Organon Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ORGANON, and when can generic versions of ORGANON drugs launch?
ORGANON has fifty-nine approved drugs.
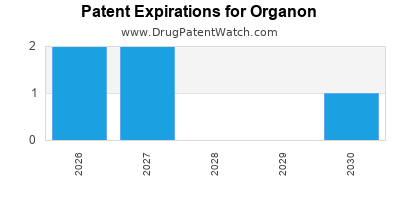
There are six US patents protecting ORGANON drugs. There is one tentative approval on ORGANON drugs.
There are three hundred and thirteen patent family members on ORGANON drugs in forty-four countries and one hundred and thirty-six supplementary protection certificates in fifteen countries.
Drugs and US Patents for Organon
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Organon | ZOCOR | simvastatin | TABLET;ORAL | 019766-003 | Dec 23, 1991 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Organon | ELOCON | mometasone furoate | CREAM;TOPICAL | 019625-002 | Apr 19, 2013 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Organon Llc | XACIATO | clindamycin phosphate | GEL;VAGINAL | 215650-001 | Dec 7, 2021 | RX | Yes | Yes | 11,129,896 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Organon
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Organon | DIPROLENE | betamethasone dipropionate | LOTION, AUGMENTED;TOPICAL | 019716-001 | Aug 1, 1988 | 4,775,529 | ⤷ Try a Trial |
| Organon | VYTORIN | ezetimibe; simvastatin | TABLET;ORAL | 021687-002 | Jul 23, 2004 | RE37721*PED | ⤷ Try a Trial |
| Organon | LOTRISONE | betamethasone dipropionate; clotrimazole | CREAM;TOPICAL | 018827-001 | Jul 10, 1984 | 3,705,172 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for ORGANON drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Oral Solution | 70 mg/75 mL | ➤ Subscribe | 2007-09-07 |
| ➤ Subscribe | Tablets | 100 mg/12.5 mL | ➤ Subscribe | 2006-04-04 |
| ➤ Subscribe | Tablets | 10 mg | ➤ Subscribe | 2006-10-25 |
| ➤ Subscribe | Tablets | 70 mg/2800 IU and 70 mg/5600 IU | ➤ Subscribe | 2007-11-20 |
| ➤ Subscribe | Injection | 250 mcg/0.5 mL, 1 mL PFS | ➤ Subscribe | 2012-03-30 |
| ➤ Subscribe | Tablets | 10 mg | ➤ Subscribe | 2007-02-20 |
| ➤ Subscribe | Tablets | 5 mg | ➤ Subscribe | 2006-06-21 |
| ➤ Subscribe | Orally Disintegrating Tablets | 5 mg and 10 mg | ➤ Subscribe | 2006-02-17 |
| ➤ Subscribe | Tablets | 50 mg/12.5 mg and 100 mg/25 mg | ➤ Subscribe | 2004-05-24 |
| ➤ Subscribe | Tablets | 10 mg/10 mg, 10 mg/20 mg, 10 mg/40 mg and 10 mg/80 mg | ➤ Subscribe | 2009-07-27 |
| ➤ Subscribe | Nasal Spray | 50 mcg/ Spray | ➤ Subscribe | 2009-08-07 |
| ➤ Subscribe | Tablets | 5 mg and 10 mg | ➤ Subscribe | 2004-09-02 |
| ➤ Subscribe | Vaginal Ring | 0.015 mg/24 hour and 0.12 mg/24 hour | ➤ Subscribe | 2013-06-17 |
| ➤ Subscribe | Topical Solution (Lotion) | 0.10% | ➤ Subscribe | 2004-06-10 |
| ➤ Subscribe | Chewable Tablets | 4 mg and 5 mg | ➤ Subscribe | 2006-12-26 |
| ➤ Subscribe | Orally Disintegrating Tablets | 2.5 mg and 5 mg | ➤ Subscribe | 2006-06-21 |
International Patents for Organon Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 2009286801 | ⤷ Try a Trial |
| Australia | 2002247019 | ⤷ Try a Trial |
| Spain | 2308460 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Organon Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0454511 | 99C0009 | Belgium | ⤷ Try a Trial | PRODUCT NAME: IRBESARTAN / HYDROCHLOROTHIAZIDE; REGISTRATION NO/DATE: EU/1/98/086/001 19981015 |
| 0613371 | 2002C/022 | Belgium | ⤷ Try a Trial | PRODUCT NAME: BUDESONID. MICRONIS. AND FORMOTEROL. FUMARAS DIHYDR., NATL REGISTRATION NO/DATE: 624 IS 234 F 0 20010129; FIRST REGISTRATION: SE 16047 20000825 |
| 0480717 | 9890027-7 | Sweden | ⤷ Try a Trial | PRODUCT NAME: MONTELUKAST, R-(E)-1-1-3-2-(7-KLOR-2KINOLINYL)-ETENYL)-FENYL)-3-(2-(1-HYDROXI-1-METYLETYL)-FENYL)-PROPYL)-TIO)METYL)-CYKLOPROPANAETTIKSYRA, ELLER ETT FARMACEUTISKT GODDTAGBART SALT DAERAV, FOERETRAEDESVIS NATRIUMSALTET; NAT REG./DATE: 13944 19880111; FIRST REG.: FI EG 12766 19970825 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.