DAPAGLIFLOZIN Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Dapagliflozin, and what generic alternatives are available?
Dapagliflozin is a drug marketed by Aizant, Ajanta Pharma Ltd, Alembic, Alkem Labs Ltd, Aurobindo Pharma, Biocon Pharma, Cipla, Inventia, Lupin Ltd, Macleods Pharms Ltd, Micro Labs, MSN, Sandoz, Teva Pharms Usa, Zydus Pharms, Cipla Ltd, Sun Pharm, Teva Pharms Usa Inc, and Torrent. and is included in twenty-seven NDAs.
The generic ingredient in DAPAGLIFLOZIN is dapagliflozin; saxagliptin hydrochloride. There are twenty-six drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the dapagliflozin; saxagliptin hydrochloride profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Dapagliflozin
A generic version of DAPAGLIFLOZIN was approved as dapagliflozin; saxagliptin hydrochloride by MSN on April 6th, 2026.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for DAPAGLIFLOZIN?
- What are the global sales for DAPAGLIFLOZIN?
- What is Average Wholesale Price for DAPAGLIFLOZIN?
Summary for DAPAGLIFLOZIN
| US Patents: | 0 |
| Applicants: | 19 |
| NDAs: | 27 |
| Finished Product Suppliers / Packagers: | 16 |
| Raw Ingredient (Bulk) Api Vendors: | 84 |
| Clinical Trials: | 564 |
| Patent Applications: | 3,060 |
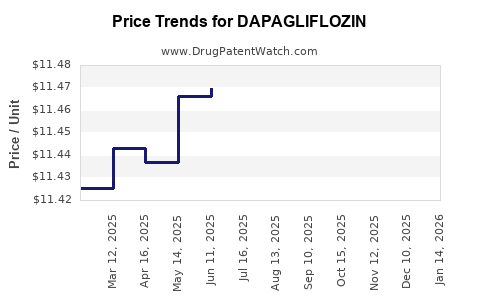
| Drug Prices: | Drug price information for DAPAGLIFLOZIN |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for DAPAGLIFLOZIN |
| What excipients (inactive ingredients) are in DAPAGLIFLOZIN? | DAPAGLIFLOZIN excipients list |
| DailyMed Link: | DAPAGLIFLOZIN at DailyMed |
Recent Clinical Trials for DAPAGLIFLOZIN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Yung NA | PHASE2 |
| Ruijin Hospital | PHASE2 |
| Huashan Hospital | PHASE2 |
Pharmacology for DAPAGLIFLOZIN
| Drug Class | Sodium-Glucose Cotransporter 2 Inhibitor |
| Mechanism of Action | Sodium-Glucose Transporter 2 Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for DAPAGLIFLOZIN
Paragraph IV (Patent) Challenges for DAPAGLIFLOZIN
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| FARXIGA | Tablets | dapagliflozin | 5 mg and 10 mg | 202293 | 20 | 2018-01-08 |
US Patents and Regulatory Information for DAPAGLIFLOZIN
EU/EMA Drug Approvals for DAPAGLIFLOZIN
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| AstraZeneca AB | Forxiga | dapagliflozin | EMEA/H/C/002322Type 2 diabetes mellitusForxiga is indicated in adults and children aged 10 years and above for the treatment of insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exerciseas monotherapy when metformin is considered inappropriate due to intolerance.in addition to other medicinal products for the treatment of type 2 diabetes.For study results with respect to combination of therapies, effects on glycaemic control, cardiovascular and renal events, and the populations studied, see sections 4.4, 4.5 and 5.1.Heart failureForxiga is indicated in adults for the treatment of symptomatic chronic heart failure.Chronic kidney diseaseForxiga is indicated in adults for the treatment of chronic kidney disease. | Authorised | no | no | no | 2012-11-11 | |
| AstraZeneca AB | Edistride | dapagliflozin | EMEA/H/C/004161Type 2 diabetes mellitusEdistride is indicated in adults and children aged 10 years and above for the treatment of insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exerciseas monotherapy when metformin is considered inappropriate due to intolerance.in addition to other medicinal products for the treatment of type 2 diabetes.For study results with respect to combination of therapies, effects on glycaemic control, cardiovascular and renal events, and the populations studied, see sections 4.4, 4.5 and 5.1.Heart failureEdistride is indicated in adults for the treatment of symptomatic chronic heart failure.Chronic kidney diseaseEdistride is indicated in adults for the treatment of chronic kidney disease. | Authorised | no | no | no | 2015-11-09 | |
| Viatris Limited | Dapagliflozin Viatris | dapagliflozin | EMEA/H/C/006006Type 2 diabetes mellitusDapagliflozin Viatris is indicated in adults and children aged 10 years and above for the treatment of insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exercise- as monotherapy when metformin is considered inappropriate due to intolerance.- in addition to other medicinal products for the treatment of type 2 diabetes.For study results with respect to combination of therapies, effects on glycaemic control, cardiovascular and renal events, and the populations studied, see sections 4.4, 4.5 and 5.1.Heart failureDapagliflozin Viatris is indicated in adults for the treatment of symptomatic chronic heart failure with reduced ejection fraction.Chronic kidney diseaseDapagliflozin Viatris is indicated in adults for the treatment of chronic kidney disease. | Authorised | yes | no | no | 2023-03-24 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
Market Dynamics and Financial Trajectory for Dapagliflozin
More… ↓