CHLORPROMAZINE - Generic Drug Details
✉ Email this page to a colleague
What are the generic drug sources for chlorpromazine and what is the scope of freedom to operate?
Chlorpromazine
is the generic ingredient in five branded drugs marketed by Glaxosmithkline, Actavis Mid Atlantic, Genus, Pharm Assoc, Rubicon, Wockhardt, Hikma, Saptalis Pharms, Abraxis Pharm, Aspiro, Deva Hlding, Dr Reddys, Eugia Pharma, Gland, Marsam Pharms Llc, MSN, Thinq Pharm-cro Pvt, Watson Labs, Wyeth Ayerst, Zydus Pharms, Alpharma Us Pharms, Chartwell Rx, Abbott, Alembic, Amneal Pharms Co, Aurobindo Pharma Ltd, Cycle, Glenmark Pharms Ltd, Ivax Sub Teva Pharms, Kv Pharm, Lannett Co Inc, Lederle, Lupin, Purepac Pharm, Pvt Form, Somerset Theraps Llc, Sun Pharm, Teva Pharms, Upsher Smith Labs, Vangard, West Ward, Zameer Pharms, Zydus Lifesciences, and Parke Davis, and is included in eighty-four NDAs. Additional information is available in the individual branded drug profile pages.There are twenty-four drug master file entries for chlorpromazine.
Summary for CHLORPROMAZINE
| US Patents: | 0 |
| Tradenames: | 5 |
| Applicants: | 44 |
| NDAs: | 84 |
| Drug Master File Entries: | 24 |
| Raw Ingredient (Bulk) Api Vendors: | 74 |
| Clinical Trials: | 40 |
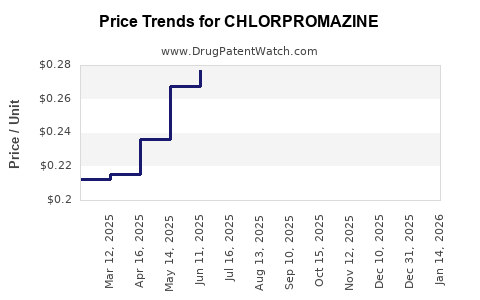
| Drug Prices: | Drug price trends for CHLORPROMAZINE |
| What excipients (inactive ingredients) are in CHLORPROMAZINE? | CHLORPROMAZINE excipients list |
| DailyMed Link: | CHLORPROMAZINE at DailyMed |
Recent Clinical Trials for CHLORPROMAZINE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Ji Xunming,MD,PhD | NA |
| University Hospital, Strasbourg, France | PHASE2 |
| Capital Medical University | PHASE1 |
Medical Subject Heading (MeSH) Categories for CHLORPROMAZINE
Anatomical Therapeutic Chemical (ATC) Classes for CHLORPROMAZINE
US Patents and Regulatory Information for CHLORPROMAZINE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lederle | CHLORPROMAZINE HYDROCHLORIDE | chlorpromazine hydrochloride | TABLET;ORAL | 084803-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Wyeth Ayerst | CHLORPROMAZINE HYDROCHLORIDE | chlorpromazine hydrochloride | INJECTABLE;INJECTION | 080370-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Somerset Theraps Llc | CHLORPROMAZINE HYDROCHLORIDE | chlorpromazine hydrochloride | TABLET;ORAL | 213590-001 | Aug 31, 2020 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Market Dynamics and Financial Trajectory for Chlorpromazine
More… ↓