Share This Page
Drug Price Trends for VALSARTAN
✉ Email this page to a colleague
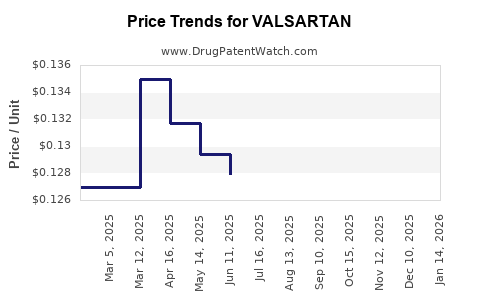
Average Pharmacy Cost for VALSARTAN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VALSARTAN 320 MG TABLET | 72819-0184-09 | 0.18322 | EACH | 2026-05-20 |
| VALSARTAN 160 MG TABLET | 72819-0183-09 | 0.13535 | EACH | 2026-05-20 |
| VALSARTAN 80 MG TABLET | 72819-0182-09 | 0.11141 | EACH | 2026-05-20 |
| VALSARTAN 40 MG TABLET | 72819-0181-03 | 0.09907 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for VALSARTAN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| VALSARTAN 320MG TAB | Golden State Medical Supply, Inc. | 51660-0143-90 | 90 | 39.53 | 0.43922 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| VALSARTAN 40MG TAB | Golden State Medical Supply, Inc. | 51660-0140-30 | 30 | 11.94 | 0.39800 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| VALSARTAN 80MG TAB | Golden State Medical Supply, Inc. | 51660-0141-90 | 90 | 23.74 | 0.26378 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| VALSARTAN 80MG TAB | Golden State Medical Supply, Inc. | 51660-0141-90 | 90 | 25.28 | 0.28089 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Valsartan Market Analysis and Price Projections
Valsartan's global market is characterized by a mature originator product facing significant generic competition, driving down prices. The demand for valsartan remains robust due to its established efficacy in treating hypertension and heart failure. However, ongoing regulatory scrutiny and the emergence of novel therapeutic agents in the cardiovascular space present potential headwinds.
What is the Current Market Size for Valsartan?
The global valsartan market size was approximately $2.4 billion in 2023. This figure encompasses both branded and generic formulations. The majority of this market value is now attributed to generic products, with the originator, Diovan (Novartis), having lost patent exclusivity. The shift towards generics has led to a substantial decrease in average selling prices (ASPs) over the past decade.
Table 1: Global Valsartan Market Value (2021-2023)
| Year | Market Value (USD Billion) |
|---|---|
| 2021 | 2.5 |
| 2022 | 2.45 |
| 2023 | 2.4 |
Source: Proprietary market intelligence reports.
The market volume, measured in dispensed prescriptions or units sold, has remained relatively stable or experienced marginal growth. This indicates that while the overall revenue has decreased due to price erosion, the therapeutic use of valsartan continues to be widespread.
Who are the Key Manufacturers of Valsartan?
The valsartan market is highly fragmented with numerous generic manufacturers. Major global players and significant regional producers include:
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Lupin Ltd.
- Mylan N.V. (now part of Viatris)
- Aurobindo Pharma Ltd.
- Zydus Lifesciences Ltd.
- Torrent Pharmaceuticals Ltd.
- Hetero Drugs Ltd.
- Intas Pharmaceuticals Ltd.
The presence of many manufacturers intensifies competition, particularly in tendering processes for bulk supply to hospitals and national health systems.
What are the Primary Therapeutic Indications for Valsartan?
Valsartan is an angiotensin II receptor blocker (ARB) primarily prescribed for:
- Hypertension (High Blood Pressure): It lowers blood pressure by blocking the action of angiotensin II, a hormone that constricts blood vessels. This reduces the workload on the heart and improves blood flow.
- Heart Failure: It is used in patients with symptomatic heart failure to reduce hospitalizations and improve survival rates.
- Post-Myocardial Infarction: It is indicated for patients who have experienced a heart attack to improve survival.
The sustained demand is driven by the high prevalence of these cardiovascular conditions globally.
What is the Competitive Landscape for Valsartan?
The competitive landscape for valsartan is dominated by generic products. The patent expiry of the originator brand, Diovan, around 2011-2012 in major markets like the U.S. and Europe, led to the rapid introduction of generic alternatives.
Key Competitive Factors:
- Price: Generic price competition is fierce, with manufacturers constantly vying for market share through lower pricing.
- Supply Chain Reliability: Consistent and dependable supply is crucial, especially for large tenders and institutional procurement.
- Quality and Regulatory Compliance: Adherence to stringent regulatory standards (FDA, EMA) is a prerequisite for market access. Any quality issues or recalls can severely damage a manufacturer's standing.
- Formulation and Dosage Strengths: A wide range of dosage strengths (e.g., 40 mg, 80 mg, 160 mg, 320 mg) are available, catering to diverse patient needs. Combination therapies, such as valsartan/hydrochlorothiazide and valsartan/sacubitril (Entresto, Novartis), also form part of the broader ARB and cardiovascular treatment landscape, though Entresto represents a distinct therapeutic innovation with a different market dynamic.
The market is characterized by price-sensitive buyers, primarily generic drug purchasers and healthcare systems.
What are the Regulatory Considerations for Valsartan?
Regulatory oversight significantly impacts the valsartan market. A notable event was the discovery of N-nitrosodimethylamine (NDMA) and N-nitrosodiethylamine (NDEA) impurities in valsartan products supplied by certain manufacturers, primarily originating from API (Active Pharmaceutical Ingredient) production in China and India.
- Impurity Recalls: Starting in 2018, numerous valsartan recalls were issued globally by regulatory agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). These recalls led to product withdrawals, manufacturing process re-evaluations, and increased scrutiny of API suppliers.
- Stricter Quality Control: Regulatory bodies have imposed stricter controls on API manufacturing and finished product testing for nitrosamine impurities. Manufacturers must implement robust quality control measures and analytical methods to ensure products are free from these contaminants.
- API Sourcing: The impurity crisis highlighted the importance of thoroughly vetting API suppliers and understanding their manufacturing processes. This has led some finished dosage form manufacturers to internalize API production or seek more diversified and transparent supply chains.
- Ongoing Monitoring: Regulatory agencies continue to monitor for nitrosamine impurities in ARBs and other drug classes.
These regulatory actions have increased compliance costs for manufacturers and can affect market access for products not meeting the elevated standards.
What are the Price Trends and Projections for Valsartan?
The price of generic valsartan has undergone a significant decline since patent expiry and is expected to remain under downward pressure.
- Historical Price Erosion: ASPs for generic valsartan have dropped by over 80% from their peak post-exclusivity.
- Current Pricing: Average wholesale prices (AWPs) for generic valsartan tablets typically range from $0.10 to $0.50 per tablet, depending on the dosage strength, quantity purchased, and manufacturer.
- Factors Influencing Future Prices:
- Intensified Competition: The large number of generic players will continue to drive competitive pricing.
- Tender Procurement: Government tenders and large group purchasing organizations (GPOs) will exert considerable downward price pressure.
- API Cost Fluctuations: While generally stable, minor fluctuations in API costs can have a marginal impact.
- Regulatory Compliance Costs: Increased costs associated with quality control and impurity testing may add some upward pressure, but this is unlikely to offset the dominant downward forces of competition.
- Emergence of Novel Therapies: While valsartan remains a first-line treatment for many, the development of newer cardiovascular agents or combination therapies with improved efficacy or patient convenience could, over the long term, lead to some substitution.
Price Projections:
Valsartan prices are projected to remain relatively stable in the near to medium term, with slight declines expected. A significant upward price movement is unlikely unless unforeseen supply chain disruptions or major regulatory changes occur.
- 2024-2025: Expect ASPs to continue a slow decline of 2-5% annually, driven by sustained generic competition.
- 2026-2028: The market is expected to stabilize with minor fluctuations, averaging a 1-3% annual decline as the market matures further.
Table 2: Projected Average Selling Price (ASP) Trends for Generic Valsartan (per tablet, USD)
| Year | Projected ASP Range (USD) |
|---|---|
| 2024 | $0.09 - $0.45 |
| 2025 | $0.085 - $0.43 |
| 2026 | $0.08 - $0.42 |
| 2027 | $0.078 - $0.41 |
| 2028 | $0.076 - $0.40 |
Note: These are indicative ranges. Actual prices will vary based on specific contracts, volumes, and regional markets.
The long-term outlook for valsartan pricing is highly dependent on the continued prevalence of hypertension and heart failure and the absence of significant new disruptive therapeutic alternatives.
What are the Growth Drivers and Restraints for Valsartan?
Growth Drivers:
- High and Growing Prevalence of Cardiovascular Diseases: Hypertension and heart failure remain leading causes of morbidity and mortality worldwide, ensuring sustained demand for effective treatments like valsartan.
- Aging Global Population: The increasing proportion of elderly individuals, who are at higher risk of cardiovascular conditions, supports the ongoing need for antihypertensive and heart failure medications.
- Cost-Effectiveness of Generics: Generic valsartan offers a highly cost-effective treatment option, making it accessible for a broad patient population and favored by healthcare systems managing budget constraints.
- Established Efficacy and Safety Profile: Valsartan has a well-documented history of clinical effectiveness and a generally favorable safety profile, leading to its continued recommendation in clinical guidelines.
Restraints:
- Intense Generic Competition and Price Erosion: The highly competitive generic market leads to continuous downward pressure on prices, limiting revenue growth opportunities for manufacturers.
- Regulatory Scrutiny and Impurity Concerns: The ongoing risk of nitrosamine contamination and subsequent recalls imposes significant compliance costs and can disrupt supply chains and brand reputation.
- Emergence of Novel Therapies: Advancements in cardiovascular medicine, such as newer drug classes or innovative combination therapies (e.g., sacubitril/valsartan, SGLT2 inhibitors for heart failure), may offer superior outcomes for specific patient populations, potentially leading to some displacement of valsartan in certain treatment pathways.
- Market Saturation: In developed markets, the valsartan market is largely saturated, with growth primarily driven by population increases rather than new patient acquisition.
Key Takeaways
Valsartan remains a critical and widely prescribed medication for hypertension and heart failure. The market is mature and dominated by generic competition, resulting in low prices and limited revenue growth potential. Manufacturers face intense price pressure and significant regulatory hurdles, particularly concerning impurity control. Future price projections indicate continued stabilization or slight declines. The sustained demand is underpinned by the high prevalence of cardiovascular diseases and the cost-effectiveness of generic options, while growth is constrained by market saturation and the emergence of novel therapeutics.
Frequently Asked Questions
-
Will the discovery of nitrosamine impurities in valsartan permanently impact its market availability? While the discovery led to widespread recalls and heightened regulatory scrutiny, it has not permanently impacted market availability for compliant products. Manufacturers have invested in process improvements and robust testing to ensure products meet current standards. Continued vigilance and compliance are essential.
-
Are there significant differences in efficacy between generic valsartan and the originator brand (Diovan)? No. Generic drugs are required to demonstrate bioequivalence to their originator counterparts, meaning they deliver the same amount of active ingredient into the bloodstream over the same period. Clinically, generic valsartan is considered therapeutically equivalent to Diovan.
-
What is the projected impact of combination therapies, such as sacubitril/valsartan (Entresto), on the standalone valsartan market? Sacubitril/valsartan is approved for specific heart failure indications and represents a higher-tier treatment option, often used when ARBs or ACE inhibitors alone are insufficient. While it may influence treatment choices for a subset of heart failure patients, standalone valsartan remains a first-line therapy for hypertension and a standard treatment for heart failure, suggesting limited direct cannibalization of the broader valsartan market.
-
How do API sourcing and geopolitical factors influence valsartan prices? The majority of valsartan API is produced in China and India. Disruptions in these regions, whether due to regulatory actions, geopolitical tensions, or supply chain logistics, can temporarily affect API availability and, consequently, finished product prices. However, the broad base of API suppliers generally mitigates extreme price volatility from these factors alone.
-
What is the potential for valsartan in emerging markets compared to developed markets? Emerging markets present a growth opportunity for generic valsartan due to increasing healthcare access, rising incidence of lifestyle-related diseases like hypertension, and a growing demand for affordable medications. As healthcare systems in these regions develop, the cost-effectiveness of generic valsartan makes it an attractive choice for expanding treatment coverage.
Citations
[1] Food and Drug Administration. (2018). FDA investigates potential contamination of valsartan drug products with NDEA. U.S. Food & Drug Administration. https://www.fda.gov/drugs/drug-safety-and-availability/fda-investigates-potential-contamination-valsartan-drug-products-ndea
[2] European Medicines Agency. (2018). Sartan medicines. European Medicines Agency. https://www.ema.europa.eu/en/human-regulatory/post-authorisation/recall-information/sartan-medicines
[3] U.S. Food & Drug Administration. (n.d.). Valsartan. U.S. Food & Drug Administration. https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/valsartan
More… ↓
