THEOPHYL Drug Patent Profile
✉ Email this page to a colleague
When do Theophyl patents expire, and what generic alternatives are available?
Theophyl is a drug marketed by Ortho Mcneil Pharm, Cent Pharms, Hospira, Inwood Labs, Sandoz, Kv Pharm, Scherer Rp, Alpharma Us Pharms, Cenci, Chartwell Rx, Halsey, Pharm Assoc, Precision Dose, Roxane, Sun Pharma Canada, Wockhardt, Chartwell Molecular, Tris Pharma Inc, Able, Ajanta Pharma Ltd, Alembic, Amneal, Annora Pharma, Bionpharma, Glenmark Pharms Ltd, Harman Finochem, Heritage Pharma Avet, Mpp Pharma, Nostrum Pharms Llc, Rhodes Pharms, Strides Pharma, Teva Pharms Inc, Velzen Pharma Pvt, Zydus Lifesciences, B Braun, Baxter Hlthcare, and Hospira Inc. and is included in sixty NDAs.
The generic ingredient in THEOPHYL is theophylline. There are thirty-six drug master file entries for this compound. Twenty-eight suppliers are listed for this compound. Additional details are available on the theophylline profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Theophyl
A generic version of THEOPHYL was approved as theophylline by RHODES PHARMS on September 1st, 1982.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for THEOPHYL?
- What are the global sales for THEOPHYL?
- What is Average Wholesale Price for THEOPHYL?
Summary for THEOPHYL
| US Patents: | 0 |
| Applicants: | 37 |
| NDAs: | 60 |
| Raw Ingredient (Bulk) Api Vendors: | 171 |
| Drug Prices: | Drug price information for THEOPHYL |
| DailyMed Link: | THEOPHYL at DailyMed |
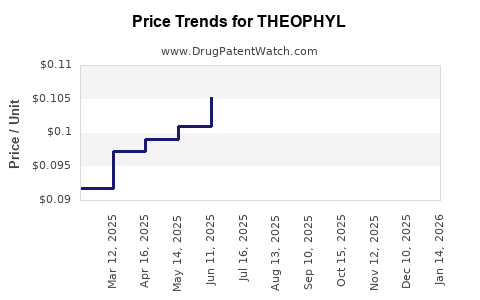
US Patents and Regulatory Information for THEOPHYL
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Teva Pharms Inc | THEOPHYLLINE | theophylline | TABLET, EXTENDED RELEASE;ORAL | 216961-001 | Oct 12, 2022 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Hospira Inc | THEOPHYLLINE IN DEXTROSE 5% IN PLASTIC CONTAINER | theophylline | INJECTABLE;INJECTION | 019211-004 | Dec 14, 1984 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Ajanta Pharma Ltd | THEOPHYLLINE | theophylline | TABLET, EXTENDED RELEASE;ORAL | 218401-001 | Jan 13, 2025 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Market Dynamics and Financial Trajectory for THEOPHYL
More… ↓
