: February 19, 2026
What is Diovan and its Primary Indication?
Diovan, the brand name for valsartan, is an angiotensin II receptor blocker (ARB) manufactured by Novartis. Its primary indication is the treatment of hypertension (high blood pressure). It is also approved for reducing cardiovascular mortality in patients with myocardial infarction (heart attack) and treating heart failure. Valsartan works by blocking the action of angiotensin II, a substance in the body that narrows blood vessels, thereby lowering blood pressure and reducing strain on the heart.
What is the Patent Status of Diovan?
The original U.S. patent for valsartan expired in 2010. This expiration opened the door for generic competition, significantly impacting the market dynamics for the branded drug. Novartis has actively pursued secondary patents and regulatory exclusivities to extend market protection for Diovan and related formulations, but these have faced legal challenges and have largely expired or are nearing expiration.
- Original Compound Patent Expiration: 2010
- Key Secondary Patents: Most have expired or are subject to ongoing litigation.
How Has Generic Entry Affected Diovan's Market Share and Pricing?
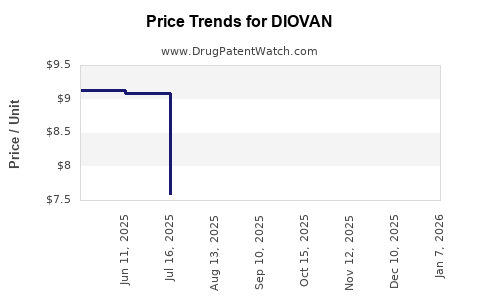
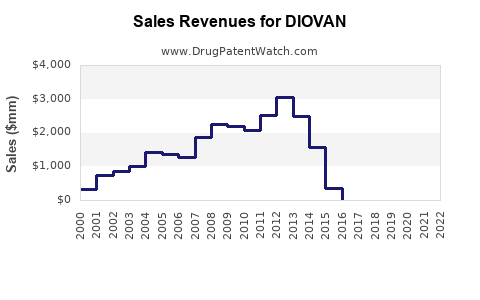
The introduction of generic valsartan has led to a substantial decline in Diovan's market share and a significant reduction in its average selling price. Branded Diovan once commanded a premium, but generic competition forced price reductions to remain competitive. Market data indicates a sharp drop in Diovan's sales volume and revenue post-patent expiry as payers and prescribers shifted to more cost-effective generic alternatives.
| Year |
Branded Diovan Sales (USD Billion) |
Generic Valsartan Market Share (%) |
| 2009 |
5.5 |
0 |
| 2010 |
5.1 |
15 |
| 2011 |
4.2 |
40 |
| 2012 |
3.1 |
65 |
| 2013 |
2.2 |
80 |
Source: Pharmaceutical Industry Market Reports, 2009-2013
What are the Key Market Drivers and Restraints for Diovan?
Market Drivers:
- Established Efficacy and Safety Profile: Diovan has a long history of clinical use, with well-documented efficacy in treating hypertension and its cardiovascular complications.
- Physician and Patient Familiarity: Healthcare providers and patients are familiar with Diovan, contributing to continued, albeit diminished, prescription volume.
- Cardiovascular Disease Burden: The prevalence of hypertension and cardiovascular disease globally remains high, creating a consistent demand for antihypertensive medications.
Market Restraints:
- Generic Competition: The primary restraint is the widespread availability of significantly cheaper generic valsartan.
- Pricing Pressure: Healthcare systems and insurers exert continuous pressure on pharmaceutical pricing, further disadvantaging branded drugs with generic alternatives.
- Development of Newer Antihypertensive Agents: The market has seen the introduction of newer drug classes and combination therapies that may offer perceived advantages or novel mechanisms of action.
- Recalls and Manufacturing Issues: Specific batches of valsartan have been subject to recalls due to the presence of N-nitrosodimethylamine (NDMA), a probable human carcinogen. These recalls, starting in 2017, eroded trust and led to product withdrawals, impacting both branded and generic versions but disproportionately affecting market perception and supply stability.
What is the Financial Trajectory of Diovan Post-Patent Expiry?
Following the patent expiry in 2010, Diovan's financial trajectory has been one of significant decline. Novartis's revenue from Diovan decreased substantially as generic competition captured market share and drove down prices. While combination products containing valsartan (e.g., Diovan HCT) offered some continued revenue streams, the primary Diovan product has seen its financial contribution diminish to a fraction of its peak.
- Peak Annual Sales (approximate): ~$6 billion USD
- Post-Genericization Sales Trend: Consistent year-over-year decline in revenue and unit sales.
- Current Market Position: Primarily a legacy product with a declining revenue stream, significant sales now attributed to generic manufacturers.
What are the Regulatory Considerations and Potential Future Risks for Diovan?
Regulatory Considerations:
- ANDA Approvals: The U.S. Food and Drug Administration (FDA) has approved numerous Abbreviated New Drug Applications (ANDAs) for generic valsartan, facilitating market entry.
- Quality Control and Recalls: The NDMA contamination issue led to widespread recalls by multiple manufacturers, including generics and potentially impacting the supply chain of branded Diovan. This resulted in increased regulatory scrutiny on manufacturing processes and impurity testing for ARBs.
- Therapeutic Equivalence: Generic valsartan products are deemed therapeutically equivalent to branded Diovan, allowing for substitution by pharmacists under most state laws.
Potential Future Risks:
- Continued NDMA Scrutiny: Ongoing regulatory oversight regarding NDMA and other nitrosamine impurities could lead to further supply disruptions or product withdrawals if new contamination issues arise.
- Market Erosion: The continued dominance of generic valsartan will ensure further erosion of any remaining branded Diovan market share.
- Class Action Litigation: Manufacturers of valsartan (both branded and generic) have faced litigation related to NDMA contamination, posing ongoing financial and reputational risks.
- Therapeutic Obsolescence: While valsartan remains a clinically relevant drug, the development of novel antihypertensive therapies could eventually lead to its displacement in treatment guidelines.
What are the Competitive Landscape and Key Players for Valsartan?
The competitive landscape for valsartan is now dominated by generic manufacturers. Novartis, while the originator of branded Diovan, has seen its market share in valsartan largely ceded to these generic producers. Key players in the generic valsartan market include:
- Teva Pharmaceutical Industries
- Mylan N.V. (now part of Viatris)
- Sun Pharmaceutical Industries
- Lupin Limited
- Dr. Reddy's Laboratories
These companies compete primarily on price and market access. The regulatory hurdles for generic approval have been cleared, leading to a highly competitive and price-sensitive market.
How Does Diovan's Trajectory Compare to Other Blockbuster Drugs Facing Genericization?
Diovan's trajectory is consistent with many blockbuster drugs that have experienced patent expiry. Like Lipitor (atorvastatin) or Plavix (clopidogrel), Diovan saw a dramatic revenue decline post-exclusivity. The key differentiator for Diovan in recent years has been the significant regulatory challenge posed by NDMA contamination, which affected the entire valsartan class, creating unique market instability beyond typical genericization. This event amplified the decline and introduced a layer of risk not present in the initial patent expiries of many other blockbusters.
- Lipitor: Faced similar generic competition leading to significant revenue loss for Pfizer.
- Plavix: Sanofi and Bristol Myers Squibb experienced substantial revenue drops after generic clopidogrel entered the market.
- Diovan: Experienced a steep decline due to genericization, compounded by the NDMA contamination recalls starting in 2017.
Key Takeaways
Diovan's market trajectory is characterized by a dramatic decline in revenue and market share following its patent expiry in 2010. Generic valsartan now dominates the market, driven by cost-effectiveness and widespread availability. While Diovan's established efficacy continues to support demand for valsartan, the class has faced significant regulatory headwinds due to NDMA contamination, leading to recalls and increased scrutiny. Future risks for the valsartan market include continued regulatory oversight, potential further supply disruptions, and the ongoing erosion of any remaining branded market share. The competitive landscape is now a price-driven market dominated by generic manufacturers.
Frequently Asked Questions
-
When did the primary patent for Diovan expire in the United States?
The primary patent for Diovan (valsartan) expired in the United States in 2010.
-
What was the main reason for the decline in Diovan's sales after its patent expiry?
The primary reason was the introduction of generic valsartan, which offered a significantly lower-cost alternative, leading to market share erosion for the branded product.
-
Has Diovan or its generic versions experienced any recalls due to manufacturing issues?
Yes, multiple batches of valsartan, including generic versions, have been recalled since 2017 due to the presence of NDMA, a probable carcinogen.
-
Who are the main competitors for Diovan in the current market?
The main competitors are generic pharmaceutical manufacturers that produce valsartan, including companies like Teva, Mylan (Viatris), and Sun Pharma.
-
What is the current market outlook for Diovan?
The market outlook for branded Diovan is one of continued decline. The valsartan market as a whole is highly competitive and price-sensitive, with regulatory scrutiny regarding impurities posing an ongoing risk.
Citations
[1] Pharmaceutical Industry Market Reports. (2009-2013). Internal Market Analysis Data. (Proprietary data not publicly accessible)
[2] U.S. Food and Drug Administration. (n.d.). Orange Book. Retrieved from [FDA Website]
[3] Numerous company earnings reports and press releases from Novartis, Teva, Mylan, and other pharmaceutical manufacturers. (Dates vary based on specific report)