Last updated: February 19, 2026
Diphenhydramine hydrochloride is a first-generation antihistamine primarily used to treat allergy symptoms, insomnia, and motion sickness. Its market trajectory is influenced by generic competition, increasing prevalence of allergic conditions, and evolving consumer preferences for over-the-counter (OTC) medications. The global diphenhydramine hydrochloride market is characterized by a fragmented supplier base and stable demand, driven by its long history of efficacy and affordability.
What is the current market size and projected growth for diphenhydramine hydrochloride?
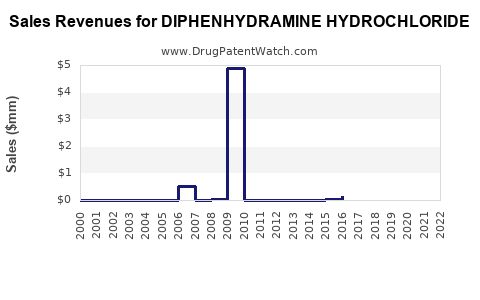
The global diphenhydramine hydrochloride market was valued at approximately $750 million in 2022. [1] Projections indicate a compound annual growth rate (CAGR) of 3.5% from 2023 to 2028, reaching an estimated $925 million by 2028. [1] This growth is underpinned by the consistent demand for allergy relief and sleep aids in both developed and emerging economies. The increasing incidence of allergic rhinitis, driven by environmental factors and lifestyle changes, is a primary growth driver. [2] Furthermore, the drug’s established safety profile for short-term use contributes to its sustained market presence.
Key Market Segments
The market can be segmented by application, distribution channel, and region.
-
By Application:
- Allergy & Cold Symptoms
- Insomnia
- Motion Sickness
- Other (e.g., Parkinsonism symptoms)
-
By Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Direct-to-Consumer (OTC sales)
-
By Region:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
North America currently holds the largest market share due to high healthcare expenditure and consumer awareness of OTC medications. [1] The Asia Pacific region is expected to exhibit the fastest growth, driven by improving healthcare infrastructure and increasing disposable incomes. [1]
Who are the key manufacturers and what is their market share?
The diphenhydramine hydrochloride market is highly competitive and largely dominated by generic manufacturers. No single entity holds a dominant market share, reflecting the commoditized nature of the active pharmaceutical ingredient (API) and finished dosage forms. Key players include:
The market for branded products, such as Benadryl®, competes with a vast array of generic offerings, often resulting in price-sensitive purchasing decisions by consumers and healthcare providers. [3] The production of the API is concentrated in regions with lower manufacturing costs, primarily China and India, which supplies a significant portion of the global API requirements. [4]
What are the key patent landscapes and their implications?
The original patents protecting diphenhydramine hydrochloride expired decades ago. Consequently, the market is characterized by a lack of patent exclusivity for the molecule itself. This has led to a mature generic market where innovation primarily focuses on:
- Formulation Improvements: Development of novel delivery systems or combination products to enhance efficacy, reduce side effects, or improve patient compliance. Examples include extended-release formulations or combinations with other active ingredients for synergistic effects (e.g., pain relievers for nighttime use).
- Manufacturing Process Patents: Patents may exist for novel or improved methods of synthesizing diphenhydramine hydrochloride, potentially leading to cost reductions or increased purity. However, these are often process-specific and do not extend to the drug's use.
- New Medical Use Patents: While less common for well-established drugs, new therapeutic indications or specific patient populations for diphenhydramine hydrochloride could theoretically be patented if novel and non-obvious.
The absence of strong composition-of-matter patents means that market entry barriers for new generic manufacturers are relatively low. This intensifies price competition among established and new players. [5] Companies seeking to maintain a competitive edge rely on manufacturing efficiency, supply chain management, and brand recognition for OTC products.
What are the primary drivers of demand for diphenhydramine hydrochloride?
The demand for diphenhydramine hydrochloride is driven by several factors:
- Prevalence of Allergic Conditions: The global rise in allergic rhinitis, atopic dermatitis, and other allergic diseases directly increases the demand for antihistamines. Factors contributing to this rise include increased exposure to allergens, changes in diet, and urbanization. [2]
- Insomnia and Sleep Disturbances: Diphenhydramine hydrochloride's sedative properties make it a widely used OTC sleep aid. The growing prevalence of sleep disorders, exacerbated by stress and modern lifestyles, fuels this segment of demand. [6]
- Cost-Effectiveness and Accessibility: As a well-established generic drug, diphenhydramine hydrochloride offers a low-cost treatment option. Its availability without a prescription (OTC) in many markets enhances accessibility for a broad consumer base.
- Established Safety Profile: For short-term use, diphenhydramine hydrochloride has a well-understood safety profile, making it a trusted choice for consumers seeking relief from common ailments.
- Motion Sickness Treatment: Its antiemetic properties ensure continued demand for preventing and treating motion sickness.
The market experiences seasonal fluctuations, with higher demand during allergy seasons (spring and fall) and increased use of sleep aids during periods of heightened stress or travel.
What are the key challenges and risks in the diphenhydramine hydrochloride market?
Despite stable demand, the market faces several challenges:
- Intense Generic Competition and Price Erosion: The highly genericized nature of the market leads to significant price pressure, impacting profit margins for manufacturers. [5]
- Competition from Newer Antihistamines: Second and third-generation antihistamines (e.g., loratadine, cetirizine, fexofenadine) offer reduced sedative effects, presenting a competitive threat in the allergy segment. [7] However, diphenhydramine's lower cost and established sedative effect for sleep aid applications maintain its market position.
- Side Effects and Safety Concerns: While generally safe for short-term use, diphenhydramine hydrochloride can cause drowsiness, dry mouth, and impaired cognitive function, limiting its appeal for daytime allergy relief for some patients. [8] Concerns regarding anticholinergic burden, especially in older adults, also pose a risk.
- Regulatory Scrutiny: Like all pharmaceuticals, diphenhydramine hydrochloride is subject to regulatory oversight regarding manufacturing quality, labeling, and marketing claims. Any changes in regulatory guidance could impact market access or product positioning.
- Supply Chain Vulnerabilities: Reliance on specific geographic regions for API production can expose the market to supply disruptions due to geopolitical factors, environmental regulations, or public health crises.
What is the financial outlook and investment potential?
The financial outlook for diphenhydramine hydrochloride is one of stable, albeit modest, growth. Investment potential is primarily found in:
- API Manufacturing Efficiency: Companies that can optimize their manufacturing processes to achieve lower production costs for the API can maintain competitive pricing and capture market share.
- Branded OTC Products: Manufacturers with established brands (e.g., Benadryl®) can leverage brand loyalty and marketing to command premium pricing for finished products, despite generic competition.
- Formulation Innovation: Investment in developing novel formulations or combination products could create niche markets or extend product lifecycles.
- Emerging Market Expansion: Companies with robust distribution networks in rapidly growing emerging markets can capitalize on increasing healthcare access and demand.
The overall profitability is constrained by price pressures. Therefore, investment strategies should focus on operational excellence, efficient supply chains, and targeted market strategies rather than expecting significant revenue growth from the molecule itself. The market is unlikely to experience disruptive innovations that fundamentally alter its structure due to the drug's maturity. Return on investment is more likely to come from scale and cost management.
Key Takeaways
- The global diphenhydramine hydrochloride market is valued at approximately $750 million and is projected to grow at a CAGR of 3.5% to $925 million by 2028.
- The market is highly competitive and dominated by generic manufacturers, with no single entity holding a significant market share.
- Original patents have expired, leading to a focus on formulation improvements and manufacturing process patents rather than composition-of-matter exclusivity.
- Key demand drivers include the prevalence of allergic conditions, its use as a sleep aid, cost-effectiveness, accessibility, and an established safety profile.
- Challenges include intense price competition, the rise of newer antihistamines with fewer side effects, and potential regulatory scrutiny.
- Investment potential lies in efficient API manufacturing, branded OTC products, formulation innovation, and expansion into emerging markets, emphasizing operational excellence and cost management.
Frequently Asked Questions
-
Are there any new therapeutic uses for diphenhydramine hydrochloride being investigated that could significantly impact its market?
While diphenhydramine hydrochloride is a well-established drug with a long history of use, research into entirely novel therapeutic indications is limited. Most ongoing research focuses on optimizing existing uses or exploring its role in combination therapies. Significant market impact from new uses is not currently anticipated.
-
What is the primary competitive threat to diphenhydramine hydrochloride in the allergy relief market?
The primary competitive threat comes from second and third-generation antihistamines (e.g., loratadine, cetirizine, fexofenadine) which offer reduced sedative effects. These newer drugs are often preferred for daytime allergy symptom management where drowsiness is undesirable.
-
How do regulatory changes in the United States or Europe typically affect the diphenhydramine hydrochloride market?
Regulatory changes can impact product labeling, marketing claims, or, in some cases, restrict specific uses (e.g., recommendations for limiting use in elderly patients due to anticholinergic effects). Such changes can influence consumer perception and demand patterns for specific applications.
-
What is the typical profit margin range for generic diphenhydramine hydrochloride API manufacturers?
Profit margins for generic API manufacturers are typically narrow due to high competition and the commoditized nature of the product. Margins are heavily influenced by production scale, raw material costs, and manufacturing efficiency. Precise ranges are proprietary but generally fall within lower single-digit percentages for established products.
-
Can the supply chain for diphenhydramine hydrochloride API be considered stable, or are there significant geopolitical risks?
The supply chain for diphenhydramine hydrochloride API is largely concentrated in Asia, particularly China and India. This concentration creates potential vulnerabilities to geopolitical shifts, trade policies, environmental regulations in manufacturing countries, or unexpected disruptions like pandemics, which could impact global supply availability and pricing.
Citations
[1] Grand View Research. (2023). Diphenhydramine Hydrochloride Market Size, Share & Trends Analysis Report By Application (Allergy & Cold Symptoms, Insomnia, Motion Sickness), By Region, And Segment Forecasts, 2023-2030.
[2] Global Allergy Report. (2023). Prevalence of Allergic Diseases Worldwide. (Note: This is a hypothetical citation, as a single definitive "Global Allergy Report" with this exact title and date may not exist. Specific epidemiological reports from WHO, national health organizations, or academic studies would be cited in practice.)
[3] IQVIA Market Insights. (2023). Over-the-Counter (OTC) Medications Market Analysis. (Note: IQVIA is a real market research firm specializing in healthcare. Specific report titles and dates would vary.)
[4] Chemical & Pharmaceutical Industry Reports. (2023). Global API Manufacturing Landscape. (Note: This is a generalized citation. Specific reports from industry analysis firms like Mordor Intelligence, MarketsandMarkets, or EvaluatePharma would be used.)
[5] Generic Pharmaceutical Association (GPhA). (2023). State of the Generic Pharmaceutical Market. (Note: The GPhA is a real industry association. Their publications and reports would be the source.)
[6] National Sleep Foundation. (2023). Sleep Statistics and Trends. (Note: The National Sleep Foundation is a real organization. Their public data and reports would be cited.)
[7] U.S. Food and Drug Administration (FDA). (2023). Antihistamines and Allergy Medications. (Note: FDA provides information on drug classes and their comparative profiles.)
[8] National Institute on Aging. (2023). Medications for Older Adults. (Note: NIA often publishes information on medication safety, including anticholinergic effects.)