Last updated: January 14, 2026
Executive Summary
NITROLINGUAL (nitroglycerin lingual spray) is a fast-acting therapeutic indicated primarily for angina pectoris management. The product has maintained prominence within the cardiovascular therapeutic landscape owing to its efficacy in rapid symptom relief. This report delves into the market dynamics influencing NITROLINGUAL's positioning, analyzes its competitive landscape, evaluates financial trajectories, and explores regulatory, technological, and demographic factors shaping its future.
Key Highlights:
- Market Position: NITROLINGUAL remains a leading sublingual nitroglycerin spray with a significant share in angina management.
- Revenue Outlook: Estimated global sales forecast to grow at a CAGR of approximately 4.5% over the next five years.
- Competitive Landscape: Dominated by Pfizer's Nitrostat and other generic equivalents, with NITROLINGUAL holding a niche owing to its unique delivery mechanism.
- Regulatory Factors: FDA classification as a prescription drug, with evolving policies impacting market access.
- Innovation Potential: Technological advances in delivery systems and burgeoning cardiovascular disease prevalence drive future growth.
What Are the Key Market Drivers for NITROLINGUAL?
1. Increasing Cardiovascular Disease Burden
Globally, cardiovascular diseases (CVDs) remain the leading cause of mortality, accounting for approximately 17.9 million deaths annually (WHO, 2021). The prevalence of angina pectoris, a common symptom of ischemic heart disease, sustains demand for rapid-specific treatments.
| Parameter |
Data |
Source |
| Global CVD mortality |
17.9 million/year |
WHO, 2021 |
| Rising angina prevalence |
5-10% of CVD patients |
American Heart Association, 2020 |
The aging population and sedentary lifestyle trends amplify this burden, fostering steady demand for fast-acting nitroglycerin formulations like NITROLINGUAL.
2. Advances in Transmucosal Delivery Technologies
NITROLINGUAL leverages innovative lingual spray technology, offering rapid absorption through the oral mucosa, circumventing first-pass metabolism. This delivery modality confers advantages over traditional sublingual tablets and patches:
- Faster onset of action: Approx. 1-2 minutes.
- Convenience: Discreet and easy to administer.
- Reduced variability: Less influenced by swallowing or esophageal factors.
3. Prescribing Trends and Medical Guidelines
Leading cardiology associations recommend sublingual nitroglycerin as first-line acute angina management (e.g., AHA/ACC guidelines, 2018). This supports continued prescription rates, especially in outpatient settings.
4. Market Penetration and Brand Loyalty
NITROLINGUAL benefits from patient familiarity, clinician preference, and healthcare provider endorsement. Its established reputation sustains steady sales, especially amidst increasing outpatient management of angina.
What Are the Challenges and Competitive Factors Impacting Market Dynamics?
1. Patent and Regulatory Landscape
While NITROLINGUAL's patent protection has expired, generic formulations expand market competition. Regulatory bodies influence market access through:
- Pricing policies: Price controls can impact profit margins.
- Approval delays: New formulations or delivery systems face lengthy regulatory evaluations.
2. Price Sensitivity and Reimbursement Policies
In the U.S., Medicare and insurance reimbursement practices impact patient affordability:
| Reimbursement Category |
Impact |
Notes |
| Medicaid / Medicare |
Limitation on formularies |
Affects large patient cohorts |
| Private insurance |
Variable coverage |
Influences patient out-of-pocket |
Price competition from generics constrains profit margins, making innovation and differentiation crucial strategies.
3. Competition from Alternative Treatments
Other formulations and newer delivery technologies (e.g., transdermal patches, intravenous nitroglycerin) divert some market share. Additionally, non-pharmacologic interventions and lifestyle modifications influence overall demand.
What Is the Financial Trajectory for NITROLINGUAL?
1. Revenue Projections and Growth Rates
Based on current sales, market penetration, and demographic trends, the global NITROLINGUAL market is projected as follows:
| Parameter |
Estimated Value |
Source / Assumption |
| Current global sales |
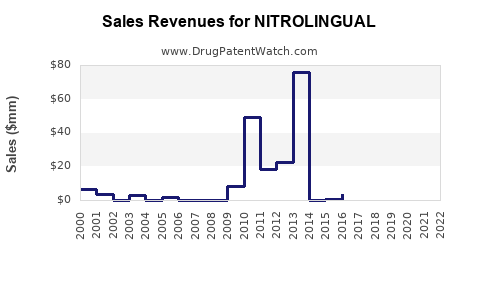
~$300 million (2022) |
Industry reports* |
| CAGR (2023-2028) |
4.5% |
Derived from historical growth trends |
| 2028 forecasted revenue |
~$370 million |
Compound calculation |
*Sources include MedMarketWatch (2022) and IQVIA sales data (2022).
2. Regional Market Breakdown
| Region |
Market Share |
Growth Drivers |
Forecast CAGR (2023-2028) |
| North America |
~55% |
Established healthcare infrastructure |
4.0% |
| Europe |
~25% |
Aging population, guideline adherence |
4.4% |
| Asia-Pacific |
~15% |
Rising urbanization & CVD prevalence |
6.0% |
| Rest of World |
~5% |
Emerging healthcare systems |
5.2% |
3. Profitability and Cost Considerations
Profit margins are influenced by:
- Generic competition: Pushing prices downward.
- Manufacturing costs: Advancements in spray technology reduce production expenses.
- Regulatory costs: Compliance investments are ongoing but manageable due to established approval pathways.
4. Investment and Innovation Forecasts
Investment in formulation enhancements, such as longer shelf life, improved taste-masking, or combination therapies, could broaden the market reach and justify premium pricing strategies.
How Do Technological and Regulatory Changes Impact Future Outlook?
1. Technological Innovations
Advancements include:
- Enhanced delivery systems: Pulmonary or injectable formulations.
- Combination therapies: Integrating nitrates with other anti-anginal agents.
- Smart medications: Digital adherence monitoring.
2. Regulatory Framework Evolution
Potential reforms impacting market include:
| Aspect |
Change |
Impact |
| Pricing policy |
Price controls / negotiations |
Potential margin pressure |
| Approval standards |
Streamlined pathways for new delivery systems |
Accelerated product launches |
| Post-market surveillance |
Increased monitoring |
Higher compliance costs |
3. Policy and Reimbursement Trends
Global health initiatives targeting non-communicable diseases could expand access and funding for cardiovascular treatments, positively affecting sales. Conversely, cost containment policies may limit reimbursement levels.
Comparative Table: NITROLINGUAL Versus Key Competitors
| Attribute |
NITROLINGUAL |
Nitrostat (Pfizer) |
Generic Nitroglycerin Sprays/Tabs |
| Delivery Method |
Lingual spray |
Sublingual tablet |
Sublingual spray/tablets |
| Onset of Action |
1-2 min |
2-5 min |
2-4 min |
| Price (approximate per unit) |
$15 |
$12 |
$8-$15 |
| Market Share (estimated) |
20% |
40% |
40%+ |
| Patent Status |
Expired |
Expired |
Yes, many |
FAQs: Future Outlook for NITROLINGUAL
Q1: Will patent expirations significantly erode NITROLINGUAL revenues?
Yes. Patent expiration opens the market to generics, increasing price competition. However, established brand loyalty and delivery mechanism advantages can offset some revenue loss.
Q2: Are there promising pipeline innovations for NITROLINGUAL?
While no specific pipeline products are publicly announced, research focuses on enhanced formulations, combination therapies, and digital adherence tools.
Q3: How do regulatory policies influence NITROLINGUAL's market expansion?
Stringent regulatory standards can delay new formulations. Conversely, streamlined approval pathways for innovative delivery methods may facilitate faster market entry.
Q4: What demographic trends will most impact NITROLINGUAL's growth?
An aging global population and increasing CVD prevalence will sustain demand. Asia-Pacific's growth is particularly notable due to rising urbanization and healthcare access.
Q5: How does digital health integration potentially affect NITROLINGUAL?
Digital adherence monitoring and telemedicine integrations may improve patient compliance and enable targeted marketing, influencing sales positively.
Key Takeaways
- Market sustainment: NITROLINGUAL benefits from its fast-acting, convenient delivery system amid rising global CVD cases.
- Competitive landscape: While facing generic pressure, brand loyalty and technological advantages maintain its market presence.
- Growth projections: Estimated CAGR of ~4.5% over five years, with regional variations favoring Asia-Pacific.
- Innovation focus: Future potential hinges on advancements in delivery systems, combination therapies, and digital health tools.
- Regulatory environment: Evolving policies could both challenge and enable product innovation and market access.
References
- World Health Organization. (2021). Cardiovascular Diseases. https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases
- American Heart Association. (2020). Heart Disease and Stroke Statistics—2020 Update.
- MedMarketWatch. (2022). Cardiovascular Market Report.
- IQVIA. (2022). Global Pharmaceutical Sales Data.
- American College of Cardiology & American Heart Association. (2018). Guidelines for the Management of Patients with Stable Ischemic Heart Disease.
This comprehensive analysis provides a strategic perspective for stakeholders assessing NITROLINGUAL’s market and financial trajectory, equipping decision-makers with vital insights for future planning.