Last updated: February 19, 2026
Lamotrigine, an anticonvulsant medication primarily used for treating epilepsy and bipolar disorder, is characterized by a mature market with established generic competition. Its financial trajectory is influenced by patent expirations, evolving treatment guidelines, and its positioning against newer therapeutic agents.
What is the Current Market Size and Growth Projection for Lamotrigine?
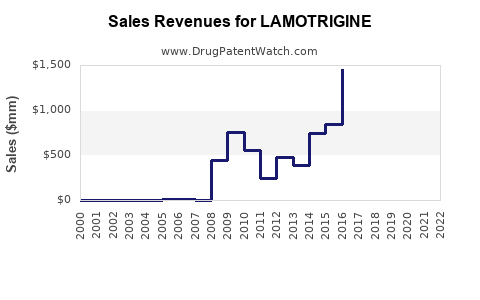
The global market for lamotrigine generated approximately $1.5 billion in 2023. The market is projected to experience a compound annual growth rate (CAGR) of 2.5% from 2024 to 2030, reaching an estimated $1.7 billion by the end of the forecast period. This growth is primarily driven by the sustained demand for epilepsy treatment and the ongoing use of lamotrigine in managing bipolar disorder, particularly in developing economies where access to newer, more expensive treatments is limited. However, the CAGR is moderate due to the widespread availability of generic versions, which limits significant price appreciation.
Who are the Key Players in the Lamotrigine Market?
The lamotrigine market is highly fragmented, dominated by generic manufacturers following the patent expiry of the originator drug, Lamictal (developed by GlaxoSmithKline). Key players include:
- Teva Pharmaceutical Industries Ltd.: A significant global producer of generic pharmaceuticals, including lamotrigine.
- Mylan N.V. (now part of Viatris Inc.): Another major generic supplier with a substantial market share.
- Sun Pharmaceutical Industries Ltd.: A leading Indian pharmaceutical company with a broad portfolio of generics.
- Dr. Reddy's Laboratories Ltd.: A prominent Indian multinational pharmaceutical company involved in lamotrigine manufacturing.
- Lupin Ltd.: An Indian multinational pharmaceutical company that manufactures and markets generic lamotrigine.
- Novartis AG (Sandoz division): Sandoz is a global leader in generic and biosimilar medicines.
- Sanofi S.A.: While less dominant in generics, Sanofi maintains a presence in certain markets.
The competitive landscape is characterized by price-based competition among these manufacturers.
What are the Primary Applications and Indications for Lamotrigine?
Lamotrigine is approved and widely used for several neurological and psychiatric conditions:
- Epilepsy:
- Adjunctive therapy for partial-onset seizures in adults and children aged 2 years and older.
- Adjunctive therapy for generalized tonic-clonic seizures in adults and children aged 2 years and older.
- Monotherapy for partial-onset seizures in adults and children aged 16 years and older.
- Treatment of Lennox-Gastaut syndrome (LGS) in children aged 2 years and older (adjunctive therapy).
- Bipolar Disorder:
- Maintenance treatment of bipolar I disorder in adults to delay the occurrence of mood episodes. It is not indicated for acute treatment of manic or depressive episodes.
The efficacy of lamotrigine in seizure control and mood stabilization underpins its continued clinical relevance.
What is the Intellectual Property Landscape for Lamotrigine?
The original compound patent for lamotrigine has long expired globally. GlaxoSmithKline’s (GSK) primary patents for Lamictal began expiring in the US around 2008 and in Europe in 2010. Subsequent secondary patents related to specific formulations, manufacturing processes, or dosage regimens have also largely expired or are nearing expiration. This lack of robust patent protection is the primary reason for the significant generic penetration and price erosion in the lamotrigine market. There are currently no major pending patent applications that are expected to create significant new market exclusivity for lamotrigine itself.
How Have Regulatory Policies Impacted the Lamotrigine Market?
Regulatory policies, particularly those concerning generic drug approvals and pricing, significantly influence the lamotrigine market.
- Generic Drug Approvals: Regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have streamlined the approval process for generic drugs. This facilitates market entry for multiple manufacturers, intensifying competition.
- Pricing Pressures: Government initiatives and payer policies in various countries aim to reduce healthcare costs by promoting the use of generic medications. This leads to downward pressure on lamotrigine prices.
- Pharmacovigilance and Safety Monitoring: Like all medications, lamotrigine is subject to ongoing pharmacovigilance. The U.S. FDA requires a Risk Evaluation and Mitigation Strategy (REMS) for certain lamotrigine formulations, particularly those associated with the risk of serious skin reactions, such as Stevens-Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TEN). While this does not directly restrict market entry, it imposes manufacturing and distribution requirements. In 2009, the FDA requested label updates for lamotrigine to include information about the risk of aseptic meningitis [1].
What are the Competitive Dynamics and Market Trends for Lamotrigine?
The competitive dynamics in the lamotrigine market are characterized by:
- Intense Generic Competition: The market is saturated with generic versions from numerous manufacturers. This leads to significant price wars and low profit margins for individual products.
- Therapeutic Alternatives: Lamotrigine faces competition from a range of other antiepileptic drugs (AEDs) and mood stabilizers. Newer generation AEDs offer different efficacy profiles, side-effect profiles, and mechanisms of action, some of which may be preferred in specific patient populations or for certain seizure types. For bipolar disorder, other mood stabilizers and atypical antipsychotics are also widely used.
- Market Penetration in Emerging Economies: While mature markets are saturated, opportunities exist for increased lamotrigine use in emerging economies due to its affordability compared to newer therapies.
- Formulation Development: While major patent expiries have occurred, some companies might pursue secondary patents for novel formulations (e.g., extended-release versions, orally disintegrating tablets) to gain a temporary competitive advantage or differentiate their product. However, these are less impactful than compound patents.
What are the Key Challenges and Opportunities in the Lamotrigine Market?
Challenges:
- Price Erosion: Persistent price declines due to generic competition limit revenue growth and profitability for manufacturers.
- Competition from Newer Drugs: The development of novel AEDs and psychiatric medications with improved efficacy or tolerability can lead to a shift in prescribing patterns away from lamotrigine.
- Skin Reaction Risk: The potential for severe skin reactions remains a significant concern, requiring careful patient titration and monitoring, which can sometimes limit its use or lead to discontinuation.
- Regulatory Scrutiny: Ongoing pharmacovigilance and potential for updated safety warnings can impact market perception and physician prescribing habits.
Opportunities:
- Affordability and Accessibility: Lamotrigine's low cost makes it a vital treatment option, especially in low- and middle-income countries, representing a significant market opportunity.
- Established Efficacy and Safety Profile: Decades of clinical use have established lamotrigine's efficacy and a well-understood safety profile (when managed appropriately), leading to physician confidence and patient familiarity.
- Combination Therapy: Its role as an adjunctive therapy in epilepsy means it continues to be prescribed alongside other AEDs, ensuring sustained demand.
- Market Expansion in Developing Nations: Growing healthcare infrastructure and increased access to medications in emerging markets present opportunities for market expansion.
What is the Financial Trajectory and Profitability Outlook for Lamotrigine?
The financial trajectory for lamotrigine is characterized by stable but modest revenue growth driven by volume increases, particularly in emerging markets, offset by significant price declines due to generic competition. Profitability for manufacturers is primarily dependent on cost-efficient production, supply chain management, and market access strategies.
- Revenue: The global revenue is projected to grow at a CAGR of 2.5%, from approximately $1.5 billion in 2023 to $1.7 billion by 2030. This growth rate is moderate and reflects the balance between increasing patient numbers and declining per-unit prices.
- Profit Margins: Gross profit margins for generic lamotrigine are typically in the range of 20-40%, depending on manufacturing efficiencies and market positioning. Net profit margins are lower after accounting for R&D (for process improvement), marketing, sales, and distribution costs.
- Investment Implications: For R&D investment, lamotrigine offers limited potential for groundbreaking innovation due to patent expirations. Investment focus would likely be on optimizing manufacturing processes for cost reduction or developing niche formulations. For investors, the market offers stable, albeit low-growth, revenue streams from established generic manufacturers. Acquisitions of smaller generic players with established lamotrigine production capabilities could be a strategy.
Key Takeaways
- Lamotrigine's market is mature and dominated by generic competition, resulting in moderate growth projections and price-based competition.
- Its established efficacy in epilepsy and bipolar disorder ensures continued demand, particularly in price-sensitive markets.
- The absence of significant patent protection for the active pharmaceutical ingredient limits innovation opportunities and perpetuates generic dominance.
- Key market players are major generic pharmaceutical manufacturers, competing primarily on cost and supply chain efficiency.
- Challenges include intense price erosion and competition from newer therapeutic agents, while opportunities lie in expanding access in emerging economies and leveraging its affordability.
FAQs
-
What is the primary driver of lamotrigine's market growth?
The primary driver is sustained demand for epilepsy and bipolar disorder treatments, especially in emerging economies where its affordability makes it a preferred option over newer, more expensive medications.
-
Are there any new patent applications that could impact the lamotrigine market?
The original compound patent for lamotrigine has expired globally. While companies might pursue secondary patents for specific formulations or delivery methods, these are unlikely to create significant new market exclusivity for the drug itself and are not expected to substantially alter the competitive landscape.
-
What is the main risk associated with lamotrigine that impacts its market use?
The main risk is the potential for serious skin reactions, including Stevens-Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TEN). This necessitates careful patient titration and monitoring, and has led to specific regulatory requirements for labeling and prescribing information.
-
How does lamotrigine compete with newer antiepileptic drugs (AEDs)?
Lamotrigine competes based on its long-standing efficacy, well-understood safety profile (when managed correctly), and significantly lower cost compared to many newer AEDs. While newer drugs may offer different benefits, lamotrigine remains a cost-effective option, especially for patients requiring adjunctive therapy or in regions with limited healthcare budgets.
-
What is the typical profit margin for a generic lamotrigine manufacturer?
Generic manufacturers typically see gross profit margins ranging from 20% to 40% for lamotrigine. Net profit margins are lower after operational costs, marketing, and distribution are factored in. Profitability relies heavily on efficient manufacturing and supply chain management.
Citations
[1] U.S. Food and Drug Administration. (2009, September 15). FDA Drug Safety Communication: Lamotrigine (Lamictal) and aseptic meningitis. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-lamotrigine-lamictal-and-aseptic-meningitis