Last updated: February 19, 2026
Morphine sulfate is a foundational opioid analgesic with persistent market demand driven by its efficacy in managing moderate to severe pain, particularly in palliative care and post-operative settings. The market is characterized by established generic competition, stringent regulatory oversight, and an ongoing focus on abuse deterrence and responsible prescribing. Financial performance is largely tied to volume-driven sales and efficient manufacturing, with limited scope for significant price escalation due to the mature nature of the product and the availability of lower-cost alternatives.
What is the Current Market Size and Growth Projection for Morphine Sulfate?
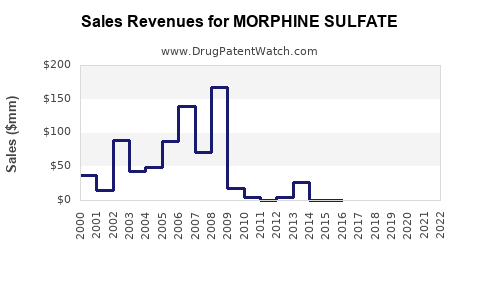
The global market for morphine sulfate is substantial, though precise figures are challenging to isolate due to its classification as a Schedule II controlled substance and its inclusion within broader analgesic market data. Industry estimates suggest the global analgesic market, which morphine sulfate contributes to significantly, is valued in the tens of billions of dollars. Projections for the broader opioid segment, including morphine, indicate a modest but stable growth rate, typically in the low single digits (2-4% annually). This growth is sustained by an aging global population, increasing incidence of chronic pain conditions, and its indispensable role in end-of-life care.
Who are the Key Manufacturers and Suppliers of Morphine Sulfate?
The manufacturing landscape for morphine sulfate is consolidated, with a few major players dominating global supply. These companies often possess vertically integrated operations, controlling the extraction of opium from poppy straw through to the synthesis and formulation of the final drug product. Key global manufacturers include:
- Insys Therapeutics (now part of Mallinckrodt Pharmaceuticals): Historically a significant producer.
- Mallinckrodt Pharmaceuticals: A major supplier with a long history in opioid production.
- Purdue Pharma (though facing significant legal challenges and bankruptcy proceedings): A historical leader in opioid production.
- Teva Pharmaceutical Industries: A large generic pharmaceutical manufacturer with morphine sulfate in its portfolio.
- Pfizer Inc.: Offers various formulations of morphine sulfate.
- Abbott Laboratories: Provides morphine sulfate products.
- Fresenius Kabi: Specializes in injectable generics, including morphine sulfate.
These manufacturers operate under strict quotas and regulations imposed by national drug enforcement agencies, such as the U.S. Drug Enforcement Administration (DEA).
What are the Primary Applications and Therapeutic Areas for Morphine Sulfate?
Morphine sulfate's primary application is as a potent analgesic. Its therapeutic uses are extensive and critical in various medical settings:
- Cancer Pain Management: A cornerstone for managing severe pain associated with advanced cancer.
- Post-Operative Pain Relief: Widely used for pain management following major surgery.
- Trauma and Injury Pain: Effective for acute, severe pain resulting from significant injuries.
- Palliative and Hospice Care: Essential for ensuring comfort and quality of life for patients with terminal illnesses.
- Myocardial Infarction (Heart Attack): Used to alleviate chest pain and reduce anxiety during a heart attack.
- Pulmonary Edema: Can be used to reduce preload in certain cases of acute pulmonary edema by inducing venodilation.
Formulations include oral tablets and solutions, immediate-release and extended-release preparations, and intravenous, intramuscular, and subcutaneous injections.
What is the Regulatory Landscape Governing Morphine Sulfate Production and Distribution?
Morphine sulfate is a Schedule II controlled substance in the United States, regulated by the DEA. This classification signifies a high potential for abuse but also medical utility. International regulation is overseen by bodies like the International Narcotics Control Board (INCB), which sets global quotas for narcotic drug production and distribution. Key regulatory aspects include:
- Manufacturing Quotas: Annual limits on the total amount of morphine sulfate that can be manufactured, set by national authorities.
- Licensing and Registration: Manufacturers, distributors, and dispensers must be licensed and registered with relevant regulatory bodies.
- Record Keeping and Reporting: Strict requirements for tracking the manufacturing, distribution, and dispensing of the drug.
- Security: Robust physical security measures are mandated for facilities storing controlled substances to prevent diversion.
- Abuse Deterrence: Increasing pressure to develop and utilize abuse-deterrent formulations (ADFs) in response to the opioid crisis. The U.S. Food and Drug Administration (FDA) has guidance on developing ADFs.
What are the Key Market Trends and Challenges Affecting Morphine Sulfate?
Several trends and challenges shape the morphine sulfate market:
- Opioid Crisis and Public Scrutiny: The ongoing opioid epidemic has led to increased regulatory scrutiny, litigation against manufacturers, and a push for reduced opioid prescribing. This impacts market dynamics, emphasizing abuse deterrence and alternative pain management strategies.
- Abuse-Deterrent Formulations (ADFs): Development and adoption of ADFs are a significant trend. These formulations are designed to make it harder to misuse the drug through chewing, crushing, or injecting.
- Generic Competition: As a long-established drug, morphine sulfate faces intense generic competition, leading to price erosion and pressure on profit margins for manufacturers.
- Supply Chain Security and Diversion Prevention: Ensuring the security of the supply chain and preventing diversion into illicit channels is paramount and requires continuous investment in tracking and security technologies.
- Shift Towards Alternative Pain Management: Growing interest and investment in non-opioid pain therapies, including non-pharmacological approaches and novel drug classes, pose a long-term challenge to traditional opioid markets.
- Strict Manufacturing Controls: The cost and complexity of adhering to stringent DEA and international regulations for controlled substance manufacturing add to operational expenses.
- Palliative Care Demand: The consistent and growing demand from palliative and end-of-life care settings remains a stable driver for morphine sulfate.
What is the Pricing Structure and Profitability for Morphine Sulfate Products?
The pricing and profitability of morphine sulfate are influenced by several factors:
- Generic Nature: The mature, generic status of most morphine sulfate products means pricing is highly competitive, driven by cost-efficiency in manufacturing.
- Volume Sales: Profitability is largely volume-dependent. Manufacturers with efficient production processes and strong distribution networks can achieve profitability.
- Formulation Complexity: More complex formulations, such as extended-release or abuse-deterrent versions, may command higher prices but also incur greater R&D and manufacturing costs.
- Regulatory Compliance Costs: Significant investment is required to meet stringent DEA and international regulatory requirements, impacting overall profitability.
- Reimbursement Policies: Payer policies and reimbursement rates influence the effective selling price.
- Distribution Channels: Markups occur at various stages of the supply chain, from manufacturer to wholesaler to pharmacy.
Typical wholesale acquisition costs for generic oral morphine sulfate tablets can range from a few cents to tens of cents per milligram, depending on the strength and quantity. Injectable forms can be more expensive per unit dose. Profit margins for pure generic morphine sulfate are generally considered to be lower than for novel or patented therapeutics.
What are the Intellectual Property and Patent Landscape Considerations?
The patent landscape for morphine sulfate itself is largely expired. As an old drug, its core chemical entity and basic therapeutic uses are in the public domain. However, opportunities for intellectual property protection exist in:
- Novel Formulations: Patents can be secured for new drug delivery systems, such as abuse-deterrent formulations (ADFs), extended-release mechanisms, or novel routes of administration.
- Manufacturing Processes: Proprietary advancements in manufacturing processes that improve efficiency, purity, or yield can be patented.
- Combination Therapies: Patents might cover specific combinations of morphine sulfate with other active pharmaceutical ingredients for synergistic effects or improved outcomes.
- Specific Medical Uses: While unlikely for basic pain relief, patents could theoretically cover highly specific, non-obvious medical indications if discovered.
The focus of IP strategy for companies involved with morphine sulfate now leans heavily towards formulation innovation, particularly in areas of abuse deterrence and improved patient adherence or convenience.
What are the Future Outlook and Potential Disruptions for Morphine Sulfate?
The future outlook for morphine sulfate is one of continued, albeit modest, demand, primarily within its established therapeutic niches. Potential disruptions include:
- Advancements in Non-Opioid Analgesics: The development and market penetration of highly effective non-opioid pain therapies could gradually reduce reliance on morphine sulfate, particularly for non-cancer-related pain.
- Enhanced Abuse Deterrence Technologies: Further innovation in ADFs could become a market differentiator and a regulatory prerequisite, potentially shifting market share to companies with superior technologies.
- Shifting Pain Management Paradigms: A broader societal and medical shift towards multimodal pain management, emphasizing physical therapy, psychological interventions, and non-pharmacological approaches, could decrease the overall volume of opioid prescriptions.
- Increased Regulatory Stringency: Further tightening of prescription guidelines, prescribing quotas, and dispensing regulations, driven by public health concerns, could limit market access and growth.
- Supply Chain Vulnerabilities: Geopolitical events or disruptions affecting opium poppy cultivation (primarily in Afghanistan, India, and Australia) could impact global supply and prices, although strategic reserves and alternative sourcing mitigate some risk.
- Novel Opioid Research: While the focus is on alternatives, continued research into safer, less addictive opioid molecules could theoretically emerge, though this remains a distant prospect given the current climate.
Key Takeaways
- Morphine sulfate maintains a stable market driven by its critical role in managing severe pain, particularly in palliative and post-operative care.
- The market is characterized by high competition from generic manufacturers and stringent regulatory oversight due to its Schedule II controlled substance status.
- Profitability hinges on manufacturing efficiency, volume sales, and compliance with strict DEA and international regulations.
- Intellectual property opportunities lie primarily in novel formulations, especially abuse-deterrent versions, rather than the drug entity itself.
- Future market dynamics will be shaped by the development of non-opioid alternatives, evolving abuse deterrence technologies, and ongoing public health initiatives to curb opioid misuse.
Frequently Asked Questions
-
What is the primary driver of current demand for morphine sulfate?
The primary driver is its established efficacy and indispensability in managing severe acute and chronic pain, particularly in palliative care, end-of-life settings, and post-operative recovery, where alternatives may be less effective or appropriate.
-
How do abuse-deterrent formulations (ADFs) impact the morphine sulfate market?
ADFs represent a significant trend. Manufacturers investing in and obtaining regulatory approval for ADFs can potentially differentiate their products, meet evolving regulatory expectations, and address some concerns surrounding opioid misuse, which can influence market share and pricing within the generic space.
-
Are there significant R&D investments in new therapeutic uses for morphine sulfate?
Substantial R&D investment is not focused on discovering new broad therapeutic uses for morphine sulfate itself, given its well-understood pharmacology and long history. Investment is primarily directed towards formulation improvements and abuse deterrence.
-
What is the impact of global opium cultivation on morphine sulfate supply?
Global opium cultivation, primarily in a few key countries, is the source of the raw material (opium) for morphine extraction. Disruptions in cultivation due to political instability, climate, or crop eradication efforts can affect raw material availability and, consequently, the supply and cost of morphine sulfate globally.
-
Will the ongoing opioid crisis lead to a complete phase-out of morphine sulfate from medical use?
A complete phase-out of morphine sulfate from legitimate medical use is highly unlikely in the foreseeable future. Its role in managing severe pain, especially in contexts where other options are insufficient or inappropriate, remains critical. The focus is on responsible prescribing, abuse deterrence, and harm reduction, not elimination from medical practice.
Cited Sources
[1] U.S. Drug Enforcement Administration. (n.d.). Schedules of Controlled Substances. Retrieved from [relevant DEA website section on drug scheduling, e.g., https://www.dea.gov/drug-scheduling]
[2] International Narcotics Control Board. (n.d.). Annual Reports. Retrieved from [relevant INCB website section on annual reports, e.g., https://www.incb.org/incb/en/publications/annual_reports.html]
[3] U.S. Food and Drug Administration. (n.d.). Information Resources on Abuse-Deterrent Opioid Analgesics. Retrieved from [relevant FDA website section on ADFs, e.g., https://www.fda.gov/drugs/information-drug-evaluation-and-research-center-der/abuse-deterrent-opioid-analgesics]