Last updated: February 19, 2026
Glimepiride, a third-generation sulfonylurea, is a well-established oral antidiabetic medication used to manage type 2 diabetes mellitus. Its mechanism of action involves stimulating insulin secretion from pancreatic beta cells and reducing hepatic glucose production. The global glimepiride market is characterized by a mature product lifecycle, driven by generic competition and its cost-effectiveness, particularly in emerging markets. Patent expiries have led to widespread availability of generic formulations, significantly impacting originator revenue streams and fostering a price-sensitive market environment.
What is the Current Market Size and Projected Growth for Glimepiride?
The global glimepiride market is a mature segment within the antidiabetic drug sector. Precise current market size figures are subject to variation based on reporting methodologies and the inclusion of different product formulations and regional sales. However, industry analysis indicates a market valued in the hundreds of millions of U.S. dollars annually.
Projections for the glimepiride market forecast a period of modest, low single-digit compound annual growth rate (CAGR) for the foreseeable future. This subdued growth is primarily attributed to several factors:
- Genericization: The majority of patents protecting the original glimepiride formulations have expired globally. This has opened the door for numerous generic manufacturers, leading to intense price competition and erosion of market share for brand-name products.
- Emergence of Newer Therapies: The antidiabetic drug market has seen rapid innovation, with the introduction of newer classes of drugs such as GLP-1 receptor agonists, SGLT2 inhibitors, and DPP-4 inhibitors. These newer agents often offer improved efficacy, additional cardiovascular or renal benefits, and different mechanisms of action, making them increasingly preferred by prescribers and patients, particularly in developed markets.
- Shifting Treatment Paradigms: Treatment guidelines for type 2 diabetes are evolving to incorporate a broader range of therapeutic options beyond traditional oral agents, focusing on patient-specific risk factors and comorbidities.
- Cost-Effectiveness in Developing Markets: Despite the advent of newer drugs, glimepiride retains significant market presence in emerging economies due to its established safety profile and, critically, its affordability. The lower cost of generic glimepiride makes it a vital treatment option for large patient populations with limited access to more expensive novel therapies.
The market is therefore bifurcated: stable but declining revenue in developed nations due to competition from newer drug classes, and consistent, albeit low-growth, demand in developing regions driven by its accessibility.
Who are the Key Manufacturers and Competitors in the Glimepiride Market?
The glimepiride market is highly fragmented, with a significant number of manufacturers globally, predominantly in the generic drug space. The original innovator company, Hoechst Marion Roussel (now part of Sanofi), launched the brand-name drug Amaryl. However, following patent expiries, the landscape has become dominated by generic producers.
Key players and types of competitors include:
- Generic Pharmaceutical Companies: These companies focus on manufacturing and marketing bioequivalent versions of glimepiride after patent expiry. Major generic players with a significant presence in various global markets include:
- Sun Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Cipla
- Aurobindo Pharma
- Teva Pharmaceutical Industries
- Lupin
- Mylan (now Viatris)
- Contract Manufacturing Organizations (CMOs): Many companies outsource the manufacturing of their glimepiride products to specialized CMOs, particularly smaller regional players or those focused on specific formulations.
- Regional Manufacturers: Numerous smaller and medium-sized pharmaceutical companies operate in specific geographic regions, catering to local market demands and regulatory environments.
Competition is primarily based on:
- Price: This is the dominant competitive factor due to the generic nature of the product.
- Quality and Compliance: Manufacturers must adhere to stringent Good Manufacturing Practices (GMP) and regulatory standards set by bodies like the FDA, EMA, and other national agencies.
- Supply Chain Reliability: Consistent availability and timely delivery are critical for healthcare providers.
- Formulation Diversity: While the primary formulation is oral tablets, some manufacturers may offer variations in dosage strengths or extended-release formulations.
The competitive intensity remains high, driven by the large number of players and the commoditized nature of the product.
What are the Dominant Patent Expiries and Their Impact on Market Entry?
The patent landscape for glimepiride is largely characterized by the expiry of primary composition-of-matter and method-of-use patents. The original U.S. patent for glimepiride, held by Hoechst Marion Roussel, expired in the early 2000s. Similar patent expiries occurred in major global markets around the same period.
The impact of these patent expiries has been profound and predictable:
- Widespread Generic Entry: The expiration of core patents removed the primary barrier to entry for generic manufacturers. This led to a rapid influx of bioequivalent glimepiride products into the market.
- Price Erosion: The increased competition among multiple generic suppliers, often with lower overheads and production costs, resulted in a significant decline in the price of glimepiride. Brand-name products, where still available, saw substantial price reductions to remain competitive.
- Increased Market Accessibility: The lower cost of generic glimepiride has made it more accessible to a broader patient population, particularly in countries with developing healthcare systems and price-sensitive markets.
- Focus on Manufacturing Efficiency: For generic manufacturers, success hinges on efficient production processes, economies of scale, and robust supply chain management to achieve profitability in a low-margin environment.
- Limited Opportunities for New Entrants (for Brand-like Products): The absence of new patent protection on the core molecule means that opportunities for novel, high-margin product differentiation are minimal. New entrants primarily compete on cost and market access.
While secondary patents related to specific formulations (e.g., extended-release) or manufacturing processes may exist, they generally offer limited protection and are often subject to challenges or workarounds by generic competitors. The fundamental patent expiries have democratized access to glimepiride, transforming it into a widely available commodity drug.
What is the Regulatory Landscape and Compliance Requirements for Glimepiride?
The regulatory landscape for glimepiride is governed by national and regional health authorities. As an established drug, it is subject to standard pharmaceutical regulations concerning manufacturing, quality control, marketing, and post-market surveillance.
Key regulatory aspects include:
- Good Manufacturing Practices (GMP): All manufacturing facilities must comply with GMP guidelines set by regulatory bodies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and their counterparts worldwide. This ensures product quality, safety, and consistency.
- Abbreviated New Drug Applications (ANDAs) / Marketing Authorisation Applications (MAAs): Generic manufacturers seeking to market glimepiride must submit applications demonstrating bioequivalence to the reference listed drug. This involves extensive analytical testing and clinical studies.
- Pharmacovigilance: Manufacturers are required to monitor and report adverse drug reactions (ADRs) to regulatory authorities. Post-market surveillance is crucial for identifying any new safety concerns.
- Labeling and Packaging: Product labeling must accurately reflect the drug's indications, dosage, contraindications, warnings, precautions, and potential side effects, in compliance with regional regulations.
- Quality Control and Assurance: Robust quality control systems are essential to verify the identity, strength, quality, and purity of glimepiride at every stage of production.
- Intellectual Property Enforcement: While primary patents have expired, manufacturers must be aware of and navigate any remaining secondary patents or trade secrets related to manufacturing processes or specific formulations. Legal challenges or patent litigation can arise in this context.
- Import/Export Regulations: International trade of glimepiride products is subject to customs regulations, import licenses, and adherence to the importing country's pharmaceutical standards.
Compliance with these evolving regulations is critical for market access and maintaining product approval. Regulatory bodies conduct regular inspections of manufacturing sites to ensure ongoing adherence to standards.
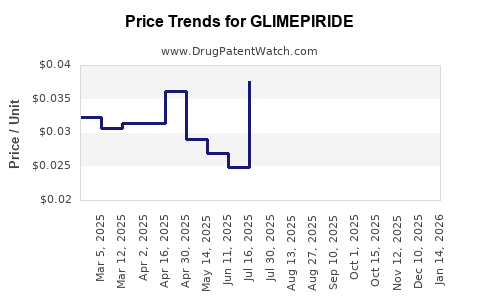
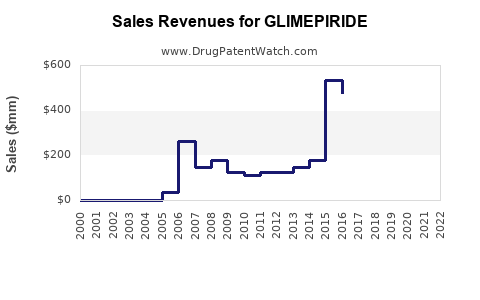
What are the Financial Trajectories and Pricing Strategies for Glimepiride Products?
The financial trajectory of glimepiride is largely defined by its post-patent expiry status, leading to a price-sensitive market.
Financial Trajectory:
- Declining Brand Revenue: For the original innovator, revenue from the branded glimepiride (e.g., Amaryl) has significantly diminished due to generic competition. The focus shifts from high-margin sales to managing residual market share or divesting the product line.
- Stable to Modest Growth for Generic Manufacturers: Generic manufacturers experience a revenue stream driven by sales volume. Profitability depends on manufacturing efficiency, economies of scale, and market access. While unit sales may remain consistent or grow modestly, profit margins per unit are typically low.
- Geographic Revenue Distribution: A substantial portion of global glimepiride revenue is generated from emerging markets where its affordability makes it a first-line or essential treatment. Developed markets contribute less revenue due to the preference for newer, higher-priced drugs.
- Portfolio Management: For diversified pharmaceutical companies, glimepiride represents a mature product within a broader portfolio. Its financial contribution is less significant compared to newer, patented drugs but provides steady, albeit low, revenue.
Pricing Strategies:
- Cost-Plus Pricing (Generic Market): Generic manufacturers typically employ a cost-plus pricing strategy. They calculate their production costs (raw materials, manufacturing, distribution, regulatory compliance) and add a profit margin. The competitive landscape forces this margin to be relatively thin.
- Volume-Based Pricing: In tenders and large-scale contracts with government health systems or large hospital networks, pricing is heavily influenced by the volume of units purchased. Deeper discounts are offered for larger orders.
- Regional Price Differentiation: Prices vary significantly across different countries and regions, influenced by local market dynamics, healthcare reimbursement policies, income levels, and the presence of specific competitors.
- Competitive Pricing: Generic manufacturers constantly monitor competitor pricing and adjust their own prices to remain competitive. This can lead to price wars, especially in highly saturated markets.
- Value-Based Pricing (Limited for Generics): For generic drugs like glimepiride, true value-based pricing is not applicable in the same way as for novel patented drugs. The perceived "value" is primarily its efficacy at a low cost.
The overall financial trajectory is one of stable, high-volume, low-margin sales, particularly for generic producers. Profitability is achieved through operational efficiency and market penetration rather than pricing power or product innovation.
What is the Future Outlook and Potential Disruptions for Glimepiride?
The future outlook for glimepiride is characterized by continued demand in specific market segments, but with limited growth potential and ongoing pressure from evolving treatment landscapes.
Future Outlook:
- Sustained Demand in Emerging Markets: Glimepiride will likely remain a cornerstone of diabetes management in low- and middle-income countries due to its affordability and established efficacy. Its role as a cost-effective option for large patient populations is secure in these regions.
- Continued Niche Use in Developed Markets: While newer drug classes dominate, glimepiride may retain a role for certain patient profiles in developed countries, particularly for individuals who cannot tolerate newer agents, have limited access to advanced therapies, or for whom cost is a primary concern and other options have failed.
- Consolidation in the Generic Market: The intense price competition may lead to consolidation among generic manufacturers as smaller, less efficient players are acquired or exit the market.
- Focus on Combination Therapies: Glimepiride may be increasingly formulated in fixed-dose combinations with other oral antidiabetic agents (e.g., metformin) to improve patient adherence and simplify treatment regimens. However, these combinations also face competition from newer fixed-dose combinations.
Potential Disruptions:
- Advancements in Diabetes Prevention and Cure: Breakthroughs in gene therapy, stem cell research, or novel preventative strategies could fundamentally alter the landscape of diabetes management, reducing the reliance on existing drug therapies over the long term. However, these are high-risk, long-horizon disruptions.
- Emergence of Even More Cost-Effective Novel Therapies: While currently more expensive, ongoing research may yield novel therapeutic classes that become more cost-competitive over time, potentially displacing older generics.
- Increased Focus on Lifestyle and Digital Health Solutions: The growing emphasis on digital health platforms, wearable devices for monitoring, and personalized lifestyle interventions could reduce the pharmacological burden of diabetes management, indirectly impacting the demand for oral antidiabetics.
- Stricter Regulatory Scrutiny on Older Generics: While unlikely for a drug with glimepiride's long safety record, any emerging safety signals, however minor, could lead to increased regulatory scrutiny or updated prescribing recommendations that favor newer agents.
- Global Health Policy Shifts: Major shifts in global health policy, particularly concerning universal healthcare access and drug pricing frameworks in developing nations, could indirectly influence the long-term demand and pricing dynamics for essential medicines like glimepiride.
The immediate future for glimepiride is one of stable, albeit low-growth, performance driven by its established role in specific markets, rather than significant expansion or innovation.
Key Takeaways
- The global glimepiride market is mature and dominated by generic competition, resulting in price sensitivity and low-margin sales.
- While patent expiries have democratized access, leading to widespread generic availability, they have also curtailed opportunities for significant revenue growth from originator products.
- Emerging markets represent the primary driver of sustained demand for glimepiride due to its affordability, whereas developed markets increasingly favor newer, more expensive antidiabetic therapies.
- Key competitors are numerous generic manufacturers focused on cost efficiency, quality compliance, and reliable supply chains.
- Future market dynamics will be shaped by continued demand in developing regions, niche use in developed markets, and potential long-term disruptions from advancements in diabetes prevention and cure.
Frequently Asked Questions
-
What is the primary indication for glimepiride?
Glimepiride is indicated for the treatment of type 2 diabetes mellitus, to improve glycemic control.
-
Why has glimepiride become a generic drug?
The primary composition-of-matter and method-of-use patents for glimepiride have expired globally, allowing multiple pharmaceutical companies to manufacture and market generic versions.
-
What are the main therapeutic advantages of glimepiride compared to older sulfonylureas?
Glimepiride is considered a third-generation sulfonylurea and may offer a more favorable pharmacokinetic profile and potentially lower risk of hypoglycemia compared to some older agents, although individual responses vary.
-
Are there any significant safety concerns associated with glimepiride?
The most common side effect is hypoglycemia (low blood sugar). Other potential side effects include weight gain and gastrointestinal disturbances. It should be used with caution in patients with renal or hepatic impairment.
-
How does the pricing of glimepiride compare to newer antidiabetic drugs like GLP-1 receptor agonists or SGLT2 inhibitors?
Glimepiride is significantly less expensive than GLP-1 receptor agonists and SGLT2 inhibitors, which are typically patented, novel therapies with higher manufacturing costs and offering additional benefits beyond glycemic control.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Approval Process. Retrieved from https://www.fda.gov/about-fda/center-drug-evaluation-and-research-cder/drug-approval-process
[2] European Medicines Agency. (n.d.). How to apply for a marketing authorisation. Retrieved from https://www.ema.europa.eu/en/how-we-work/research-and-development/applicant-guidance/how-apply-marketing-authorisation
[3] International Diabetes Federation. (2021). IDF Diabetes Atlas 10th edition 2021. Retrieved from https://www.diabetesatlas.org/