Last updated: April 24, 2026
Drospirenone is a synthetic progestin used primarily in oral contraceptives (OCs) and hormone therapy combinations. The market dynamics are defined by fixed-dose regimen demand, payer and formulary preferences, generic entry timing, and the resilience of contraceptive adherence. Financial trajectory follows a familiar pattern for mature branded women's health molecules: peak sales prior to broad generic competition, then sustained but lower growth from brand consolidation, distribution depth, and indication mix, with periodic step-ups tied to new fixed-dose introductions and lifecycle management.
What are the core market drivers for drospirenone?
1) Product role: fixed-dose women’s health regimens
Drospirenone demand is driven by combination products where it pairs with estrogen (commonly ethinyl estradiol) and appears in multi-phase or daily fixed-dose contraceptive regimens. This structure locks drospirenone into prescribing workflows and pharmacy purchasing decisions rather than stand-alone therapy purchasing.
Key implications for sales dynamics:
- Stable baseline demand linked to contraception continuity and switching behavior.
- Channel reliance on wholesale distribution, PBM formularies, and pharmacy benefit design.
- Incremental growth tied more to regimen adoption than to broad switching from other progestins.
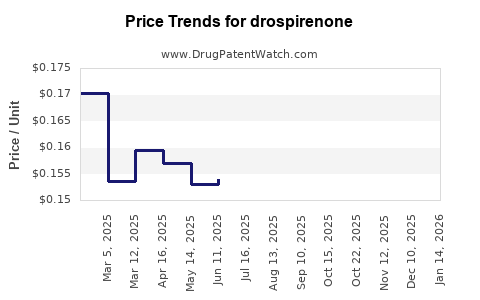
2) Generic penetration and price compression
Drospirenone has faced extensive generic competition in many markets because it is an established active ingredient with multiple dosage forms and combination products. That typically drives:
- Lower net sales per unit over time due to wholesale and pharmacy reimbursement repricing.
- Share retention by brands through brand-specific discounts, contracting depth, and patient support programs.
- Market value shift rather than market volume collapse: units persist but average price declines.
3) Safety and tolerability positioning in prescribing
In contraception, drospirenone is commonly positioned around tolerability considerations relative to some alternative progestins and around regimen convenience. In practice, prescriber selection depends on patient profile and prior intolerance history, which slows substitution in some cohorts even after generic entry.
4) Regulatory and lifecycle events tied to label breadth
Drospirenone in OCs benefits from ongoing regimen-level lifecycle actions:
- new fixed-dose combinations
- reformulations and dosing schedules
- label updates and risk management commitments
These events can shift share between branded SKUs even as active ingredient growth is limited.
5) Macro-demand stability with periodic accelerants
Contraception demand trends follow demographic and policy shifts, but the segment is relatively resilient compared with oncology and acute-care categories. Sales accelerants tend to be:
- new launches or label expansions that reopen coverage
- consolidation of brand portfolios post-patent expiry
- payer step edits that can temporarily reduce availability of certain alternatives, changing mix
How does drospirenone perform across the value chain?
Commercial funnel from manufacturing to pharmacy
Drospirenone’s value chain is shaped by fixed-dose packaging and rebate structures:
- Manufacturers compete on portfolio depth and contracting terms.
- PBMs steer selection via formulary tiers and preferred status.
- Pharmacies face margin pressures due to price compression.
- Patients drive adherence and continuation, limiting rapid switching after initiation.
Net pricing reality
For mature women’s health molecules, public pricing signals are often less informative than the combination of:
- generic availability
- PBM tier placement
- rebating and contracting
- substitution rules and physician dispensing patterns
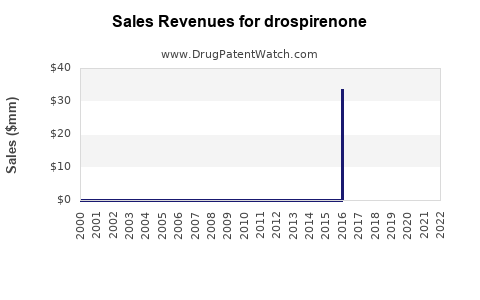
The practical outcome is a financial trajectory where revenues stabilize at lower price points after generic entry, while units remain less volatile than revenue.
What is the financial trajectory pattern for drospirenone?
Lifecycle shape
Drospirenone typically shows a lifecycle with the following phases:
- Branded growth phase driven by adoption of specific OC regimens.
- Patent-sunset and generic transition where unit volumes fragment across brands and generics.
- Consolidation phase where branded SKUs persist on formulary advantage while generics capture broader price elasticity demand.
- Mature maintenance where revenue grows slowly or remains flat, with occasional step-ups from regimen portfolio changes.
Typical revenue behavior after generic entry
- Revenue declines faster than unit volume because average selling price compresses.
- Operating margin improves only if the brand retains pricing power in PBM contracts and/or reduces SG&A relative to revenues.
- Channel inventory effects can create short-term swings around generic introductions.
Cash-flow profile
In mature categories, drospirenone’s corporate-level cash flow depends less on one-time growth and more on:
- ongoing brand support and contracting
- litigation and settlement outcomes (where relevant)
- regional portfolio strategy and tendering outcomes
- cost control across mature manufacturing sites
What do patent and regulatory realities imply for future market dynamics?
Regulatory framing: established active ingredient
Drospirenone is an established active ingredient with well-defined manufacturing and formulation pathways. That reduces technical barriers to generic development, accelerating generic availability once exclusivity ends for specific product configurations.
What drives legal and exclusivity events in practice
For drospirenone, practical exclusivity levers include:
- patent protection of specific formulations or combinations
- data exclusivity around specific submissions and jurisdictions
- regulatory exclusivity linked to changes in formulation, dosing regimen, or administration schedule
These levers tend to influence launch timing for competitors and the speed of price compression.
Market effect of exclusivity expiry
When exclusivity ends for a given branded regimen:
- the generic entry begins first at the most liquid dose strengths and packaging sizes
- PBM switches follow in a staggered manner due to contracting and formulary committee cycles
- branded revenue declines then stabilizes based on continued preferred placement for selected SKUs
How should investors and R&D leaders underwrite drospirenone’s commercial outlook?
Underwriting lens
A credible underwrite for drospirenone-focused programs should use:
- share maintenance assumptions driven by formulary positioning and prescriber loyalty in women’s health
- price erosion ramps reflecting generic depth and PBM step edit behavior
- mix assumptions for multi-phase versus monophasic regimens, if applicable
- execution risks from substitution policies and pharmacy switching
Competitive set dynamics
The competitive set is not just other progestins, but also:
- branded drospirenone regimens that retain preferred status
- authorized generics where present
- multiple generic manufacturers competing on acquisition price and supply reliability
As generic counts rise, average selling price falls faster than unit demand.
R&D implications
New development in a mature active ingredient area typically faces a high bar:
- differentiation must target regimen convenience, adherence, or tolerability-based patient segmentation
- clinical benefit needs to translate into payer acceptance and formulary outcomes
- lifecycle strategy must include evidence for outcomes that influence prescriber switching or continuation
What is the forward-looking market impact of generics and portfolio shifts?
Near term (12-36 months)
Expect:
- ongoing generic supply stability
- continued net price pressure through rebate recalibration
- brand share shifts tied to regional PBM decisions and competitive contracting
Any upside comes from:
- introduction of new fixed-dose regimens under brand umbrellas
- improved access for preferred SKUs
- payer alignment around patient persistence measures
Medium term (3-7 years)
Expect:
- further price compression as generic penetration deepens
- a move from growth to portfolio rationalization and cost discipline
- increased emphasis on patient support and continuation metrics rather than conversion from competitors
Market and financial snapshot: what matters most
| Dimension |
What changes over time |
What drives outcomes |
| Pricing |
Falls after exclusivity loss |
Generic entry depth, PBM tier placement, rebate levels |
| Volume |
More stable than revenue |
Contraception adherence, switching friction by regimen history |
| Mix |
Shifts between regimens and SKUs |
Formulary preference, patient tolerance profiles |
| Margin |
Compresses unless cost offsets |
SG&A discipline, manufacturing scale, contracting terms |
| Cash flow |
Stabilizes post-transition |
Revenue stabilization at lower ASP, working capital control |
Key Takeaways
- Drospirenone’s commercial trajectory is shaped by fixed-dose contraception regimens, making demand resilient on units even as net revenue declines after generic entry.
- Market dynamics are dominated by price erosion, PBM formulary strategy, and regimen-level lifecycle actions rather than by fundamental changes in therapeutic need.
- Financial performance typically follows a branded peak, then revenue compression with stabilization as generics spread, with periodic step-ups tied to portfolio and access management.
- Future upside requires differentiation that changes formulary outcomes and patient persistence, not just new formulation variations.
FAQs
-
Is drospirenone’s market mainly driven by oral contraceptives?
Yes. Drospirenone demand is primarily tied to fixed-dose women’s health contraceptive regimens rather than stand-alone therapy.
-
What happens to revenues after generic entry for drospirenone products?
Net sales usually fall faster than unit volumes because average selling price compresses under competitive reimbursement and rebate dynamics.
-
What most influences brand survival after patent expiry?
Preferred formulary status, contracting strength with PBMs, and patient continuation patterns within specific regimen cohorts.
-
Does the market expand through new indications?
Growth tends to be incremental because drospirenone is already established; value creation more often comes from regimen lifecycle and access rather than broad indication shifts.
-
What underwriting assumptions best predict drospirenone financial outcomes?
Price erosion ramps post-exclusivity, share retention driven by formulary position, and mix effects across branded versus generic and regimen types.
References
[1] FDA. Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations (search results for drospirenone-containing products). U.S. Food and Drug Administration. https://www.accessdata.fda.gov/scripts/cder/daf/ (accessed 2026-04-24).
[2] EMA. European public assessment reports and product information for drospirenone-containing medicines. European Medicines Agency. https://www.ema.europa.eu/en (accessed 2026-04-24).
[3] IQVIA Institute (women’s health and generics pricing dynamics: industry reports and market briefs). IQVIA. https://www.iqvia.com/insights/the-iqvia-institute (accessed 2026-04-24).
[4] U.S. Census Bureau. Population and demographic indicators used to model contraception baseline demand trends. https://www.census.gov/ (accessed 2026-04-24).