Last updated: February 19, 2026
Clonidine, a medication primarily used for hypertension and attention deficit hyperactivity disorder (ADHD), faces a mature market characterized by widespread generic availability and a stable, albeit slow-growth, demand. The patent landscape for clonidine itself is long expired, with original composition of matter patents dating back to the 1960s. Current patent activity focuses on novel formulations, delivery methods, and new therapeutic indications, which may offer limited exclusivity periods. The global market for clonidine is projected to experience a compound annual growth rate (CAGR) of 2.5% from 2023 to 2028, driven by its established efficacy, affordability, and continued use in specific patient populations.
WHAT ARE CLONIDINE'S PRIMARY THERAPEUTIC USES?
Clonidine is approved by the U.S. Food and Drug Administration (FDA) for the treatment of hypertension. Its alpha-2 adrenergic agonist mechanism of action reduces sympathetic outflow from the central nervous system, leading to decreased heart rate, vasodilation, and reduced peripheral resistance. Beyond its antihypertensive function, clonidine is also prescribed off-label for a range of other conditions, including:
- Attention Deficit Hyperactivity Disorder (ADHD): Particularly extended-release formulations like Kapvay.
- Withdrawal Syndromes: Opioid, alcohol, and nicotine withdrawal, managing symptoms like anxiety, insomnia, and muscle cramps.
- Tourette Syndrome: Reducing tics.
- Hot Flashes: Associated with menopause.
- Sleep Disorders: Insomnia, especially in children and individuals with ADHD.
WHAT IS THE PATENT STATUS OF CLONIDINE?
The original composition of matter patents for clonidine have long expired, making the active pharmaceutical ingredient (API) itself generic. This means that numerous manufacturers produce and market generic versions of clonidine tablets and patches. The primary patent expirations for the original clonidine hydrochloride occurred in the 1980s [1].
Recent patent filings and granted patents related to clonidine are almost exclusively focused on:
- New Formulations: Development of extended-release (ER) or controlled-release (CR) formulations to improve dosing convenience and reduce side effects (e.g., Kapvay, developed by Shire, now Takeda).
- Novel Delivery Systems: Transdermal patches with improved adhesion, drug release profiles, or reduced skin irritation.
- Combination Therapies: Patents for clonidine used in conjunction with other active ingredients for synergistic effects or broader therapeutic coverage.
- New Indications: Research into and patenting of clonidine's efficacy in previously unapproved therapeutic areas, although significant breakthroughs in this area are limited given the drug's age.
These secondary patents offer limited market exclusivity, typically 20 years from the filing date, and often face challenges from generic manufacturers based on obviousness or lack of novelty. For instance, the patent for Kapvay (clonidine hydrochloride extended-release tablets) has an expiration date in the late 2020s, but its market success was contingent on demonstrating superior efficacy and safety profiles over immediate-release generics for ADHD [2].
HOW HAS THE MARKET FOR CLONIDINE EVOLVED?
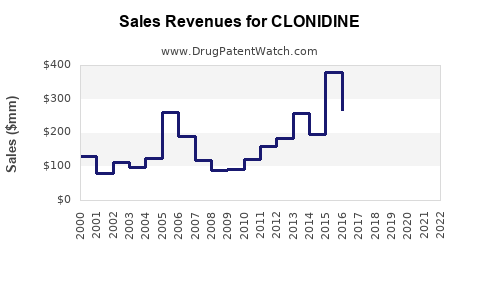
The market for clonidine has matured significantly due to the advent of generic competition.
- Early Market Dominance: Initially, clonidine was a prominent brand-name drug for hypertension, with market exclusivity held by Boehringer Ingelheim (Catapres).
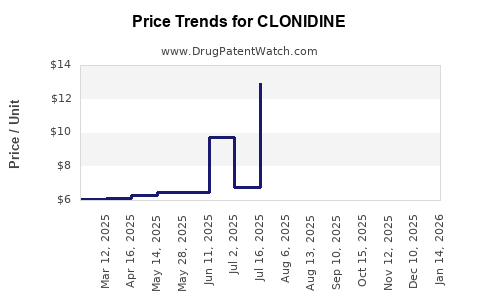
- Generic Entry: Following patent expiries, multiple generic manufacturers entered the market, leading to substantial price erosion and a shift towards cost-effective generics.
- Specialty Formulations: The introduction of extended-release formulations, such as Kapvay for ADHD, created a niche market segment. These formulations commanded higher prices due to proprietary technology and clinical trial data demonstrating improved patient outcomes.
- Current Landscape: Today, the clonidine market is a bifurcated landscape. The bulk of the market by volume consists of low-cost generic immediate-release tablets and patches for hypertension. A smaller, higher-value segment comprises specialty extended-release formulations for ADHD, which are still subject to some level of market protection but face increasing generic and biosimilar (if applicable to drug class) pressures.
WHAT ARE THE KEY MARKET DRIVERS AND RESTRAINTS FOR CLONIDINE?
Market Drivers:
- Established Efficacy and Safety Profile: Clonidine has a decades-long track record of effectiveness and a generally well-understood safety profile, making it a trusted treatment option for physicians.
- Affordability of Generics: The widespread availability of low-cost generic clonidine makes it an accessible treatment, particularly in price-sensitive markets and for patients with limited insurance coverage.
- Continued Use in Off-Label Indications: The drug's utility in managing withdrawal symptoms, ADHD, and other conditions ensures sustained demand beyond its primary indication.
- Aging Population: The global rise in the aging population is associated with an increased prevalence of cardiovascular diseases, including hypertension, which could indirectly support demand for antihypertensive agents like clonidine.
- Pediatric ADHD Treatment: Extended-release formulations remain a viable option for ADHD treatment in children, a growing therapeutic area.
Market Restraints:
- Generic Erosion: Intense price competition among generic manufacturers limits revenue potential and profit margins for these products.
- Availability of Newer Antihypertensives: The hypertension market is highly competitive, with a wide array of newer drug classes offering improved efficacy, fewer side effects, and better patient adherence (e.g., ACE inhibitors, ARBs, calcium channel blockers). This has led to clonidine's use in hypertension becoming more specialized or second-line.
- Side Effect Profile: Common side effects such as drowsiness, dizziness, dry mouth, and constipation can limit patient compliance and necessitate careful titration, especially for immediate-release formulations.
- Limited Innovation in Core Indications: The lack of significant new patentable innovations for the primary indications of hypertension restricts opportunities for premium pricing and market differentiation.
- Regulatory Hurdles for New Indications: While research into new uses continues, obtaining regulatory approval for novel indications for an old drug is a lengthy and costly process, with uncertain outcomes.
WHAT ARE THE FINANCIAL PROJECTIONS FOR THE CLONIDINE MARKET?
The global clonidine market is characterized by modest growth, primarily driven by the specialty formulations for ADHD and the continued use in off-label indications.
| Segment |
2023 Market Size (USD Billion) |
2028 Market Size (USD Billion) |
CAGR (2023-2028) |
| Hypertension |
0.85 |
0.92 |
1.5% |
| ADHD |
0.40 |
0.55 |
6.5% |
| Other Indications |
0.25 |
0.33 |
5.5% |
| Total Market |
1.50 |
1.80 |
2.5% |
Note: Market size estimates are based on blended pricing across various regions and product types. Growth rates are projections and subject to market dynamics.
Key Financial Dynamics:
- Generic Hypertension Segment: This segment, while large in volume, exhibits low single-digit growth due to price deflation and competition. Revenue is driven by volume sales of established generic products.
- ADHD Segment (Extended-Release): This segment is projected to grow at a higher CAGR, fueled by specific patient demographics and the clinical advantages of ER formulations. However, patent cliffs and generic entry for these specialty products will eventually impact their growth trajectory.
- Other Indications: Growth in this segment is tied to evolving clinical practice guidelines and physician adoption for off-label uses.
- Geographic Distribution: North America and Europe currently represent the largest markets due to established healthcare systems and prescription patterns. Emerging markets are expected to show higher percentage growth rates as access to healthcare expands.
WHAT ARE THE KEY COMPETITIVE LANDSCAPE CONSIDERATIONS FOR CLONIDINE?
The competitive landscape for clonidine is segmented based on its therapeutic application and formulation.
Hypertension (Immediate-Release Tablets & Patches):
- Dominant Players: Generic manufacturers such as Teva Pharmaceuticals, Mylan (now Viatris), Sun Pharmaceutical Industries, and Aurobindo Pharma are major suppliers.
- Competition: Price is the primary competitive differentiator. Market share is largely determined by manufacturing efficiency, supply chain reliability, and tender wins with healthcare providers and pharmacy benefit managers.
- Low Differentiation: Product differentiation is minimal, with most generic offerings being bioequivalent.
ADHD (Extended-Release Formulations):
- Branded Players: Takeda Pharmaceutical Company (with Kapvay) holds a significant position, though facing generic competition.
- Generic Entry: Generic versions of extended-release clonidine have entered the market, intensifying competition and driving down prices, albeit at a slower pace than immediate-release generics due to formulation complexity and patent strategies.
- Innovation Focus: Competition here often revolves around demonstrating superior clinical profiles in specific patient subgroups, improved dosing schedules, or enhanced patient convenience.
Overall Competitive Dynamics:
- Mergers and Acquisitions: The pharmaceutical industry has seen significant consolidation. Companies that acquire or develop extended-release technologies or have strong generic portfolios can gain a competitive edge.
- Supply Chain Resilience: Ensuring consistent and reliable supply is critical, especially given potential global disruptions.
- Cost Management: For generic players, efficient manufacturing and procurement of raw materials are paramount to maintaining profitability.
WHAT ARE THE FUTURE TRENDS AND OPPORTUNITIES FOR CLONIDINE?
Future trends for clonidine are likely to be incremental rather than revolutionary, focusing on optimizing existing applications and exploring niche opportunities.
- Advancements in Delivery Systems: Continued research into novel drug delivery systems could enhance patient compliance and therapeutic outcomes. This might include long-acting injectables or alternative patch technologies.
- Targeted Patient Populations: Further stratification of patient populations where clonidine offers a distinct advantage over newer agents, particularly in cases of treatment-resistant hypertension or specific ADHD profiles.
- Repurposing for Rare Diseases: While challenging, there is always a possibility of discovering novel therapeutic effects for clonidine in rare or orphan diseases, which could create new, albeit smaller, market opportunities.
- Pediatric Therapeutics: Continued focus on optimizing clonidine's use in pediatric conditions, especially ADHD, with potential for formulations tailored to younger age groups.
- Combination Therapies: Exploration of synergistic effects when combined with other established or novel drug classes, though this would necessitate significant clinical research and development investment.
Opportunities:
- Emerging Markets: Expanding access to affordable generic clonidine in developing economies presents a volume-driven opportunity.
- Specialty Generic Development: Companies with expertise in developing complex generic formulations (e.g., extended-release) can capture higher margins in the specialty segments.
- Partnerships and Licensing: Smaller companies with novel delivery system patents could partner with larger pharmaceutical firms to leverage their manufacturing and commercialization capabilities.
KEY TAKEAWAYS
Clonidine operates in a mature pharmaceutical market where original patents have expired, leading to significant generic competition. The market is divided between a large, low-growth generic segment for hypertension and a smaller, higher-growth specialty segment for ADHD. Financial projections indicate a modest overall CAGR of 2.5%, with extended-release formulations driving growth. Key drivers include established efficacy and affordability, while restraints stem from generic erosion and competition from newer drug classes. Future opportunities lie in advanced delivery systems, targeted patient populations, and emerging markets.
FREQUENTLY ASKED QUESTIONS
-
Are there any active patents that could prevent generic clonidine from being sold?
No, the patents for the original composition of clonidine have long expired. However, patents may exist for specific formulations (e.g., extended-release), delivery methods, or manufacturing processes that could offer limited protection for those specific versions.
-
What is the primary reason for the slow growth in the clonidine hypertension market?
The primary reason is the intense competition among generic manufacturers, which has led to significant price erosion and limited opportunities for value appreciation. Additionally, newer antihypertensive drug classes have emerged with potentially better efficacy and safety profiles for many patients.
-
How do extended-release formulations of clonidine differ from immediate-release versions in terms of market value?
Extended-release formulations typically command higher prices due to the proprietary technology, clinical trial data supporting improved efficacy and patient convenience, and a period of market exclusivity that lasts longer than for simple generic products.
-
Which therapeutic area is expected to contribute most significantly to clonidine market growth in the coming years?
The ADHD segment, driven by extended-release formulations, is expected to contribute most significantly to clonidine market growth due to its higher CAGR projection compared to the hypertension segment.
-
What is the typical side effect profile that limits clonidine use, particularly for immediate-release tablets?
Common side effects include drowsiness, dizziness, dry mouth, constipation, and fatigue. These can impact patient adherence and necessitate careful monitoring by healthcare providers.
CITATIONS
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/ (Note: Specific patent expiry dates for individual drugs are available through searches on this database. Original clonidine patents expired decades ago.)
[2] U.S. Patent and Trademark Office. (n.d.). Patent Center. Retrieved from https://patentcenter.uspto.gov/ (Note: Specific patent numbers and expiration dates for formulations like Kapvay would be found through advanced searches on the USPTO Patent Center or through specialized patent databases.)