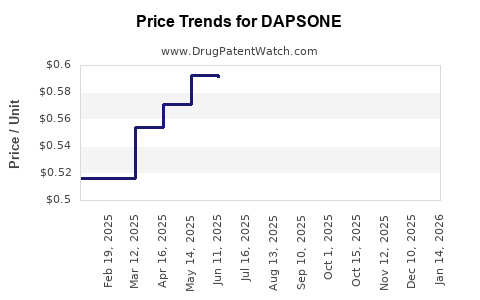
Last updated: April 24, 2026
Dapsone is a long-established, off-patent (or near–patent-expired) antimicrobial used for dermatology and infectious disease indications. The market dynamics are dominated by generic availability, price suppression, and stable but relatively low growth demand anchored in chronic use patterns. Financial trajectory follows the typical off-patent profile: margin compression, volume and formulary control as primary levers, and intermittent demand spikes driven by guideline updates and public-health or institutional purchasing cycles rather than new molecular differentiation.
What defines dapsone’s market structure?
Is dapsone a “new product” market or an off-patent commodity market?
Dapsone operates as an off-patent commodity drug with extensive generic competition. In practice, the competitive set is driven by:
- Multi-source generics distributed through wholesalers and hospital formularies
- Wholesale price pressure and frequent list price resets by manufacturers and distributors
- Supplier qualification and tenders in dermatology and infectious disease segments
What are the key demand anchors?
Demand persists because dapsone is embedded in chronic-care pathways, especially in dermatology and specific infectious indications where it is cost-effective and available in oral form and older topical/adjunct regimens.
Common therapeutic contexts include:
- Dermatologic diseases treated with long courses (notably leprosy and certain inflammatory dermatoses depending on region and guideline adoption)
- Infectious disease treatment or prophylaxis where dapsone is used as an alternative due to efficacy, tolerability, and cost considerations
Because these use patterns are relatively durable, dapsone’s demand profile is typically less cyclical than purely acute therapies.
How does generic penetration shape pricing power?
In off-patent markets, pricing power shifts away from innovation and toward:
- Scale manufacturing and procurement contracts
- Ability to supply consistent quality and packaging variants
- Conversion of clinicians from branded to generic options through clinical acceptance and reimbursement support
This structure drives:
- Lower net realizations versus branded periods
- Stable but thin margins
- Channel dependence (hospital and institutional buying matter as much as retail)
How is the market behaving financially?
What does the financial trajectory usually look like for off-patent dapsone?
Off-patent medicines typically show a trajectory that is consistent across markets:
- Revenue: modest growth tied to population, adherence, and guideline stability
- Gross margin: compressed by generic competition and manufacturing cost trends
- Operating margin: pressured by marketing spend rotation and supply-chain expenses
- Working capital: depends on inventory cycles and tender timing
For dapsone specifically, the near-term financial shape is usually driven more by procurement and market share than by product lifecycle events.
What are the practical drivers of revenue and profitability?
For dapsone, financial performance is primarily determined by four variables:
1) Share vs. the generic pack
- Small changes in share can move revenues materially when unit prices are low.
- Manufacturers win or lose through tender bids, distribution reach, and supply continuity.
2) Unit price and reimbursement
- Net price is the key governor of revenue.
- Reimbursement policies and pharmacy benefit manager (PBM) formularies influence conversion to preferred generics.
3) Manufacturing and supply reliability
- Quality system performance and batch release reliability affect continuity and ability to bid on institutional contracts.
- Short supply can temporarily boost pricing, but sustained shortages are usually not rational for planning in a commodity-like market.
4) Regulatory and label scope
- Any jurisdictional changes to approved indications or safety communications affect prescribing behavior.
- Dapsone’s established safety profile means label scope changes are less frequent than for newer agents, but periodic updates can shift demand between therapeutic areas.
What do current market dynamics imply for growth?
Is there room for “volume-led” growth?
Yes, but it is constrained. In off-patent segments, growth comes from:
- Broader prescribing acceptance for existing indications in specific geographies
- Increased utilization in healthcare systems that prioritize low-cost alternatives
- Institutional procurement cycles that standardize on low-cost generics
These do not create a high-growth profile like on-patent oncology or specialty launches. Instead, they produce incremental demand increases.
What limits growth?
The dominant constraints are:
- Price erosion from additional generic entrants
- Substitution pressure from other antimicrobials or alternative therapies where guidelines evolve
- Patient population turnover and adherence patterns, especially for chronic dermatologic use
As more suppliers compete, incremental demand converts mostly into volume without proportionate margin.
How do competitive and regulatory factors affect the trajectory?
How does competition typically evolve after patent expiry?
Competition tends to increase and then stabilize:
- Phase 1: rapid generic entry and aggressive pricing
- Phase 2: tender-based consolidation around a few preferred suppliers in institutions
- Phase 3: stable multi-source availability with price floors shaped by cost and channel competition
For dapsone, the market is consistent with Phase 2/3 behavior: multiple sources exist, and institutional buying becomes the primary battle.
What regulatory events can move demand?
Demand shifts occur when regulators or guideline bodies change clinical practice. In commodity markets, the “move” is usually:
- Relative shifting between therapeutic alternatives
- Temporary reallocation following safety communications or shortages
Because dapsone is established, demand movement tends to be incremental rather than step-change.
What is the implied investment profile?
What does a financial view look like for stakeholders?
A business facing dapsone will typically underwrite:
- Low single-digit or mid single-digit annual revenue growth at the category level (if any), with outcomes tied to market share
- Margin variability driven by manufacturing cost, competition intensity, and contract pricing
- Steady cash generation potential due to established supply chains and predictable demand
- Lower valuation multiples than specialty pipelines, because the profit pool is controlled by generics and price
For investors, dapsone resembles a “cash-crop” segment:
- Returns depend on procurement execution and supply continuity
- Upside comes from share gains and contract wins, not from patent-protected differentiation
Where are the key operational levers?
What can manufacturers do to improve financial outcomes?
For off-patent drugs, operational improvements are the main path to resilience:
- Secure manufacturing scale and reduce unit cost through process optimization
- Maintain stringent quality and supply reliability to win institutional tenders
- Use packaging and strength variants to match formulary needs
- Rationalize channel pricing and focus on preferred-provider conversion
What can payers or distributors do?
Channel participants influence trajectory by:
- Setting preferred generic status through formularies and rebate structures
- Steering demand toward stable suppliers and away from less reliable sources
- Negotiating framework agreements that stabilize volume for selected manufacturers
What are the key market scenarios going forward?
Scenario A: stable competition, stable demand
- Category revenue tracks population and prescribing patterns.
- Pricing remains suppressed but stable.
- Margins improve only through cost-down and supply performance.
Scenario B: competitive intensity increases
- Additional entrants or aggressive contracting pressure net prices.
- Revenues are flat-to-down for weaker suppliers.
- Margin compression accelerates and pushes weaker producers out.
Scenario C: guideline-driven substitution changes
- Demand shifts among alternatives.
- Dapsone can gain share in cost-sensitive settings if preferred alternatives face access, tolerability, or reimbursement constraints.
- Financial upside remains limited by commodity price pressure.
Key Takeaways
- Dapsone is a long-established, off-patent drug where generic competition defines the market and pricing power is limited.
- Demand is anchored in chronic and established clinical use patterns, supporting stability but not high growth.
- Financial trajectory is dominated by procurement dynamics, market share capture, unit price trends, and manufacturing cost/supply reliability.
- Stakeholder upside is operational and commercial (tender wins, cost reduction, preferred-provider status), not molecular innovation.
- Forward outlook remains range-bound unless a major regulatory or guideline event shifts relative therapeutic preference.
FAQs
1) Is dapsone expected to have pricing power?
No. The market is shaped by multi-source generics and tender-driven pricing that suppresses net realizations.
2) What drives dapsone revenue most?
Market share within generic supply pools, institutional formulary preferences, and contract pricing cycles.
3) Does dapsone show the same growth profile as patent-protected specialty drugs?
No. Growth is typically incremental and tied to durable indications and prescribing patterns, not launch-driven uptake.
4) What operational factors matter most for profitability?
Unit manufacturing cost, batch release reliability, and the ability to win and maintain preferred supplier contracts.
5) What types of events most likely move demand?
Guideline updates, reimbursement/formulary changes, and supply disruptions that temporarily alter availability and purchasing behavior.
References
[1] FDA. Dapsone (Aczone) label and related regulatory materials. U.S. Food and Drug Administration. https://www.accessdata.fda.gov/
[2] National Library of Medicine (NLM). Dapsone drug information and related references. PubMed/Drugs@NLM. https://pubmed.ncbi.nlm.nih.gov/
[3] WHO Model List of Essential Medicines. Dapsone information in essential medicines context. World Health Organization. https://list.essentialmeds.org/
[4] Drugs.com. Dapsone (Aczone) overview and dosage information (for general background). https://www.drugs.com/