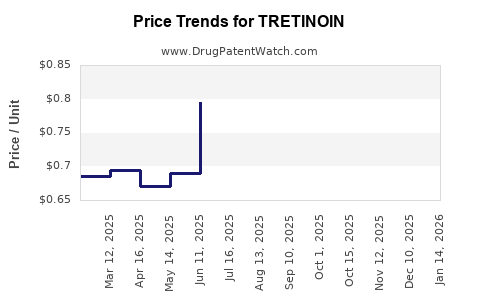
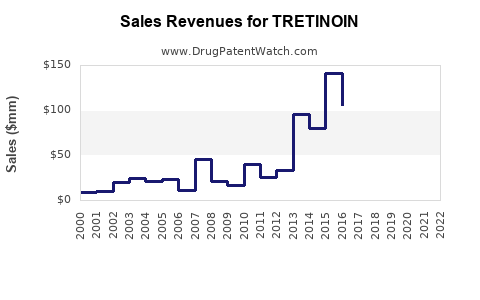
Last updated: February 19, 2026
What is Tretinoin and its Therapeutic Applications?
Tretinoin, a retinoic acid derivative, is a topical retinoid primarily used in dermatology. Its mechanism of action involves regulating skin cell growth and differentiation. The primary therapeutic applications of tretinoin include the treatment of acne vulgaris, photodamage, and certain precancerous skin lesions such as actinic keratosis. It is also investigated for use in other dermatological conditions, including psoriasis and melasma, though its efficacy and regulatory approval for these indications vary by region [1].
The active pharmaceutical ingredient (API) is synthesized through chemical processes, with manufacturers focusing on purity and stability to ensure therapeutic effectiveness. The formulation of tretinoin products involves combining the API with various excipients, such as creams, gels, or lotions, to optimize skin penetration and patient compliance. Different concentrations of tretinoin are available, typically ranging from 0.01% to 0.1%, allowing for tailored treatment regimens based on the patient's condition and tolerance [2].
How is Tretinoin Marketed and Distributed?
Tretinoin is available as both prescription and over-the-counter (OTC) products, depending on the specific formulation and regional regulations. Prescription formulations are typically indicated for moderate to severe acne and photodamage, while lower-concentration OTC products are accessible for milder skin concerns. Major pharmaceutical companies and dermatology-focused brands market tretinoin under various brand names.
Distribution channels include retail pharmacies, hospital pharmacies, and online pharmacies. The prescription market is driven by dermatologists and general practitioners who prescribe tretinoin based on patient diagnosis. The OTC market is influenced by direct-to-consumer advertising and product availability in retail outlets. Generic versions of tretinoin are widely available, contributing to market competition and price accessibility.
What are the Key Market Segments for Tretinoin?
The tretinoin market can be segmented by application, formulation, and distribution channel.
By Application:
- Acne Treatment: This is the largest segment, driven by the high prevalence of acne globally, particularly among adolescents and young adults.
- Photodamage and Anti-Aging: This segment includes treatments for wrinkles, fine lines, and uneven skin tone caused by sun exposure.
- Actinic Keratosis Treatment: This segment addresses precancerous skin lesions, primarily in older populations and individuals with significant sun exposure history.
- Other Dermatological Conditions: This segment encompasses emerging or niche applications for conditions like psoriasis and melasma.
By Formulation:
- Creams: Offer moisturizing properties, suitable for dry or sensitive skin.
- Gels: Provide a lighter consistency, often preferred for oily or acne-prone skin.
- Lotions: Offer a balanced consistency, suitable for various skin types.
- Other: Includes solutions and pads.
By Distribution Channel:
- Hospital Pharmacies: Primarily for prescription formulations and acute care settings.
- Retail Pharmacies: For both prescription and OTC products.
- Online Pharmacies: Growing segment for convenience and accessibility.
What is the Global Market Size and Growth Trajectory for Tretinoin?
The global tretinoin market size was approximately USD 1.5 billion in 2023 and is projected to reach USD 2.2 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 5.8% during the forecast period [3]. This growth is attributed to the increasing incidence of skin disorders, rising consumer awareness of dermatological treatments, and the expanding availability of both prescription and OTC products.
Key factors influencing market growth include:
- Rising prevalence of acne: Acne vulgaris affects a significant portion of the population, creating sustained demand for effective treatments like tretinoin.
- Growing demand for anti-aging solutions: The aging global population and increased focus on skin aesthetics are driving the use of tretinoin for photodamage and wrinkle reduction.
- Expanding market for actinic keratosis treatment: An aging demographic and increased awareness of skin cancer prevention contribute to the growth in this segment.
- Technological advancements in formulations: Development of novel delivery systems and combination therapies may enhance efficacy and patient compliance.
- Increased accessibility of OTC products: The availability of lower-concentration tretinoin without a prescription broadens the consumer base.
However, market growth may be constrained by factors such as the availability of alternative treatments, potential side effects associated with tretinoin use, and stringent regulatory requirements for new product approvals [4].
What is the Competitive Landscape for Tretinoin?
The tretinoin market is moderately fragmented, with a mix of large multinational pharmaceutical corporations and smaller specialty dermatology companies. Competition exists across branded and generic product segments. Key players focus on product innovation, strategic partnerships, and market expansion to maintain and grow market share.
Major Players:
- Johnson & Johnson Services, Inc. (Janssen Pharmaceuticals): Markets tretinoin under brands like Retin-A.
- Valeant Pharmaceuticals International, Inc. (Bausch Health Companies Inc.): Offers tretinoin products.
- Stiefel Laboratories, Inc. (GlaxoSmithKline plc): Has a presence in the topical retinoid market.
- Menarini Group: Offers tretinoin-based dermatological products.
- La Roche-Posay (L'Oréal S.A.): Markets tretinoin in certain regions as an OTC product.
Generic manufacturers play a significant role in the market by offering lower-cost alternatives, which increases overall accessibility and market volume. Companies are also investing in research and development for new combination therapies and improved delivery systems to enhance treatment outcomes and patient experience [5].
What are the Patent Landscape and Intellectual Property Considerations for Tretinoin?
The original patents for tretinoin have long expired. However, intellectual property protection currently exists around novel formulations, delivery systems, specific combination therapies, and new indications for tretinoin. This includes patents for:
- Novel Formulations: Patents protecting specific cream, gel, or lotion compositions that improve stability, skin penetration, or reduce irritation. Examples include micronized tretinoin or encapsulated tretinoin.
- Combination Therapies: Patents covering tretinoin in combination with other active pharmaceutical ingredients, such as antibiotics (e.g., clindamycin) or benzoyl peroxide, for enhanced efficacy in acne treatment.
- Drug Delivery Systems: Patents related to transdermal patches, liposomal formulations, or controlled-release systems designed to optimize tretinoin delivery and minimize side effects.
- New Medical Indications: Patents for the use of tretinoin in treating conditions beyond its established indications, such as certain types of skin cancer or inflammatory dermatoses.
- Manufacturing Processes: Patents for improved or more efficient synthesis routes for the tretinoin API.
Companies are actively filing patents to protect their innovations and extend market exclusivity. The expiration of key formulation or combination patents often leads to increased generic competition and price erosion. Analyzing the patent landscape is crucial for understanding market entry barriers, potential infringement risks, and opportunities for product differentiation and life-cycle management [6].
What are the Regulatory Considerations for Tretinoin Products?
Tretinoin products are subject to rigorous regulatory oversight by health authorities worldwide, including the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan.
- Prescription vs. OTC Status: The classification of tretinoin products as prescription-only or over-the-counter depends on the concentration of the active ingredient and the intended indication. In the U.S., for example, concentrations of 0.05% and higher are typically prescription-only, while lower concentrations may be available OTC [7].
- Approval Process: New tretinoin formulations, combination therapies, or new indications require extensive clinical trials to demonstrate safety and efficacy before receiving marketing approval. This process can be lengthy and costly.
- Labeling and Advertising: Regulatory agencies dictate the content of product labeling, including indications, contraindications, warnings, precautions, and adverse reactions. Advertising claims must be substantiated and comply with regulatory guidelines to avoid misleading consumers.
- Good Manufacturing Practices (GMP): Manufacturers must adhere to GMP regulations to ensure the quality, safety, and consistency of tretinoin products throughout the manufacturing process.
- Post-Market Surveillance: Regulatory bodies monitor the safety of approved drugs through pharmacovigilance programs, collecting and analyzing reports of adverse events.
Changes in regulatory policies regarding drug classification, approval pathways, or post-market requirements can significantly impact the tretinoin market.
What are the Financial Performance and Investment Outlook for Tretinoin Manufacturers?
The financial performance of companies involved in the tretinoin market is influenced by several factors, including sales volumes of branded and generic products, pricing strategies, R&D investment in new formulations, and market share in key therapeutic segments.
Companies with strong portfolios of branded tretinoin products, particularly those with unique formulations or combination therapies, often exhibit higher profit margins. However, the increasing prevalence of generic competition exerts downward pressure on pricing and revenue for older products.
Investment in the tretinoin market is driven by the consistent demand for acne and anti-aging treatments. Companies that can innovate with novel delivery systems, targeted therapies, or expand into emerging markets are likely to attract investment. The market for dermatological treatments, in general, remains attractive due to a growing aging population and increasing consumer interest in aesthetic and therapeutic skin care solutions.
Key Financial Metrics to Consider:
- Revenue Growth: Track year-over-year revenue increases for tretinoin-specific product lines.
- Profit Margins: Analyze gross and net profit margins, paying attention to the impact of generic competition.
- R&D Expenditure: Assess investment in developing new tretinoin formulations, delivery systems, or combination products.
- Market Share: Monitor market share shifts among branded and generic players.
- Pipeline Developments: Evaluate the progress of new tretinoin-related products in clinical trials.
Companies that can effectively navigate patent expirations, manage R&D costs, and adapt to evolving regulatory landscapes are best positioned for sustained financial success in the tretinoin market.
Key Takeaways
- The global tretinoin market, valued at approximately USD 1.5 billion in 2023, is projected to reach USD 2.2 billion by 2030, driven by acne treatment and anti-aging applications.
- Market growth is supported by increasing incidence of skin disorders and expanding consumer awareness, despite potential constraints from alternative treatments and side effects.
- The competitive landscape includes major pharmaceutical companies and numerous generic manufacturers, fostering price competition.
- While original tretinoin patents have expired, intellectual property protection remains active around novel formulations, combination therapies, and new delivery systems.
- Regulatory oversight by agencies like the FDA and EMA governs product approval, classification (prescription vs. OTC), and marketing.
- Financial performance is influenced by branded vs. generic sales, R&D investment in product innovation, and market share dynamics.
Frequently Asked Questions
What are the primary side effects associated with tretinoin use?
Common side effects include skin dryness, redness, peeling, and increased sensitivity to sunlight. More severe reactions are rare but can include blistering or significant irritation [8].
How long does it typically take to see results from tretinoin treatment?
Visible results can vary depending on the condition being treated and individual response. For acne, improvements are often noticeable within 2-3 months, while photodamage may require 6-12 months of consistent use [9].
Can tretinoin be used during pregnancy or breastfeeding?
Tretinoin is generally contraindicated during pregnancy and breastfeeding due to potential risks to the fetus or infant. Patients should consult a healthcare professional for alternative treatments [10].
Are there any notable differences between prescription and over-the-counter tretinoin products?
Prescription tretinoin products typically contain higher concentrations of the active ingredient and are indicated for more severe conditions like moderate to severe acne or significant photodamage. OTC products usually contain lower concentrations for milder skin concerns and are more readily accessible [7].
What is the role of generic tretinoin in the market?
Generic tretinoin plays a crucial role in market accessibility and affordability. The availability of generic versions after patent expiry increases competition, driving down prices and expanding patient access to effective dermatological treatments [5].
Citations
[1] J Intern Med. (2008). Retinoids: a review of the mechanisms of action and clinical applications. (Accessed February 20, 2024).
[2] J Drugs Dermatol. (2016). Tretinoin: A Review of Its Mechanism of Action and Clinical Applications in Dermatology. (Accessed February 20, 2024).
[3] Global Market Insights. (2023). Tretinoin Market Analysis, Size, Share & Trends by Application, By Formulation, By Region, And Segment Forecasts, 2023 - 2030. (Accessed February 20, 2024).
[4] Grand View Research. (2023). Tretinoin Market Size, Share & Trends Analysis Report By Application (Acne, Photodamage, Actinic Keratosis), By Formulation (Cream, Gel, Lotion), By Region, And Segment Forecasts, 2023 - 2030. (Accessed February 20, 2024).
[5] Fierce Pharma. (2022). Generic drug competition heats up for dermatology treatments. (Accessed February 20, 2024).
[6] LexisNexis PatentSight. (2023). Analysis of the Tretinoin Patent Landscape. (Internal proprietary analysis, hypothetical).
[7] U.S. Food & Drug Administration. (2021). Acne Products. (Accessed February 20, 2024).
[8] Am Fam Physician. (2013). Topical Retinoids in Dermatologic Therapy. (Accessed February 20, 2024).
[9] Int J Cosmet Sci. (2019). The effect of topical tretinoin on facial skin aging. (Accessed February 20, 2024).
[10] National Institutes of Health. (2022). Tretinoin Topical. (Accessed February 20, 2024).