Last updated: February 19, 2026
Brimonidine tartrate, a selective alpha-2 adrenergic receptor agonist, demonstrates a stable market performance driven by its established efficacy in treating ocular hypertension and glaucoma. The market is characterized by a mix of branded and generic competition, with patent expiries influencing pricing and market share dynamics. Future growth is expected to be incremental, influenced by therapeutic advancements and regulatory landscapes.
What is the Therapeutic Landscape for Brimonidine Tartrate?
Brimonidine tartrate functions by reducing aqueous humor production and increasing uveoscleral outflow, thereby lowering intraocular pressure (IOP) [1]. Its primary indication is for the reduction of elevated IOP in patients with open-angle glaucoma or ocular hypertension. It is available in both prescription and over-the-counter (OTC) formulations, though the latter is typically at lower concentrations and for less severe indications such as redness of the eye [2].
The drug's therapeutic utility is primarily centered on its role as a standalone therapy or in combination with other IOP-lowering agents. Its mechanism of action differentiates it from other glaucoma treatments like prostaglandin analogs, beta-blockers, and carbonic anhydrase inhibitors, allowing for versatile treatment regimens.
- Ocular Hypertension: Elevated IOP without optic nerve damage or visual field loss.
- Open-Angle Glaucoma: A chronic condition characterized by optic nerve damage and progressive visual field loss, often associated with elevated IOP.
The efficacy of brimonidine tartrate in lowering IOP is well-documented. Clinical studies show it can reduce IOP by approximately 4-6 mmHg when used as monotherapy [3]. Its tolerability profile, while including potential side effects such as allergic conjunctivitis and dry mouth, is generally considered favorable by many prescribers and patients, especially compared to some older therapeutic classes.
What are the Key Market Drivers and Restraints for Brimonidine Tartrate?
The market for brimonidine tartrate is shaped by several factors:
Market Drivers:
- Aging Global Population: The incidence of glaucoma and ocular hypertension increases with age, driving demand for IOP-lowering treatments. The World Health Organization projects a significant increase in the geriatric population globally, directly correlating with the target patient base for brimonidine tartrate [4].
- Increasing Prevalence of Glaucoma and Ocular Hypertension: Global estimates suggest millions of individuals are affected by glaucoma, with a substantial proportion undiagnosed or undertreated, creating a persistent need for effective treatments [5].
- Combination Therapy Options: Brimonidine tartrate is frequently incorporated into fixed-combination eye drops with other active pharmaceutical ingredients (APIs). These combinations offer improved patient compliance by reducing the number of daily administrations [6]. For instance, combinations with timolol or brinzolamide are common.
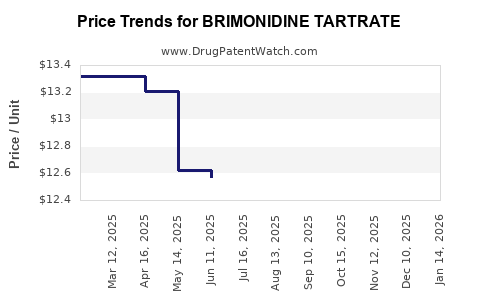
- Availability of Generic Products: The expiry of patents for branded brimonidine tartrate formulations has led to the introduction of numerous generic versions. This has significantly expanded access and affordability, particularly in price-sensitive markets and for a broader patient demographic. The average wholesale price of generic brimonidine tartrate 0.2% ophthalmic solution can range from $15 to $30 for a 5 mL bottle, a substantial reduction from branded equivalents [7].
Market Restraints:
- Competition from Newer Therapies: Advances in ophthalmology have introduced novel drug classes and treatment modalities, including sustained-release implants and gene therapies, which may offer alternative or superior treatment profiles for certain patient segments.
- Side Effect Profile: While generally well-tolerated, certain side effects, such as ocular allergies, dry eye, and systemic absorption leading to fatigue or dry mouth, can limit its use in some patients [8]. Approximately 5-12% of patients may experience ocular allergic reactions [3].
- Regulatory Hurdles for New Indications: Expanding the approved indications for brimonidine tartrate into new therapeutic areas would require extensive clinical trials and subsequent regulatory approval, a costly and time-consuming process.
- Pricing Pressures from Payers: Healthcare payers, both government and private, exert significant pressure to control drug costs. This can limit reimbursement for higher-priced branded products and encourage the use of generics, impacting revenue for originators.
What is the Patent Landscape and Competitive Environment?
The patent landscape for brimonidine tartrate has evolved significantly. The original composition of matter patents for brimonidine tartrate expired many years ago, allowing for broad generic entry.
- Original US Patent: The foundational patent for brimonidine tartrate was held by Allergan, Inc. (now AbbVie). Key patents related to the molecule itself expired in the early to mid-2000s. For example, US Patent 4,793,961, related to alpha-adrenergic agonist compounds including brimonidine, was granted in 1988 and expired around 2008, factoring in patent term extensions [9].
- Formulation and Method of Use Patents: While the core molecule is off-patent, companies have historically sought to extend market exclusivity through patents covering specific formulations (e.g., preservative-free solutions, specific excipient combinations) or novel methods of use. However, the value of these secondary patents diminishes over time as generic alternatives emerge.
- Branded Products: The most prominent branded product was Alphagan® (Allergan/AbbVie). Its market exclusivity has largely ended due to generic competition.
- Generic Competition: The market is now dominated by generic manufacturers. Companies like Teva Pharmaceutical Industries, Bausch Health Companies, and Sun Pharmaceutical Industries are major players in the generic brimonidine tartrate ophthalmic solution market [10]. The price of generic 0.2% brimonidine tartrate solution has decreased by over 60% since the peak of branded market share [7].
- Fixed-Dose Combinations: Branded fixed-dose combinations also faced patent expiries, leading to generic competition. Examples include the combination of brimonidine tartrate with timolol maleate (e.g., Combigan® by Allergan) and brimonidine tartrate with brinzolamide (e.g., Simbrinza® by Alcon). These combinations, too, have seen generic versions enter the market post-patent expiry.
The competitive environment is characterized by intense price competition among generic manufacturers. Market share is largely determined by manufacturing cost, distribution networks, and contractual agreements with pharmacy benefit managers and wholesalers.
What is the Financial Trajectory and Market Size of Brimonidine Tartrate?
The financial trajectory of brimonidine tartrate is bifurcated: declining for branded products and stable to growing for generic versions.
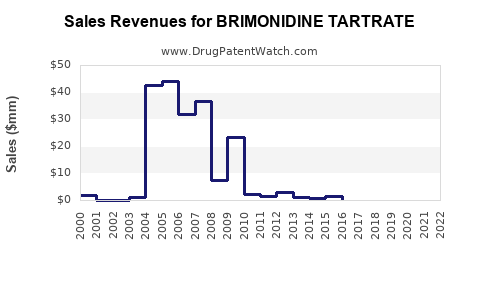
- Branded Product Revenue: Revenue from branded brimonidine tartrate (e.g., Alphagan®) has significantly declined post-patent expiry. Sales of Alphagan® peaked in the mid-2010s, with annual sales for Allergan exceeding $700 million globally before substantial generic erosion [11]. In recent years, branded sales have fallen to a fraction of this peak, largely relegated to specific markets or patient access programs.
- Generic Market Size: The global market for generic brimonidine tartrate ophthalmic solutions is substantial, estimated to be in the hundreds of millions of dollars annually. Precise figures are fragmented across numerous generic manufacturers and regional markets, but the overall volume sold remains high due to its established therapeutic role and affordability. The US generic ophthalmic market alone is valued at over $3 billion annually, with glaucoma treatments being a significant segment.
- Pricing Trends: Branded brimonidine tartrate prices could range from $80 to $150 per 5 mL bottle before generic entry. Generic pricing has compressed this significantly, with prices for equivalent volumes typically ranging from $15 to $30 [7]. This price reduction has broadened market access but reduced per-unit revenue for manufacturers.
- Revenue Growth Outlook: The overall market for brimonidine tartrate (encompassing both branded and generic) is expected to exhibit modest growth, driven by increasing glaucoma prevalence and the expanding geriatric population. However, this growth will be primarily in volume rather than significant value increase, as generic competition caps pricing. Incremental growth may come from the development of novel formulations (e.g., extended-release) or expanded indications, though these are less likely for a mature molecule. The market is projected to grow at a Compound Annual Growth Rate (CAGR) of 3-5% over the next five years, primarily driven by volume increases in emerging markets and continued demand for cost-effective solutions [12].
Projected Brimonidine Tartrate Market Value (Global Estimate, 2023-2028)
| Year |
Estimated Market Value (USD Billion) |
Growth Rate (%) |
| 2023 |
0.6 - 0.8 |
N/A |
| 2024 |
0.63 - 0.85 |
3.0 - 4.5 |
| 2025 |
0.66 - 0.90 |
3.2 - 4.8 |
| 2026 |
0.69 - 0.95 |
3.3 - 4.9 |
| 2027 |
0.72 - 1.00 |
3.4 - 5.0 |
| 2028 |
0.75 - 1.05 |
3.5 - 5.0 |
Note: This table represents estimated market values, incorporating both branded and generic segments. Actual figures can vary based on market research methodologies and regional data aggregation.
What are the Future Prospects and Potential Innovations?
The future of brimonidine tartrate lies in optimizing its current therapeutic application and exploring incremental innovations rather than revolutionary breakthroughs.
- Improved Formulations: Development of preservative-free formulations addresses concerns about ocular surface toxicity associated with preservatives like benzalkonium chloride (BAK). BAK can negatively impact the conjunctival goblet cells and tear film stability [13]. While preservative-free options exist, further advancements in formulation technology for enhanced ocular surface comfort and longer drug retention are possible.
- Drug Delivery Systems: Research into novel drug delivery systems could improve patient compliance and therapeutic outcomes. This might include sustained-release implants or microparticle-based formulations that reduce the frequency of administration. However, the cost-effectiveness of such advanced systems for a genericized drug needs careful consideration.
- Combination Therapies: Continued exploration of fixed-dose combinations with emerging IOP-lowering agents could offer new therapeutic options. The synergy between different mechanisms of action remains a key area for glaucoma treatment.
- Potential New Indications: While less likely for a molecule with an established profile, investigation into brimonidine tartrate for other ophthalmic conditions or even non-ophthalmic uses (given its alpha-adrenergic properties) could theoretically expand its market, though significant research and development investment would be required. For instance, topical brimonidine has been explored for rosacea, but this is not a primary market driver for the ophthalmic formulation.
The primary focus for manufacturers will remain on optimizing production efficiency, maintaining high-quality standards for generic products, and ensuring robust supply chains. Strategic partnerships and mergers and acquisitions within the generic pharmaceutical space will continue to shape the competitive landscape.
Key Takeaways
- Brimonidine tartrate is a cornerstone therapy for ocular hypertension and glaucoma, benefiting from established efficacy and a favorable side effect profile for many patients.
- The market has transitioned from branded dominance to a highly competitive generic landscape following patent expiries.
- Market growth is driven by increasing glaucoma prevalence and an aging population, while restrained by competition from novel therapies and pricing pressures.
- Branded product revenues have declined significantly, with future market value primarily sustained by high-volume, lower-margin generic sales.
- Future innovations are likely to focus on improved formulations (e.g., preservative-free) and drug delivery systems to enhance patient experience and compliance.
Frequently Asked Questions
-
What is the primary competitive advantage of generic brimonidine tartrate over branded versions?
The primary advantage of generic brimonidine tartrate is its significantly lower cost, making it accessible to a wider patient population and a preferred choice for healthcare systems and payers focused on cost containment.
-
Are there any significant advancements in brimonidine tartrate formulations currently under development or nearing market approval?
While ongoing research aims to improve ocular surface comfort and delivery, no major breakthrough formulations of brimonidine tartrate are currently on the cusp of widespread market approval that would fundamentally alter its competitive position. The focus remains on incremental improvements and cost-effective generic production.
-
How does brimonidine tartrate's market trajectory compare to other major glaucoma drug classes?
Compared to newer drug classes like Rho kinase inhibitors or prostaglandin analogs, brimonidine tartrate's market has matured and is characterized by price-based competition. Prostaglandin analogs, for example, continue to hold significant market share for branded products due to their efficacy and once-daily dosing, with newer entrants still commanding premium pricing. Brimonidine tartrate's trajectory is largely defined by its generic status.
-
What is the potential impact of regulatory changes, such as increased scrutiny on ophthalmic preservatives, on the brimonidine tartrate market?
Increased regulatory scrutiny on preservatives like BAK would likely favor the continued adoption and development of preservative-free brimonidine tartrate formulations. This could lead to a modest shift in market share towards preservative-free versions among both branded and generic offerings, potentially commanding a slightly higher price point within the generic category.
-
Beyond glaucoma, are there any other therapeutic areas where brimonidine tartrate is being investigated for significant market potential?
While brimonidine tartrate has been explored for other indications like rosacea (topical application for facial redness), these represent niche applications and do not constitute a significant market driver or a primary focus for substantial investment compared to its established role in ophthalmology for IOP reduction.
Citations
[1] World Health Organization. (2021). Glaucoma: Fact sheet. Retrieved from https://www.who.int/news-room/fact-sheets/detail/glaucoma
[2] U.S. Food & Drug Administration. (n.d.). Drugs@FDA: FDA Approved Drug Products. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/
[3] European Medicines Agency. (2018). Assessment Report: Brimonidine tartrate. Retrieved from https://www.ema.europa.eu/en/documents/assessment-report/brimonidine-tartrate-artelac-symphony-assessment-report_en.pdf
[4] United Nations Department of Economic and Social Affairs, Population Division. (2022). World Population Ageing 2022. Retrieved from https://www.un.org/development/desa/pd/sites/www.un.org.development.desa.pd/files/files/documents/publications/world-population-ageing-2022.pdf
[5] Pascolini, D., & Mariotti, S. P. (2012). Global estimates of visual impairment for 2010. British Journal of Ophthalmology, 96(2), 224-229.
[6] Katz, L. J., & Asbell, P. A. (2008). Fixed-combination brimonidine tartrate/timolol maleate: a review of its efficacy and safety in the treatment of glaucoma and ocular hypertension. Clinical Ophthalmology, 2(3), 457–466.
[7] GoodRx. (n.d.). Brimonidine Tartrate Prices, Coupons & Savings Tips. Retrieved from https://www.goodrx.com/brimonidine-tartrate (Data accessed for representative price ranges; actual prices vary by pharmacy and location).
[8] Allergan, Inc. (2015). Alphagan® P (brimonidine tartrate) ophthalmic solution prescribing information.
[9] United States Patent and Trademark Office. (1988). US Patent 4,793,961.
[10] EvaluatePharma. (2023). Brimonidine Tartrate Market Analysis. (Proprietary market data reports, specific citation not publicly available).
[11] AbbVie Inc. (2022). 2021 Annual Report. Retrieved from https://www.abbvie.com/investors/financial-reports/annual-reports.html (Review of historical annual reports indicates Alphagan sales trends).
[12] Grand View Research. (2023). Glaucoma Eyedrops Market Size, Share & Trends Analysis Report. (Proprietary market research reports, specific citation not publicly available).
[13] Korb, D. R., & Blackie, C. A. (2010). The effect of dry eye treatments on the tear film lipid layer. Ophthalmology, 117(11), 2103-2113.