HYDRALAZINE Drug Patent Profile
✉ Email this page to a colleague
When do Hydralazine patents expire, and what generic alternatives are available?
Hydralazine is a drug marketed by Watson Labs, Abraxis Pharm, Am Regent, Baxter Hlthcare Corp, Eugia Pharma, Fresenius Kabi Usa, Hikma, Kindos, Navinta Llc, Pharmobedient, Rising, Smith And Nephew, Solopak, Teva Parenteral, Actavis Elizabeth, Actavis Grp Ptc, Alkem Labs Ltd, Ascot, Cadila Pharms Ltd, Chartwell Rx, Glenmark Pharms Ltd, Halsey, Heritage, Heritage Pharms Inc, Hetero Labs Ltd Iii, Impax Labs, Invagen Pharms, Ivax Sub Teva Pharms, Macleods Pharms Ltd, Mutual Pharm, Pliva, Purepac Pharm, Quantum Pharmics, Sciegen Pharms, Strides Pharma, Strides Pharma Intl, Superpharm, Tp Anda Holdings, Upsher Smith Labs, Usl Pharma, Vangard, Vitarine, West Ward, Solvay, Ivax Pharms, and Mylan. and is included in ninety-nine NDAs.
The generic ingredient in HYDRALAZINE is hydralazine hydrochloride; hydrochlorothiazide; reserpine. There are twenty-one drug master file entries for this compound. Additional details are available on the hydralazine hydrochloride; hydrochlorothiazide; reserpine profile page.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for HYDRALAZINE?
- What are the global sales for HYDRALAZINE?
- What is Average Wholesale Price for HYDRALAZINE?
Summary for HYDRALAZINE
| US Patents: | 0 |
| Applicants: | 46 |
| NDAs: | 99 |
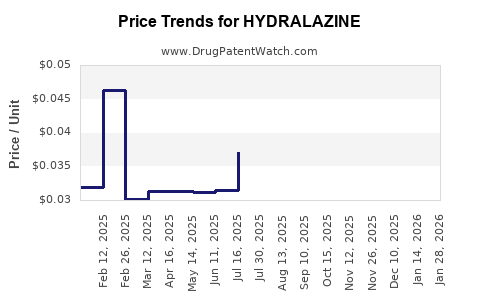
| Drug Prices: | Drug price information for HYDRALAZINE |
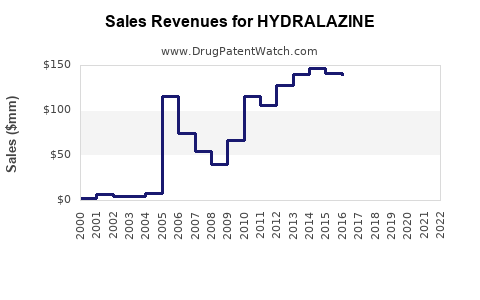
| Drug Sales Revenues: | Drug sales revenues for HYDRALAZINE |
| DailyMed Link: | HYDRALAZINE at DailyMed |
US Patents and Regulatory Information for HYDRALAZINE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pharmobedient | HYDRALAZINE HYDROCHLORIDE | hydralazine hydrochloride | TABLET;ORAL | 090413-001 | Dec 8, 2010 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Strides Pharma Intl | HYDRALAZINE HYDROCHLORIDE | hydralazine hydrochloride | TABLET;ORAL | 088391-001 | Sep 27, 1983 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Halsey | HYDRALAZINE HYDROCHLORIDE | hydralazine hydrochloride | TABLET;ORAL | 089222-001 | Jan 22, 1986 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Chartwell Rx | HYDRALAZINE HYDROCHLORIDE | hydralazine hydrochloride | TABLET;ORAL | 088847-001 | Feb 26, 1985 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |