Share This Page
Drug Sales Trends for NITROLINGUAL
✉ Email this page to a colleague
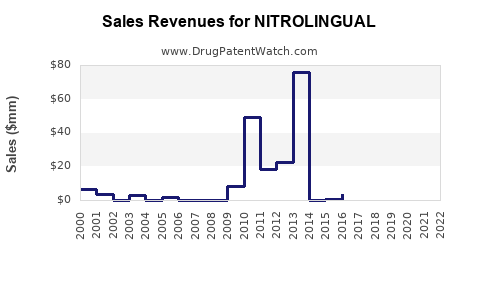
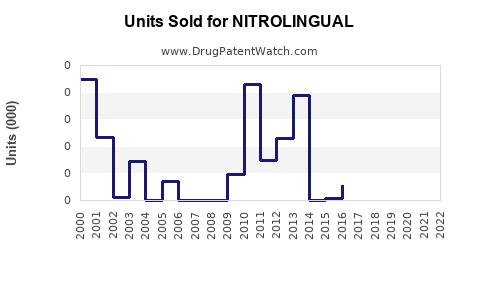
Annual Sales Revenues and Units Sold for NITROLINGUAL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| NITROLINGUAL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| NITROLINGUAL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| NITROLINGUAL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| NITROLINGUAL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Nitrolingual: Market Trajectory and Sales Forecast
Nitrolingual, a sublingual nitroglycerin spray for acute relief of angina pectoris, faces a mature market with established competitors. Its future sales performance will be influenced by generic competition, evolving treatment guidelines for cardiovascular disease, and the potential for market penetration in underserved patient populations.
What is Nitrolingual's Current Market Position?
Nitrolingual (nitroglycerin) is an established medication for the symptomatic treatment of angina pectoris, a condition characterized by chest pain due to insufficient blood flow to the heart muscle. The drug delivers nitroglycerin directly to the bloodstream via sublingual absorption, offering rapid onset of action.
- Active Ingredient: Nitroglycerin
- Mechanism of Action: Vasodilation, primarily through relaxation of vascular smooth muscle, leading to reduced preload and afterload on the heart, and improved coronary blood flow.
- Indication: Acute relief of an attack or prevention of angina pectoris.
- Formulation: Sublingual spray.
The market for acute angina relief is largely occupied by generic nitroglycerin products in various formulations, including sublingual tablets and sprays. Nitrolingual, while a branded product, competes directly with these generics on price and accessibility. The prevalence of cardiovascular disease, particularly angina, remains significant globally, providing a consistent demand for effective relief medications. However, the advent of newer therapeutic classes for chronic angina management and preventative cardiovascular therapies has altered the landscape, shifting focus towards disease modification rather than solely symptomatic relief of acute events.
What are the Key Drivers of Nitrolingual Sales?
Nitrolingual's sales are primarily driven by the immediate need for acute angina relief in patients diagnosed with this condition. The sublingual spray formulation offers a distinct advantage in terms of speed of administration and absorption compared to some other delivery methods.
- Patient Population: Individuals experiencing acute anginal episodes, including those with stable and unstable angina.
- Convenience of Delivery: The spray format is perceived as easy to use and less prone to degradation compared to some older sublingual tablet formulations, particularly in terms of dosing accuracy and stability [1].
- Rapid Onset of Action: The sublingual route bypasses first-pass metabolism in the liver, allowing for swift entry into systemic circulation and rapid symptom relief, typically within minutes.
- Physician Prescribing Habits: Continued physician familiarity and preference for established treatment protocols for acute angina contribute to sustained prescription volumes.
The market is also influenced by the increasing incidence of cardiovascular diseases, linked to aging populations and lifestyle factors such as obesity and sedentary behavior. This demographic trend underpins a sustained demand for treatments that can rapidly alleviate anginal symptoms.
What are the Primary Challenges Facing Nitrolingual?
Nitrolingual faces several significant challenges that will impact its future sales trajectory, including intense generic competition, evolving treatment paradigms, and pricing pressures.
- Generic Competition: The expiration of key patents for nitroglycerin has led to the widespread availability of generic sublingual tablets and sprays. These generic alternatives are typically offered at significantly lower price points, eroding the market share and price premium of branded products like Nitrolingual.
- Evolving Treatment Guidelines: Current cardiovascular guidelines increasingly emphasize disease modification and prevention of ischemic events through medications such as beta-blockers, calcium channel blockers, antiplatelets, and statins. While rapid-acting nitrates remain crucial for acute symptom relief, their role in overall cardiovascular management has been contextualized within a broader therapeutic strategy.
- Alternative Therapies: The development of novel anti-anginal agents and the wider adoption of interventional cardiology procedures (e.g., angioplasty and stenting) can reduce the reliance on or frequency of acute nitrate use in some patient subsets.
- Healthcare System Cost Containment: Payers and healthcare systems are increasingly focused on cost-effectiveness, which favors generic medications and may lead to formulary restrictions or prior authorization requirements for branded products.
The therapeutic landscape for cardiovascular disease is dynamic, with ongoing research into new drug classes and treatment approaches. This continuous innovation presents a challenge for older, established medications that primarily address symptomatic relief without disease modification.
What is the Competitive Landscape for Nitrolingual?
Nitrolingual operates within a competitive market dominated by generic nitroglycerin products. The primary differentiation for branded spray formulations lies in their convenience, consistent dosing, and perceived stability.
Key Competitors:
- Generic Nitroglycerin Sublingual Tablets: These are the most prevalent and cost-effective alternatives. Brands are less significant than the active ingredient and its availability as a generic.
- Other Nitroglycerin Sublingual Sprays (Generic): Multiple manufacturers offer generic versions of nitroglycerin spray, directly competing with Nitrolingual on price.
- Other Rapid-Acting Nitrates: While less common, other nitrate formulations or drugs with similar immediate vasodilatory effects could be considered by some prescribers.
Comparison Table: Nitrolingual vs. Generic Sublingual Tablets
| Feature | Nitrolingual | Generic Nitroglycerin Sublingual Tablets |
|---|---|---|
| Formulation | Sublingual spray | Sublingual tablet |
| Onset of Action | Rapid (minutes) | Rapid (minutes) |
| Dosing | Metered dose spray | Tablet count |
| Convenience | High (easy to administer, portable) | Moderate (requires counting, potential for dissolution issues) |
| Cost | Higher (branded product) | Lower (generic) |
| Stability | Generally considered stable if stored properly | Can be susceptible to degradation from moisture/heat |
| Market Share | Niche within the broader nitrate market | Dominant by volume and value |
The market is highly price-sensitive, and the availability of inexpensive generic tablets means that branded sprays must demonstrate significant clinical or convenience advantages to command a premium.
What are the Projected Sales and Market Growth for Nitrolingual?
Projecting sales for Nitrolingual requires consideration of multiple factors, including the entrenched generic competition and the evolving treatment landscape for angina. The market for acute angina relief is relatively stable but not experiencing significant growth.
Global Market Size for Acute Angina Relief (Estimated):
- The overall market for acute angina relief, encompassing all nitrate formulations and related products, is estimated to be in the range of \$1.5 billion to \$2.0 billion globally.
- Nitroglycerin, as the primary active ingredient, accounts for a substantial portion of this market.
Nitrolingual Specific Projections:
- Current Annual Sales (Estimated): \$50 million - \$75 million (global, for the branded product). This figure can vary based on specific market reporting and product lifecycle stage.
- Projected Annual Sales (5-Year Outlook): A slight decline or stagnation is anticipated.
- Year 1-2: -2% to 0% growth per annum.
- Year 3-5: -1% to -3% growth per annum.
Factors Influencing Projections:
- Erosion from Generics: Continued price pressure from generic nitroglycerin sprays and tablets will cap any potential price-driven growth.
- Market Penetration: While the spray format offers convenience, its adoption is limited by cost compared to generics, especially in cost-sensitive healthcare systems.
- Therapeutic Shift: The increasing focus on disease modification over solely symptomatic relief may lead to a gradual decrease in the overall demand for acute nitrates as first-line therapy for some patients.
- Limited New Indications: Nitrolingual is unlikely to see significant new indications approved, limiting its expansion potential.
The market is expected to remain stable in terms of patient numbers requiring acute relief, but the revenue generated by branded Nitrolingual will likely face downward pressure due to the dominance of generic alternatives.
What is the Regulatory and Patent Landscape for Nitrolingual?
Nitrolingual, as a pharmaceutical product, is subject to rigorous regulatory oversight and intellectual property protection. However, the core active ingredient, nitroglycerin, has long been off-patent.
- Active Ingredient Patent Status: Nitroglycerin itself is a well-established and long-off-patent compound.
- Formulation Patents: Patents related to the specific sublingual spray formulation, device technology, or manufacturing processes may exist. However, the expiry of these formulation patents is a critical factor in the entry of generic competitors. The original patents protecting Nitrolingual's specific formulation and delivery system have largely expired or are nearing expiry in key markets.
- Regulatory Approvals: Nitrolingual requires marketing authorization from regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- Exclusivity Periods: The branded product likely benefited from periods of market exclusivity following its initial approval, but these have expired in most major regions.
- Generic Entry: The expiry of formulation and manufacturing patents, or the successful navigation of patent challenges by generic manufacturers, has led to the availability of generic versions of nitroglycerin sublingual spray. This is the primary driver of the projected sales decline for the branded product.
Manufacturers of generic nitroglycerin sprays aim to replicate the efficacy and safety profile of the innovator product while offering a lower-cost alternative. The regulatory pathway for generic drug approval focuses on demonstrating bioequivalence.
What are the Opportunities for Nitrolingual?
Despite the challenges, limited opportunities exist for Nitrolingual to maintain or marginally grow its market presence. These opportunities are largely niche and dependent on specific market dynamics.
- Underserved Patient Populations:
- Elderly Patients: For whom the convenience of a spray may be particularly beneficial compared to manipulating small tablets.
- Patients with Dexterity Issues: Individuals with conditions like arthritis who find it difficult to handle tablets.
- Patients in Remote or Resource-Limited Settings: Where consistent storage and administration of tablets might be more challenging.
- Improved Patient Education and Adherence Programs: Enhancing awareness among patients and caregivers about the benefits of the spray formulation for rapid relief could sustain demand.
- Partnerships with Healthcare Providers: Collaborating with cardiology practices to emphasize the specific advantages of the spray for acute symptom management.
- Market Access in Emerging Economies: While price is a barrier, in markets where generic options are less prevalent or consistent, a branded product with established quality may find a foothold, albeit at a lower price point than in developed markets.
These opportunities do not suggest a significant reversal of the overall market trend but rather strategies to preserve existing market share against generic erosion.
Key Takeaways
Nitrolingual operates in a mature market for acute angina relief, facing significant pressure from generic nitroglycerin products. Its sublingual spray formulation offers convenience and rapid action, but this is increasingly offset by the cost advantage of generic alternatives. While the prevalence of cardiovascular disease supports a baseline demand, evolving treatment guidelines emphasize disease modification, contextualizing the role of acute nitrate therapy. Projected sales for Nitrolingual are expected to stagnate or decline modestly due to generic competition and limited opportunities for substantial market expansion. Regulatory hurdles for generic entry are largely overcome due to the expiry of key formulation patents. Opportunities lie in targeting specific patient demographics with dexterity issues or seeking market access in emerging economies.
FAQs
-
What is the primary difference between Nitrolingual and generic nitroglycerin tablets? Nitrolingual is a sublingual spray formulation, designed for rapid absorption and ease of administration. Generic nitroglycerin tablets are traditional solid dosage forms administered under the tongue. While both deliver nitroglycerin for acute angina relief, the spray offers greater convenience and potentially more consistent dosing.
-
Will new patents extend Nitrolingual's market exclusivity? The active pharmaceutical ingredient, nitroglycerin, is off-patent. Any remaining patent protection for Nitrolingual would pertain to specific formulation, device, or manufacturing processes. The expiry of these secondary patents has enabled generic manufacturers to enter the market with their own nitroglycerin spray products.
-
How does the market for acute angina relief compare to the market for chronic angina management? The market for acute angina relief, where Nitrolingual competes, focuses on immediate symptom management of anginal episodes. The market for chronic angina management involves therapies aimed at preventing angina attacks, improving exercise tolerance, and modifying disease progression (e.g., beta-blockers, calcium channel blockers, antiplatelets, statins). These are distinct therapeutic areas with different treatment goals and competitive dynamics.
-
What is the projected impact of new cardiovascular therapies on Nitrolingual sales? The development and adoption of new therapies for cardiovascular disease, particularly those focusing on disease modification and prevention of ischemic events, may indirectly impact Nitrolingual sales. As these newer therapies become more prevalent, the reliance on symptomatic relief for acute angina might shift within the overall treatment paradigm, potentially leading to a gradual decrease in the volume of acute nitrate prescriptions.
-
Are there any off-label uses for Nitrolingual that could drive future sales? Nitrolingual is approved and indicated solely for the acute relief of angina pectoris. Off-label uses are not officially supported by regulatory bodies and would not be a basis for reliable sales projections or market analysis. Prescribing for unapproved indications carries regulatory and liability risks.
Citations
[1] N. B. T. H. Committee, The British National Formulary (BNF) (Vol. 85). Pharmaceutical Press, 2023.
More… ↓
