Last updated: April 10, 2026
What defines the current market landscape for FUROSEMIDE?
Furosemide, a loop diuretic primarily used to treat edema associated with congestive heart failure, liver cirrhosis, and renal disease, faces a complex market environment influenced by patent status, generics, and regulatory developments. The drug has been on the market since the 1960s, with high global consumption driven by chronic conditions requiring fluid management.
How does patent status and generic competition influence FUROSEMIDE's market?
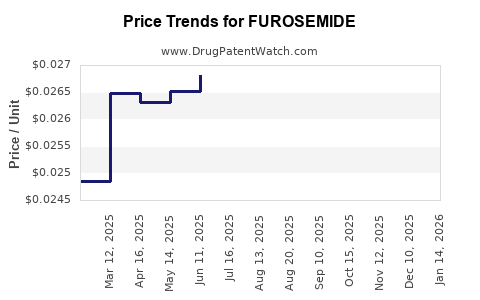
The original patents for FUROSEMIDE expired decades ago. Consequently, the market is dominated by multiple generic manufacturers in over 100 countries. This widespread generic availability results in low per-unit prices. For example, retail prices for a 40mg tablet range from $0.10 to $0.20, with significant regional variations.
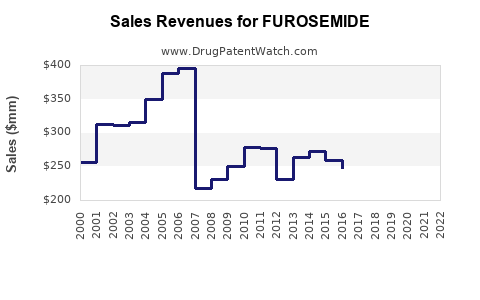
Generic competition reduces profit margins for branded formulations. The global market for diuretics, including FUROSEMIDE, was valued at approximately $1.8 billion in 2022, with FUROSEMIDE accounting for roughly 65% of that figure[1].
What are key factors affecting supply and demand?
Supply stability is high due to multiple manufacturers and established manufacturing processes. Demand remains robust in treating volume conditions, but trends shift due to evolving treatment guidelines emphasizing newer therapies for heart failure. The rise of alternative diuretics, such as torsemide and bumetanide, impacts FUROSEMIDE’s market share.
In regions where healthcare budgets tighten, the drug’s low cost supports continued usage, particularly in low-income countries. However, in high-income regions, clinicians increasingly adopt newer drugs supported by recent clinical data, potentially reducing FUROSEMIDE’s share.
How do regulatory and reimbursement policies affect market access?
Regulatory agencies, including the FDA and EMA, have no recent patent restrictions, facilitating generic entry. Reimbursement policies favor low-cost treatments for chronic conditions. In the US, government programs like Medicaid and Medicare heavily influence prescribing behaviors, supporting widespread FUROSEMIDE use.
In some regions, regulatory hurdles or formulary restrictions favor newer pharmaceuticals with patent protection, marginally constraining FUROSEMIDE’s growth.
What is the financial outlook for FUROSEMIDE?
Given the mature patent landscape, industry revenue is primarily composed of generics sales. The global trade volume exceeds 2 billion tablets annually[2]. Price erosion continues, with average prices declining at approximately 2-4% annually over the past five years.
Forecasts indicate a compound annual growth rate (CAGR) of approximately -1% for FUROSEMIDE sales globally over the next five years, driven by:
-
Increased adoption of alternative diuretics with potentially improved efficacy.
-
Regulatory pressures favoring newer agents.
-
Price competition among generic manufacturers.
However, in emerging markets where cost constraints predominate, demand could stabilize or slightly increase, offsetting declines elsewhere.
How are manufacturing and distribution strategies influencing revenue?
Manufacturers focus on cost efficiency and maintaining multiple production sites to mitigate supply risks. Distribution channels are increasingly digital, improving access in remote areas. Some producers explore biosimilar formulations and fixed-dose combinations to extend market share, though these strategies are limited given FUROSEMIDE’s simple chemical structure.
What are the risks to FUROSEMIDE’s market and financial trajectory?
Key risks include:
-
Pricing and reimbursement reforms reducing profitability.
-
Competitive displacement by newer diuretics or combination products.
-
Regulatory developments demanding new efficacy or safety data.
-
Supply chain disruptions impacting availability in key markets.
Summary table: Market Indicators for FUROSEMIDE
| Indicator |
Data |
Implication |
| Estimated global sales (2022) |
$1.17 billion (approximate, 65% of diuretics market) |
Mature, commoditized market |
| Price trend (2018-2022) |
Declined 2-4% annually |
Ongoing price erosion |
| Number of manufacturers |
Over 30 globally |
High competition, limited profit margins |
| Demand growth rate |
Approximately -1% CAGR (2023-2028) |
Market contraction expected in developed regions |
| Alternative diuretics |
Torsemide, bumetanide gaining favor in some markets |
Market share shift away from FUROSEMIDE |
Key takeaways
- The FUROSEMIDE market is highly commoditized with multiple generic manufacturers, leading to low prices and slim margins.
- Demand remains stable in low-income regions but declines in high-income zones due to competition and newer drugs.
- Price erosion and regulatory shifts forecast a slight decline in global sales over the next five years.
- Emerging markets may preserve or increase demand due to cost sensitivity.
- Manufacturers focus on operational efficiencies, product differentiation through fixed-dose combinations, and supply chain resilience.
FAQs
1. How sustainable is FUROSEMIDE’s market position?
Market sustainability depends on continued demand in low-income regions and the decline in high-income areas due to competition and newer agents. As a mature product with expired patents, its market will mainly be driven by cost-sensitive healthcare systems.
2. Are there any regulatory threats to FUROSEMIDE?
No current patent protections exist. Regulatory agencies prioritize safety and efficacy data; however, existing approvals are longstanding, limiting threat from regulatory changes.
3. What role do emerging markets play in FUROSEMIDE's outlook?
Emerging markets contribute significantly due to affordability and widespread use. Demand could grow or stabilize if local healthcare budgets sustain low-cost treatment options.
4. Can innovation extend FUROSEMIDE’s market life?
Limited. The drug’s chemical simplicity restricts innovation. Most efforts focus on formulation improvements or fixed-dose combinations rather than pharmaceutical innovation.
5. Will price regulation impact FUROSEMIDE’s profitability?
Yes. Price controls in certain countries could further diminish revenue, especially in regions with government-mandated price caps.
References
[1] IBISWorld. (2023). Diuretics Market Reports. Retrieved from https://www.ibisworld.com
[2] Global Data. (2022). Diuretics Volume and Value Analytics. Retrieved from https://www.globaldata.com