Last updated: February 19, 2026
Nifedipine, a dihydropyridine calcium channel blocker, continues to hold a significant position in the cardiovascular therapeutics market, primarily for managing hypertension and angina pectoris. Its established efficacy and broad availability contribute to consistent demand, though facing increasing competition from newer drug classes and generics.
What are Nifedipine's Primary Therapeutic Indications?
Nifedipine is indicated for the treatment of:
- Hypertension: It is used as monotherapy or in combination with other antihypertensive agents to lower blood pressure.
- Angina Pectoris: It is effective in managing chronic stable angina and vasospastic (Prinzmetal's) angina by reducing myocardial oxygen demand and increasing oxygen supply.
Off-label uses include the treatment of Raynaud's phenomenon and preterm labor, though these are not its primary market drivers.
What is the Global Market Size and Projected Growth for Nifedipine?
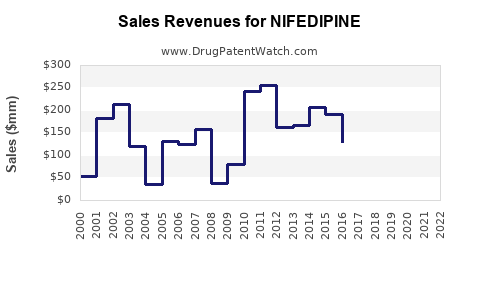
The global nifedipine market is a mature segment within the broader cardiovascular drug market. Exact figures for nifedipine alone are often aggregated within broader calcium channel blocker or antihypertensive drug categories. However, analysis of these segments indicates a stable, albeit modest, growth trajectory.
- Market Value: While precise current figures are proprietary, the global market for calcium channel blockers, which nifedipine is a part of, was valued in the billions of dollars in recent years. Projections suggest continued growth, driven by the aging global population and the increasing prevalence of cardiovascular diseases.
- Projected CAGR: For the broader antihypertensive drug market, Compound Annual Growth Rates (CAGRs) are typically estimated in the range of 4% to 6% over the next five to seven years. Nifedipine's share within this market is expected to grow at a lower rate, likely between 1% and 3%, due to its generic status and competition.
Who are the Key Manufacturers and Generics Suppliers of Nifedipine?
The production and supply of nifedipine are characterized by a significant number of generic manufacturers, particularly following patent expirations of originator products.
Major Branded Nifedipine Products (Historical and Current):
- Procardia (Pfizer): A historically significant branded formulation, now largely off-patent.
- Adalat (Bayer): Another key originator brand with extensive global market presence.
- Nifedipine XL/ER Formulations: Extended-release formulations remain important for improved patient compliance and efficacy.
Key Generic Manufacturers and Suppliers:
The market is highly fragmented with numerous generic players. Prominent entities involved in the production and distribution of nifedipine API (Active Pharmaceutical Ingredient) and finished dosage forms include:
- Teva Pharmaceutical Industries
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Cipla Ltd.
- Lupin Ltd.
- Aurobindo Pharma
- Novartis AG (through Sandoz division)
These companies compete primarily on price and market access in various regions.
What is the Patent Landscape for Nifedipine?
Original patents for nifedipine have long expired. The composition of matter patents were granted in the 1970s.
- Initial Patents: Composition of matter patents for nifedipine expired in the late 1990s to early 2000s, depending on the jurisdiction and any extensions.
- Formulation and Method of Use Patents: While the core molecule is off-patent, companies have historically pursued patents on novel formulations (e.g., extended-release), delivery systems, or specific therapeutic uses. However, the expiration of these secondary patents has further opened the market to generics.
- Generic Entry: Widespread generic entry occurred globally following the expiration of primary patents, leading to significant price erosion.
How Have Regulatory Approvals and Market Exclusivities Shaped Nifedipine's Market Position?
Regulatory approvals have historically defined nifedipine's market presence. As an older drug, it has undergone extensive regulatory review and has established dossiers with major health authorities.
- FDA Approval: Nifedipine received its first FDA approval in 1981.
- EMA Approval: It has also been approved by the European Medicines Agency (EMA) and numerous other national regulatory bodies.
- Generic Approvals: Regulatory agencies worldwide have approved numerous Abbreviated New Drug Applications (ANDAs) for generic nifedipine, reflecting its generic status.
- Market Exclusivity: Any remaining market exclusivity for specific formulations or indications has largely expired, allowing for broad generic competition.
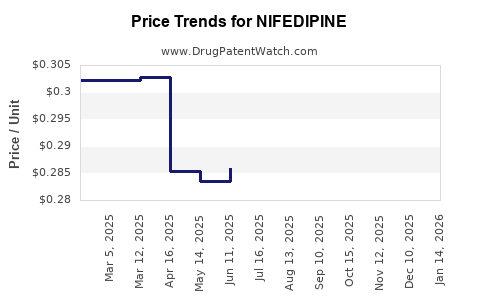
What are the Pricing Dynamics and Revenue Streams for Nifedipine?
Pricing for nifedipine is predominantly driven by generic competition, leading to lower per-unit costs compared to newer therapies.
- Price Erosion: The introduction of generic versions has led to significant price erosion, particularly in developed markets.
- Volume-Driven Revenue: Manufacturers of generic nifedipine generate revenue primarily through high sales volumes.
- Contract Manufacturing and API Sales: Many companies that produce nifedipine also engage in contract manufacturing for other pharmaceutical entities or sell nifedipine API to formulators.
- Regional Pricing Variations: Pricing varies considerably by region, influenced by local regulatory policies, healthcare systems, and the competitive intensity of generic players in that market. For instance, prices in emerging markets may differ from those in North America or Europe.
What is the Competitive Landscape for Nifedipine?
Nifedipine faces competition from several fronts:
- Other Dihydropyridine Calcium Channel Blockers: Amlodipine, felodipine, and nifedipine itself compete within this class, often differentiated by pharmacokinetic profiles and dosing regimens.
- Non-Dihydropyridine Calcium Channel Blockers: Verapamil and diltiazem are also used for hypertension and angina but have different mechanisms and side effect profiles.
- Other Antihypertensive Drug Classes: Angiotensin-converting enzyme (ACE) inhibitors, angiotensin II receptor blockers (ARBs), beta-blockers, and diuretics represent major therapeutic alternatives. These classes often offer different cardiovascular protective benefits and have varying efficacy in specific patient populations.
- Combination Therapies: Many patients are treated with combination therapies, where nifedipine might be one component, but the overall treatment efficacy is driven by multiple drug classes.
- Newer Agents: While not direct competitors in all cases, newer agents with novel mechanisms of action or improved cardiovascular outcomes are also vying for market share in the management of cardiovascular conditions.
What are the Key Market Trends and Future Outlook for Nifedipine?
The future outlook for nifedipine is characterized by stability and continued generic competition.
- Aging Population and Disease Prevalence: The persistent rise in cardiovascular diseases and an aging global population will maintain a baseline demand for effective antihypertensive and anti-anginal medications, including nifedipine.
- Cost-Effectiveness: As a mature, generic drug, nifedipine remains a cost-effective option, particularly favored in healthcare systems with budget constraints and in emerging markets.
- Shift to Newer Therapies: In developed markets, there is a gradual trend towards newer drug classes offering potentially better tolerability, broader cardiovascular protection, or simplified dosing.
- Focus on Formulations: Continued innovation might focus on improved nifedipine formulations for enhanced patient compliance or specific therapeutic niches, though major breakthroughs are unlikely given its maturity.
- Market Saturation: The market for nifedipine is largely saturated with generic options, leading to intense price competition and limiting significant growth potential.
Key Takeaways
Nifedipine remains a foundational therapy for hypertension and angina, supported by a well-established efficacy profile and cost-effectiveness. The market is characterized by mature patent protection, leading to a highly competitive generic landscape. While demand is sustained by rising cardiovascular disease prevalence and an aging population, growth is tempered by the availability of numerous generic alternatives and the ongoing shift towards newer therapeutic classes. Revenue generation for nifedipine producers is largely volume-driven, with pricing heavily influenced by generic competition and regional market dynamics.
Frequently Asked Questions
1. What is the primary reason for nifedipine's continued market presence despite being an older drug?
Nifedipine's continued market presence is primarily due to its established efficacy, broad regulatory approvals, and its status as a cost-effective generic medication, making it an accessible treatment option globally.
2. How does nifedipine's patent status impact its market competitiveness?
The expiration of nifedipine's primary patents has led to widespread generic entry, significantly increasing competition and driving down prices, which is a defining characteristic of its current market competitiveness.
3. What is the impact of emerging markets on the nifedipine market?
Emerging markets contribute significantly to nifedipine demand due to their growing populations, increasing prevalence of cardiovascular diseases, and a greater reliance on cost-effective generic medications.
4. Are there any significant research and development efforts currently focused on nifedipine?
Current R&D efforts concerning nifedipine are generally limited to developing improved formulations for enhanced patient adherence or exploring its use in combination therapies, rather than novel molecular entities.
5. What are the main challenges faced by manufacturers in the nifedipine market?
Manufacturers face challenges including intense price competition from numerous generic suppliers, limited potential for market differentiation, and the gradual shift of healthcare providers and patients towards newer therapeutic agents.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases (General source for approval dates and information)
[2] European Medicines Agency. (n.d.). Human Medicines. Retrieved from https://www.ema.europa.eu/en/human-medicines (General source for EMA approval information)
[3] Market Research Reports (Various Publishers: e.g., Grand View Research, Mordor Intelligence, Allied Market Research). (Recent Years). Global Antihypertensive Drugs Market Analysis and Forecasts. (Specific report titles and publishers vary, but these are common sources for market size and CAGR data).
[4] Pharmaceutical Industry Databases (e.g., EvaluatePharma, Cortellis). (Ongoing). Drug Lifecycles and Patent Expirations. (Proprietary databases providing detailed patent information and market exclusivity timelines).