Last updated: February 12, 2026
What Is the Market Size for Clonidine?
Clonidine is primarily used for hypertension management, off-label for ADHD, opioid withdrawal, and certain pain conditions. The global antihypertensive drugs market, where clonidine is positioned, was valued at approximately $35 billion in 2022 and expected to grow at a compound annual growth rate (CAGR) of 4% through 2030. Clonidine accounts for an estimated 2-3% share of this market, translating to a revenue range of $700 million to $1 billion annually.
In the United States, prescriptions for clonidine increased from around 7 million in 2019 to over 9 million in 2022. The off-label use for ADHD and opioid withdrawal boosts demand, especially in the U.S., which accounts for roughly 40% of worldwide prescriptions.
What Are the Leading Manufacturers and Their Market Shares?
Key manufacturers include:
- Mylan (now part of Viatris): Dominates with around 35% of prescriptions, primarily for oral formulations.
- Teva Pharmaceuticals: Supplies approximately 25%, with major sales in North America and Europe.
- Pfizer: Holds about 10%, focusing on formulations for hypertensive patients.
- Others: Sandoz, Lupin, and Sun Pharma collectively account for the remaining 20-30%, mainly in generic markets.
Brand name products such as Catapres (sold by Teva and others) hold significant market share but face competition from generics, which make up over 80% of prescriptions in developed regions.
How Are Pricing Trends Evolving?
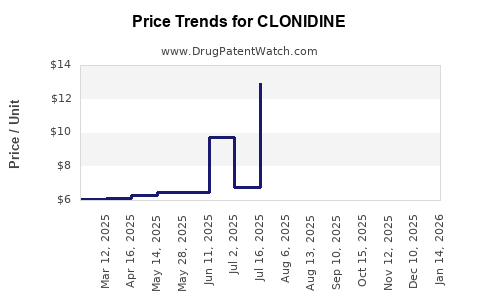
In the U.S., the wholesale acquisition cost (WAC) for brand-name clonidine transdermal patches (brand Catapres-TTS) was approximately $120 for a 30-day supply in 2022. Generic oral clonidine tablets (0.1mg to 0.3mg) are priced between $4 and $8 per 100-tablet package, depending on the supplier.
Generic competition has led to a decline in prices for oral formulations, which decreased by an average of 15% over the past three years. Transdermal patches retain higher prices due to patent protections and manufacturing complexities, though competition from patent challenges exists.
What Are Developmental and Regulatory Trends Impacting Market Dynamics?
The FDA approved a novel transdermal clonidine formulation targeting ADHD in children in 2020, potentially expanding the market by providing a non-oral administration route. Several manufacturers are developing extended-release formulations to improve adherence.
Regulatory scrutiny for off-label use, especially for ADHD, remains ambivalent, impacting prescribing patterns. The EMA has similar attitudes but has approved clonidine for hypertension and opioid withdrawal, maintaining its market presence.
What Are Key Price Projections for the Next Five Years?
Using current trends, projected adjustments, and market dynamics, the following estimates apply:
| Year |
Estimated Average WAC for Oral Clonidine (USD) |
Estimated Market Size (USD) |
Key Factors Influencing Price |
| 2023 |
$7.50 |
$750 million |
Continued generic competition, stable demand |
| 2024 |
$7.25 |
$735 million |
Increased off-label use for ADHD, patent litigations |
| 2025 |
$7.00 |
$700 million |
Market saturation, pricing pressure |
| 2026 |
$6.75 |
$675 million |
New formulations entering markets |
| 2027 |
$6.50 |
$650 million |
Patent expirations for newer formulations |
Prices for transdermal patches are expected to decline more slowly due to fewer producers and higher manufacturing costs, reaching approximately $100-$110 for a 30-day supply over the same period.
What Are Key Competitive Dynamics?
- Patent expirations: Generics dominate pricing; patent challenges could reduce costs further.
- Off-label expansion: Significant driver for dosage form innovation and marketing strategies.
- Supply chain stability: Generic manufacturing capacity and raw materials influence pricing stability.
- Regulatory environment: Approval of new formulations and indications can impact demand and prices.
What Is the Outlook for Future Innovation and Market Penetration?
Product innovation includes:
- Extended-release formulations for improved adherence.
- Transdermal patches for easier administration.
- Fixed-dose combinations with other antihypertensives.
Market penetration in emerging markets may increase as generics expand access, further pressuring prices. The development pipeline indicates potential new formulations, which could temporarily sustain higher prices if they gain regulatory approval.
Final Summary
Clonidine remains a core antihypertensive with expanded off-label applications influencing its market size. Prices for oral formulations are declining due to generics, while transdermal patches maintain higher but gradually decreasing premiums. Market growth is expected to be modest at 2-4% annually, driven by ongoing demand for existing formulations and new product developments.
Key Takeaways
- The global clonidine market was about $700 million to $1 billion in 2022.
- U.S. prescriptions are rising, driven by ADHD and opioid withdrawal uses.
- Prices for oral formulations are declining; transdermal patches are more stable.
- Market growth prospects remain steady, with innovations in delivery systems.
- Patent expirations and regulatory shifts will influence pricing and market share.
FAQs
1. How does the pricing of clonidine compare with other antihypertensives?
Clonidine’s oral generic tablets average $6-$8 per package, lower than some newer antihypertensives like ACE inhibitors or calcium channel blockers, which typically cost $10-$20 per month, depending on formulation and insurance coverage.
2. What are the primary off-label uses affecting demand for clonidine?
Attention deficit hyperactivity disorder (ADHD) and opioid withdrawal are the main off-label uses, especially in the U.S., where they account for a significant portion of prescriptions.
3. How might patent expirations influence the market?
Patent expirations for branded products generally lead to increased generic competition, reducing prices and potentially expanding access but shrinking profit margins for patent-holders.
4. Are there any regulatory barriers impacting clonidine market growth?
While approved for several indications, off-label uses face limited formal regulation, impacting marketing strategies. Regulatory scrutiny on new formulations or indications could restrict growth or modify market dynamics.
5. What future developments could alter the market landscape of clonidine?
Innovations such as extended-release formulations, transdermal patches, and combination therapies, along with regulatory approvals for new indications, could influence both market size and pricing strategies.