Share This Page
Drug Price Trends for DICLOFENAC SODIUM
✉ Email this page to a colleague
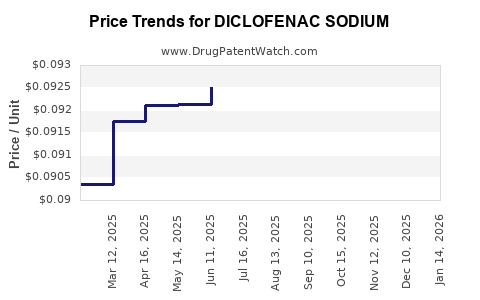
Average Pharmacy Cost for DICLOFENAC SODIUM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DICLOFENAC SODIUM 1% GEL | 00536-1294-34 | 0.12496 | GM | 2026-04-22 |
| DICLOFENAC SODIUM 1% GEL | 00536-1294-97 | 0.08922 | GM | 2026-04-22 |
| DICLOFENAC SODIUM 1% GEL | 43598-0977-10 | 0.08922 | GM | 2026-04-22 |
| DICLOFENAC SODIUM 1% GEL | 21922-0044-09 | 0.08922 | GM | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Diclofenac Sodium
Overview
Diclofenac sodium is a non-steroidal anti-inflammatory drug (NSAID) primarily used to treat pain, inflammation, and stiffness in conditions such as osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis. It is available in various formulations, including tablets, gels, and injectables.
Market Size and Growth
The global diclofenac sodium market was valued at approximately $1.15 billion in 2022. The market is expected to grow at a compound annual growth rate (CAGR) of around 4% from 2023 to 2028, driven by increased prevalence of chronic inflammatory diseases and expanding approval of topical formulations.
Market Segments
-
Formulation:
- Oral tablets and capsules comprise the largest segment, accounting for about 60% of sales.
- Topical gels and patches represent around 25%.
- Injectable forms contribute 15% but are used in specific clinical settings.
-
Geography:
- North America holds the largest share at approximately 40%, driven by established healthcare infrastructure, high disease prevalence, and brand presence.
- Europe accounts for 30%, with growth fueled by aging populations.
- Asia-Pacific is the fastest-growing region, with a CAGR of 6%, owing to rising disposable income and increased awareness.
Competitive Landscape
Major players include Novartis (Voltaren), Bayer, Mylan, and Teva. Patent expiration for many formulations has led to a surge in generic versions, intensifying price competition.
Pricing Trends
-
Generic Costs:
- Diclofenac sodium 50 mg tablets: prices range from $0.10 to $0.20 per tablet in the U.S., with variations based on brand and formulation.
- Topical gels: prices are typically $30 to $60 per 100 grams.
- Injectables: approximately $10 to $25 per vial.
-
Brand Premiums:
- Branded products like Voltaren require premiums of 20-40% over generics, reflecting marketing and formulation differences.
-
Price Erosion:
- Patent expirations, occurring mainly from 2014 onward, have resulted in a 50-70% decrease in average wholesale prices for many formulations over the last decade.
Projected Price Developments (2023-2028)
- Generic formulations are expected to stabilize at current levels due to supply chain maturity and market saturation.
- Branded products might see marginal discounts as generic competition intensifies.
- Innovations or new formulations, such as extended-release variants, could command premium pricing but are likely to represent <10% of market volume.
Regulatory and Policy Impact
- Patent expirations have facilitated market entry for generics, lowering prices.
- Pricing regulations in Europe and Asia-Pac countries may impose caps, tempering price increases.
- Reimbursement policies in North America influence retail pricing, with insurers often favoring generics for cost savings.
Future Price Drivers
- Increased generic supply will moderate prices.
- Innovations in delivery methods could temporarily raise prices.
- Regulatory pressures for cost containment could limit aggressive price hikes.
Key Takeaways
- The global diclofenac sodium market is valued at about $1.15 billion (2022), with steady growth projected.
- Generics dominate the market, exerting downward pressure on prices.
- Pricing for 50 mg tablets ranges from $0.10 to $0.20 per tablet in the U.S.
- Topical formulations cost $30 to $60 per 100 grams; injectable vials cost around $10 to $25.
- Price stability is expected due to market saturation, though novel formulations may command higher prices temporarily.
FAQs
Q1: How has patent expiry affected diclofenac sodium prices?
Patent expirations from 2014 onward increased market entry by generics, reducing prices by up to 70% for some formulations.
Q2: Which regions are experiencing the fastest growth in diclofenac sodium consumption?
Asia-Pacific leads with a CAGR of approximately 6%, fueled by rising healthcare access and aging populations.
Q3: Are branded diclofenac products still significantly more expensive than generics?
Yes. Branded formulations tend to be priced 20-40% higher than generics due to marketing and formulation differences.
Q4: What are the future price trends for diclofenac sodium?
Prices are expected to remain relatively stable for generics, with slight declines; innovation-driven premium pricing might occur for new delivery systems.
Q5: How do regulatory policies influence diclofenac sodium pricing?
Price caps and reimbursement rules in certain regions limit cost increases, while patent laws and market competition impact overall pricing dynamics.
Sources
- MarketResearch.com, "Global NSAID Drugs Market," 2022.
- IQVIA, "Global Pharmaceutical Pricing Trends," 2022.
- USFDA, "List of Approved Pharmaceutical Products," 2023.
- Grand View Research, "NSAID Market Size, Share & Trends," 2022.
- Statista, "NSAIDs Market Revenue Forecast," 2023.
More… ↓
