MICARDIS Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Micardis, and what generic alternatives are available?
Micardis is a drug marketed by Boehringer Ingelheim and is included in two NDAs.
The generic ingredient in MICARDIS is hydrochlorothiazide; telmisartan. There are thirty-two drug master file entries for this compound. Nine suppliers are listed for this compound. Additional details are available on the hydrochlorothiazide; telmisartan profile page.
Summary for MICARDIS
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 151 |
| Clinical Trials: | 71 |
| Patent Applications: | 4,727 |
| Formulation / Manufacturing: | see details |
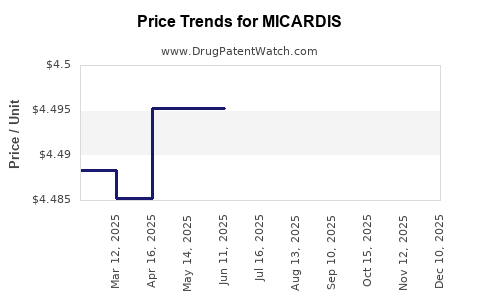
| Drug Prices: | Drug price information for MICARDIS |
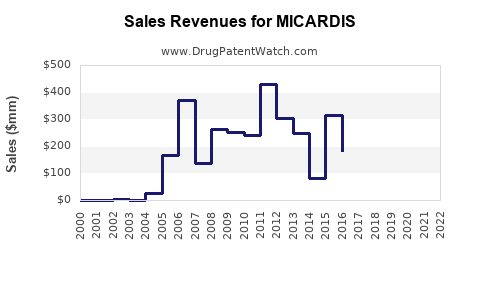
| Drug Sales Revenues: | Drug sales revenues for MICARDIS |
| What excipients (inactive ingredients) are in MICARDIS? | MICARDIS excipients list |
| DailyMed Link: | MICARDIS at DailyMed |
Recent Clinical Trials for MICARDIS
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Queens Medical Center | Phase 2 |
| University of Hawaii | Phase 2 |
| HK inno.N Corporation | Phase 1 |
Pharmacology for MICARDIS
| Drug Class | Angiotensin 2 Receptor Blocker |
| Mechanism of Action | Angiotensin 2 Receptor Antagonists |
Anatomical Therapeutic Chemical (ATC) Classes for MICARDIS
Paragraph IV (Patent) Challenges for MICARDIS
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| MICARDIS | Tablets | telmisartan | 20 mg, 40 mg and 80 mg | 020850 | 1 | 2006-12-26 |
US Patents and Regulatory Information for MICARDIS
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-003 | Apr 4, 2000 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Boehringer Ingelheim | MICARDIS HCT | hydrochlorothiazide; telmisartan | TABLET;ORAL | 021162-001 | Nov 17, 2000 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-001 | Nov 10, 1998 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for MICARDIS
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-001 | Nov 10, 1998 | ⤷ Try a Trial | ⤷ Try a Trial |
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-002 | Nov 10, 1998 | ⤷ Try a Trial | ⤷ Try a Trial |
| Boehringer Ingelheim | MICARDIS | telmisartan | TABLET;ORAL | 020850-003 | Apr 4, 2000 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for MICARDIS
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim International GmbH | Micardis | telmisartan | EMEA/H/C/000209 HypertensionTreatment of essential hypertension in adults.Cardiovascular preventionReduction of cardiovascular morbidity in patients with:manifest atherothrombotic cardiovascular disease (history of coronary heart disease, stroke, or peripheral arterial disease) or;type-2 diabetes mellitus with documented target-organ damage. |
Authorised | no | no | no | 1998-12-16 | |
| Krka, d.d., Novo mesto | Tolura | telmisartan | EMEA/H/C/001196 HypertensionTreatment of essential hypertension in adults.Cardiovascular preventionReduction of cardiovascular morbidity in patients with:manifest atherothrombotic cardiovascular disease (history of coronary heart disease or peripheral arterial disease) or;type 2 diabetes mellitus with documented target organ damage. |
Authorised | yes | no | no | 2010-06-04 | |
| Teva B.V. | Telmisartan Teva Pharma | telmisartan | EMEA/H/C/002511 Treatment of essential hypertension in adults. |
Authorised | yes | no | no | 2011-10-03 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for MICARDIS
See the table below for patents covering MICARDIS around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Canada | 2060624 | BENZIMIDAZOLES; COMPOSITIONS PHARMACEUTIQUES CONTENANT CES PRODUITS ET METHODES DE PREPARATION (BENZIMIDAZOLES, PHARMACEUTICAL COMPOSITIONS CONTAINING THESE COMPOUNDS AND PROCESSES FOR PREPARING THEM) | ⤷ Try a Trial |
| Hungary | 9200355 | ⤷ Try a Trial | |
| Israel | 100864 | Benzimidazole compounds their preparation and pharmaceutical compositions containing them | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for MICARDIS
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0502314 | 99C0014 | Belgium | ⤷ Try a Trial | PRODUCT NAME: TELMISARTAN REGISTRATION NO/DATE: EU/1/98/089/001 / 19981211 |
| 0502314 | SPC/GB99/014 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: TELMISARTAN, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT; REGISTERED: UK EU/1/98/089/001 19981211; UK EU/1/98/089/002 19981211; UK EU/1/98/089/003 19981211; UK EU/1/98/089/004 19981211; UK EU/1/98/089/005 19981211; UK EU/1/98/089/006 19981211; UK EU/1/98/089/007 19981211; UK EU/1/98/089/008 19981211; UK EU/1/98/089/009 19981211; UK EU/1/98/089/010 19981211 |
| 0502314 | 91802 | Luxembourg | ⤷ Try a Trial | 91802, EXPIRES: 20170131 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |


