EFAVIRENZ Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Efavirenz, and what generic alternatives are available?
Efavirenz is a drug marketed by Aurobindo Pharma, Aurobindo Pharma Ltd, Cipla, Hetero Labs Ltd Iii, Macleods Pharms Ltd, Mylan, Pharmobedient, Strides Pharma, Hetero Labs Ltd V, Laurus, Teva Pharms Usa, and Chartwell Rx. and is included in twenty-one NDAs.
The generic ingredient in EFAVIRENZ is efavirenz; lamivudine; tenofovir disoproxil fumarate. There are twenty-six drug master file entries for this compound. Two suppliers are listed for this compound. Additional details are available on the efavirenz; lamivudine; tenofovir disoproxil fumarate profile page.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for EFAVIRENZ?
- What are the global sales for EFAVIRENZ?
- What is Average Wholesale Price for EFAVIRENZ?
Summary for EFAVIRENZ
| US Patents: | 0 |
| Applicants: | 12 |
| NDAs: | 21 |
| Finished Product Suppliers / Packagers: | 5 |
| Raw Ingredient (Bulk) Api Vendors: | 93 |
| Clinical Trials: | 437 |
| Patent Applications: | 5,354 |
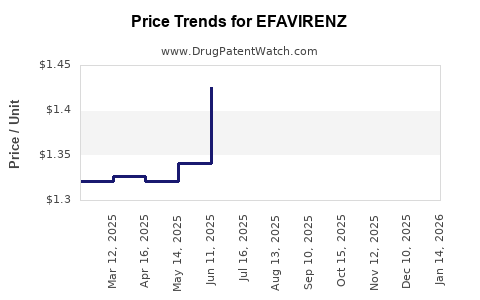
| Drug Prices: | Drug price information for EFAVIRENZ |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for EFAVIRENZ |
| DailyMed Link: | EFAVIRENZ at DailyMed |
Recent Clinical Trials for EFAVIRENZ
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Sunshine Lake Pharma Co., Ltd. | PHASE1 |
| Gilead Sciences | PHASE4 |
| National Center for AIDS/STD Control and Prevention, China CDC | PHASE4 |
Pharmacology for EFAVIRENZ
Anatomical Therapeutic Chemical (ATC) Classes for EFAVIRENZ
US Patents and Regulatory Information for EFAVIRENZ
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aurobindo Pharma | EFAVIRENZ | efavirenz | CAPSULE;ORAL | 078064-001 | Dec 15, 2017 | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Aurobindo Pharma Ltd | EFAVIRENZ, LAMIVUDINE AND TENOFOVIR DISOPROXIL FUMARATE | efavirenz; lamivudine; tenofovir disoproxil fumarate | TABLET;ORAL | 022343-001 | Aug 15, 2018 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Hetero Labs Ltd Iii | EFAVIRENZ | efavirenz | TABLET;ORAL | 078886-001 | Apr 27, 2018 | AB | RX | No | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Aurobindo Pharma | EFAVIRENZ | efavirenz | TABLET;ORAL | 205322-001 | Aug 30, 2018 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for EFAVIRENZ
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Teva B.V. | Efavirenz Teva | efavirenz | EMEA/H/C/002352Efavirenz is indicated in antiviral combination treatment of human-immunodeficiency-virus-1 (HIV-1)-infected adults, adolescents and children 3 years of age and older.Efavirenz has not been adequately studied in patients with advanced HIV disease, namely in patients with CD4 counts < 50 cells/mm3, or after failure of protease inhibitor (PI)-containing regimens. Although cross-resistance of efavirenz with protease inhibitors (PIs) has not been documented, there are at present insufficient data on the efficacy of subsequent use of PI-based combination therapy after failure of regimens containing efavirenz. | Authorised | yes | no | no | 2012-01-09 | |
| Merck Sharp & Dohme B.V. | Stocrin | efavirenz | EMEA/H/C/000250Stocrin is indicated in antiviral combination treatment of human-immunodeficiency-virus-1 (HIV-1)-infected adults, adolescents and children three years of age and older.Stocrin has not been adequately studied in patients with advanced HIV disease, namely in patients with CD4 counts < 50 cells/mm3, or after failure of protease-inhibitor (PI)-containing regimens. Although cross-resistance of efavirenz with PIs has not been documented, there are at present insufficient data on the efficacy of subsequent use of PI-based combination therapy after failure of regimens containing Stocrin. | Authorised | no | no | no | 1999-05-28 | |
| Bristol-Myers Squibb Pharma EEIG | Sustiva | efavirenz | EMEA/H/C/000249Sustiva is indicated in antiviral combination treatment of human-immunodeficiency-virus-1 (HIV-1)-infected adults, adolescents and children three years of age and older.Sustiva has not been adequately studied in patients with advanced HIV disease, namely in patients with CD4 counts < 50 cells/mm3, or after failure of protease-inhibitor (PI)-containing regimens. Although cross-resistance of efavirenz with PIs has not been documented, there are at present insufficient data on the efficacy of subsequent use of PI-based combination therapy after failure of regimens containing Sustiva. | Authorised | no | no | no | 1999-05-28 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
Market Dynamics and Financial Trajectory for Efavirenz
More… ↓