Last updated: February 19, 2026
Fluconazole, an orally and intravenously administered triazole antifungal medication, has maintained a significant market presence since its initial patent expiration. Its efficacy against a broad spectrum of fungal infections, including Candida species and Cryptococcus neoformans, has cemented its role as a first-line treatment. The compound's pharmacokinetic profile, characterized by good oral bioavailability and penetration into various bodily tissues, further contributes to its clinical utility. While originator patents have long expired, the intellectual property landscape continues to evolve through new formulations, polymorphs, and combination therapies, influencing its market dynamics and the financial trajectory of companies involved in its production and distribution.
What is the Current Patent Status of Fluconazole?
The foundational patents for fluconazole, originally developed by Pfizer Inc. and marketed as Diflucan, have long since expired. The primary U.S. patent (U.S. Patent No. 4,404,216) for fluconazole was granted on September 13, 1983, and expired in 2001. Similar expirations occurred internationally.
However, the patent landscape is not static. Post-expiration, companies have pursued and continue to seek patents related to:
- New Formulations: Patents may cover improved drug delivery systems, such as sustained-release formulations, topical preparations, or novel intravenous solutions designed to enhance efficacy, reduce side effects, or improve patient compliance. For example, patents might claim specific excipients or manufacturing processes that result in a more stable or bioavailable formulation.
- Polymorphs and Crystalline Forms: Different crystalline structures of an active pharmaceutical ingredient (API) can affect its stability, solubility, and manufacturing characteristics. Companies have patented specific polymorphs of fluconazole that offer advantages in production or therapeutic performance. A notable example is the patenting of specific anhydrous and hydrated forms.
- Combination Therapies: Research into combining fluconazole with other antifungal agents or therapeutic compounds to achieve synergistic effects or overcome resistance is ongoing. Patents may be granted for specific ratios or co-formulations of fluconazole with other APIs.
- Manufacturing Processes: While the core synthesis is well-established, innovative and more efficient or environmentally friendly manufacturing processes can be patented. These patents focus on novel synthetic routes, purification methods, or scalable production techniques.
- Methods of Treatment: New uses or specific treatment protocols for fluconazole, particularly for rare or resistant fungal infections, can be the subject of method-of-use patents. These patents claim the use of fluconazole in a specific dosage regimen or for a particular patient population.
The expiration of the original composition-of-matter patents has significantly opened the market to generic manufacturers, leading to price erosion. However, these subsequent patents, though often narrower in scope, can still create opportunities for market differentiation and extended exclusivity for specific innovations.
How Has the Market for Fluconazole Evolved Post-Patent Expiration?
Following the expiration of its primary patents, the fluconazole market has undergone substantial transformation, characterized by increased generic competition and a shift in market dynamics.
Key Market Evolutions:
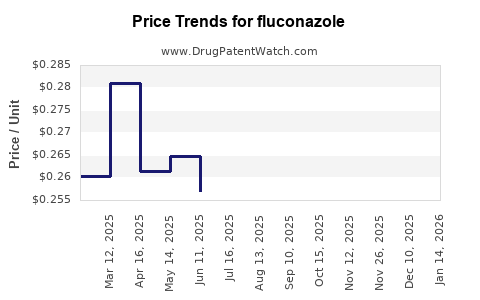
- Generic Entry and Price Compression: The period immediately following patent expiration saw a rapid influx of generic manufacturers. This led to significant price competition, reducing the cost of fluconazole therapy for healthcare systems and patients. The average selling price (ASP) for generic fluconazole has fallen dramatically since its peak. For instance, in the U.S., the cost of a standard 150 mg tablet can range from approximately \$0.20 to \$2.00, a stark contrast to the branded Diflucan pricing prior to generic competition.
- Increased Market Volume: While ASPs decreased, the overall volume of fluconazole prescribed and dispensed increased. The lower cost made the medication more accessible, leading to broader utilization in both hospital and outpatient settings for prophylaxis and treatment of common fungal infections. Global sales volume for fluconazole has remained robust, estimated to be in the hundreds of millions of units annually.
- Dominance of Generic Brands: The market is now dominated by generic brands. Companies that specialize in generic drug manufacturing, such as Teva Pharmaceutical Industries, Mylan N.V. (now part of Viatris), and Sandoz, hold significant market share. These companies leverage efficient manufacturing processes and extensive distribution networks to compete.
- Shift from Brand to Generic Manufacturers: The financial trajectory for the originator, Pfizer, shifted from the high-margin branded product to licensing or divesting rights, and focusing R&D on newer therapeutic areas. Conversely, generic manufacturers have seen sustained revenue from fluconazole based on volume.
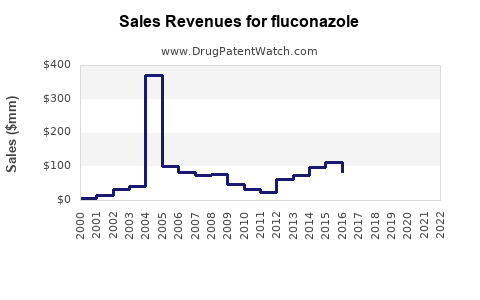
- Therapeutic Niche Persistence: Despite the development of newer antifungal agents (e.g., voriconazole, posaconazole, isavuconazole), fluconazole retains its importance for uncomplicated candidiasis, yeast infections, and prophylaxis in immunocompromised patients due to its favorable safety profile, oral bioavailability, and cost-effectiveness. The estimated annual global market size for fluconazole, considering all formulations and generics, is in the range of \$300 million to \$500 million, with the majority of this revenue derived from generic sales.
- Emergence of Resistance: While fluconazole remains effective against many common fungal pathogens, the increasing prevalence of fluconazole-resistant Candida strains (e.g., C. glabrata, certain C. auris strains) has led to a more nuanced prescribing landscape. This has created opportunities for newer, broader-spectrum antifungals in specific clinical scenarios, but has not eliminated fluconazole's role for susceptible infections.
What is the Financial Trajectory and Market Size of Fluconazole?
The financial trajectory of fluconazole is a textbook example of a drug lifecycle post-patent expiration, transitioning from high-margin branded revenue to volume-driven generic sales.
Financial Data and Projections:
- Peak Branded Revenue: Prior to patent expiry, Diflucan was a blockbuster drug for Pfizer, generating annual sales exceeding \$1 billion in its prime in the late 1990s.
- Generic Market Size: The current global annual market for fluconazole, encompassing all generic formulations and brands, is estimated to be between \$300 million and \$500 million. This figure represents the wholesale acquisition cost (WAC) and does not include rebates or payer discounts.
- Revenue Breakdown: The vast majority of this revenue, approximately 85-90%, is generated by generic manufacturers. The remaining revenue is derived from any niche branded formulations or combination products that may still hold market exclusivity or offer unique benefits.
- Profit Margins: For generic manufacturers, profit margins on fluconazole are significantly lower than those of the originator. However, these are often high-volume products, contributing consistent revenue streams. Margins for established generics typically range from 10-25% on revenue.
- Manufacturing Costs: The manufacturing cost of generic fluconazole API is highly competitive, often in the range of \$20 to \$70 per kilogram, depending on scale and purity. This low cost of goods sold (COGS) is a key driver of profitability for generic players.
- Market Growth Rate: The fluconazole market, as a whole, exhibits low single-digit annual growth (1-3%), primarily driven by population growth and the continued incidence of fungal infections in vulnerable populations. Growth is tempered by the emergence of resistance and the availability of newer agents for specific indications.
- Key Market Participants: Leading generic manufacturers include Viatris (formed by the merger of Mylan and Pfizer's Upjohn division), Teva Pharmaceuticals, Sanofi (through its generics arm), and numerous smaller regional players.
The financial future of fluconazole remains tied to its established role in treating common fungal infections and its cost-effectiveness. While not a growth driver for major pharmaceutical companies, it is a stable, albeit lower-margin, revenue generator for generic manufacturers and a critical, affordable therapeutic option globally.
What are the Key Intellectual Property Strategies Employed for Fluconazole?
Despite the expiration of core patents, companies have continued to employ various intellectual property strategies to maintain market relevance and generate revenue from fluconazole.
Intellectual Property Strategies:
- Formulation Patents:
- Example: Patents claiming specific ratios of fluconazole with permeation enhancers for topical applications.
- Effect: Creates exclusivity for specific delivery methods, potentially capturing niche markets or improving patient adherence.
- Polymorph Patents:
- Example: U.S. Patent No. 6,756,379 describes a novel anhydrous crystalline form of fluconazole, claimed to offer improved stability and processability.
- Effect: Can extend market exclusivity by protecting a therapeutically equivalent but structurally distinct form of the API, requiring generic competitors to avoid the patented form.
- Manufacturing Process Patents:
- Example: Patents for stereoselective synthesis routes that increase yield or reduce hazardous byproducts.
- Effect: Protects proprietary manufacturing know-how, potentially providing a cost advantage or a barrier to entry for those without the patented process.
- Method of Use Patents:
- Example: Patents claiming the use of fluconazole in combination with another agent for treating specific resistant fungal strains, or for particular prophylactic regimens in organ transplant recipients.
- Effect: Extends exclusivity for specific therapeutic applications, even if the base compound is generic. These are often more challenging to enforce due to the need to prove off-label use circumvention.
- Patent Linkage and Data Exclusivity:
- Effect: Regulatory pathways, such as the Hatch-Waxman Act in the U.S., provide periods of market exclusivity based on patent listings in the Orange Book and the initial approval of a New Chemical Entity (NCE). While fluconazole is long off NCE exclusivity, subsequent patent filings can trigger these linkages, leading to litigation and potentially delaying generic entry.
- Evergreening Strategies:
- Effect: The cumulative effect of multiple, often narrow, patents covering minor variations or improvements can create a complex web of intellectual property that can deter or delay generic competition, a practice sometimes referred to as "evergreening."
These strategies, while not granting the broad monopoly of a composition-of-matter patent, can provide limited market protection, create licensing opportunities, and influence the timing and nature of generic competition.
How Does Fluconazole Compare to Newer Antifungal Agents?
Fluconazole occupies a distinct position in the antifungal armamentarium when compared to newer agents. Its profile is characterized by a balance of efficacy, safety, cost, and spectrum of activity.
Comparative Analysis:
| Feature |
Fluconazole |
Newer Azoles (e.g., Voriconazole, Posaconazole, Isavuconazole) |
| Spectrum |
Primarily Candida spp. (except C. glabrata, C. krusei), Cryptococcus neoformans. |
Broader spectrum, including Aspergillus spp., Scedosporium, Fusarium, and more resistant Candida spp. (e.g., C. glabrata, C. auris). |
| Efficacy |
High efficacy against fluconazole-susceptible organisms. First-line for many candidemias. |
Generally higher efficacy against broader range of pathogens and resistant strains. Often preferred for invasive or life-threatening infections. |
| Oral Bioavailability |
Excellent (>90%). |
Variable. Posaconazole has improved oral absorption with food. Isavuconazole has good oral bioavailability. Voriconazole is good but less consistent than fluconazole. |
| CNS Penetration |
Good. |
Variable, generally good. |
| Safety Profile |
Generally well-tolerated. Common side effects: nausea, headache, abdominal pain. Fewer drug-drug interactions than newer azoles. |
More potential for drug-drug interactions (especially CYP450 mediated). Liver toxicity, visual disturbances (voriconazole), and infusion reactions are concerns. |
| Cost |
Very low (generic). |
Significantly higher. Branded pricing can be 10-50 times that of generic fluconazole. |
| Indications |
Vulvovaginal candidiasis, oropharyngeal candidiasis, esophageal candidiasis, candidemia (non-neutropenic), cryptococcal meningitis (initial therapy). |
Invasive aspergillosis, invasive candidiasis (including resistant strains), candidemia, prophylaxis in immunocompromised patients, mucormycosis (posaconazole). |
| Resistance |
Increasing prevalence of resistant Candida spp. (e.g., C. glabrata). |
Developed for strains resistant to fluconazole. Resistance still possible, but generally less common than for fluconazole. |
| Development Stage |
Mature, off-patent. |
Newer, may still have some remaining patent protection or be subject to recent market entry. |
Fluconazole remains a cornerstone for empirical treatment of common fungal infections and prophylaxis in specific patient populations due to its cost-effectiveness and favorable safety profile. Newer agents are reserved for documented or suspected infections by resistant organisms, invasive diseases, or when fluconazole has failed. The continued rise of resistant fungal pathogens, however, necessitates ongoing vigilance and the judicious use of fluconazole.
Key Takeaways
Fluconazole's market trajectory illustrates the profound impact of patent expiration. Originator revenue has been replaced by volume-driven sales for generic manufacturers. While core patents have expired, subsequent intellectual property strategies, such as those for new formulations and polymorphs, continue to shape the competitive landscape. Fluconazole remains a cost-effective and vital antifungal agent for common infections, coexisting with newer, broader-spectrum, and more expensive agents that address resistant strains and invasive diseases. The global fluconazole market, estimated at \$300-500 million annually, is dominated by generic players, with low single-digit growth anticipated.
Frequently Asked Questions
- Are there any active patents for fluconazole that could impact generic manufacturers?
Yes, while the original composition-of-matter patents have expired, patents covering new formulations, specific crystalline forms (polymorphs), manufacturing processes, and methods of use can exist. Generic manufacturers must navigate this patent landscape to ensure their products do not infringe on these later-expiring patents.
- What is the projected growth rate for the global fluconazole market?
The global fluconazole market is projected to experience low single-digit annual growth, estimated between 1% and 3%, driven primarily by population growth and the persistent incidence of fungal infections.
- How has the emergence of antifungal resistance affected fluconazole's market position?
The increasing prevalence of fluconazole-resistant fungal strains has led to more specific prescribing practices. While fluconazole remains a first-line treatment for susceptible infections, newer, broader-spectrum antifungals are increasingly used for resistant pathogens or more severe invasive infections. This has not eliminated fluconazole's market but has refined its therapeutic niche.
- What is the estimated total annual revenue generated by the fluconazole market globally?
The estimated total annual global market for fluconazole, encompassing all generic and any remaining branded products, falls within the range of \$300 million to \$500 million.
- Which companies are the primary players in the current fluconazole market?
The current fluconazole market is dominated by generic manufacturers. Key players include Viatris (formed by Mylan and Upjohn), Teva Pharmaceuticals, Sanofi (generics division), and other established generic pharmaceutical companies.
Citations
[1] U.S. Patent No. 4,404,216. (1983). 2-(2,4-difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)-2-propanol. United States Patent and Trademark Office.
[2] U.S. Patent No. 6,756,379. (2004). Anhydrous crystalline fluconazole. United States Patent and Trademark Office.