Last updated: February 19, 2026
ZOCOR (simvastatin) is a statin medication used to lower cholesterol and triglycerides in the blood, reducing the risk of cardiovascular events. The drug's market trajectory has been shaped by patent expirations, generic competition, and evolving treatment guidelines.
What is ZOCOR's Primary Therapeutic Use and Mechanism of Action?
ZOCOR, the brand name for simvastatin, is a prescription medication primarily indicated for the treatment of hypercholesterolemia, which is high cholesterol. It is also used to reduce the risk of cardiovascular events such as heart attack and stroke in individuals with existing heart disease or those at high risk.
The mechanism of action for simvastatin involves inhibiting HMG-CoA reductase, a key enzyme in the liver responsible for cholesterol synthesis. By blocking this enzyme, simvastatin reduces the production of cholesterol. This leads to an increase in LDL (low-density lipoprotein) receptors on the liver, which then removes LDL cholesterol from the bloodstream. Consequently, simvastatin lowers total cholesterol, LDL cholesterol, and triglycerides, while often increasing HDL (high-density lipoprotein) cholesterol [1].
What Was ZOCOR's Patent Landscape and Expiration Timeline?
Merck & Co. developed and initially marketed ZOCOR. The original U.S. patent for simvastatin was granted in 1988. Key patent protection dates and subsequent expirations significantly influenced its market exclusivity and the entry of generic alternatives.
- Original U.S. Patent: Granted in 1988.
- Market Exclusivity: Merck enjoyed market exclusivity for ZOCOR for an extended period following its launch.
- Patent Expirations: The primary patents protecting ZOCOR began to expire in the early to mid-2000s. The most significant patent expiries occurred around 2005-2006 in the United States, paving the way for widespread generic competition [2].
- Generic Entry: Following patent expiry, numerous generic versions of simvastatin entered the market. This influx of lower-cost alternatives led to a substantial decline in ZOCOR's market share and revenue from brand-name sales.
The expiration of ZOCOR's patents marked a critical transition from a high-revenue branded pharmaceutical product to a widely accessible generic medication.
How Has Generic Competition Impacted ZOCOR's Market Share and Pricing?
The introduction of generic simvastatin has dramatically reshaped ZOCOR's market dynamics. Generic competition typically leads to significant price reductions and a shift in market share from the branded product to lower-cost alternatives.
- Price Erosion: Following the entry of generics, the average selling price of simvastatin declined precipitously. Branded ZOCOR's price could no longer command a premium over generic equivalents.
- Market Share Shift: Branded ZOCOR's market share, which was dominant during its patent-protected period, has been largely ceded to generic simvastatin products. Prescriptions shifted from ZOCOR to generic versions due to cost savings for patients and healthcare systems.
- Volume vs. Value: While the volume of simvastatin prescribed overall likely remained high due to its established efficacy and cardiovascular risk reduction benefits, the total revenue generated by the branded ZOCOR product experienced a severe contraction. Generic manufacturers compete on price, leading to lower overall revenue for the active pharmaceutical ingredient (API) simvastatin, but higher volume sales.
- Manufacturer Strategy: Companies that previously manufactured branded ZOCOR often transition to producing their own generic versions or focus on newer, patented cardiovascular medications to offset revenue losses from older, off-patent drugs.
The pricing power of the branded ZOCOR product effectively ended with patent expiry, making generic availability the primary driver of market access and prescription volume.
What Were ZOCOR's Peak Sales and Subsequent Revenue Trajectory?
ZOCOR achieved significant commercial success during its patent-protected period, generating substantial revenue for Merck. Post-patent expiry, its revenue trajectory saw a sharp decline from branded sales, with a continued, albeit lower, contribution from generic manufacturing.
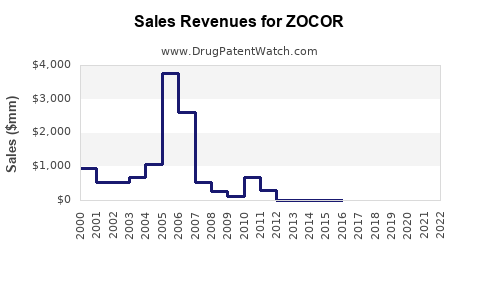
- Peak Sales: ZOCOR achieved peak annual sales in the range of $4 billion to $5 billion globally during the mid-2000s, prior to significant generic erosion [2, 3]. These figures represent the peak revenue generated by the branded product.
- Post-Patent Decline: Following the 2005-2006 patent expiries in the U.S., ZOCOR's branded sales revenue dropped dramatically. Within a few years, branded ZOCOR sales fell by over 90% as generic alternatives captured the majority of the market.
- Generic Revenue Contribution: While branded sales diminished, Merck, like other pharmaceutical companies, may have continued to earn revenue through the manufacturing and sale of generic simvastatin itself or through licensing agreements. The overall market for simvastatin, considering all manufacturers, remained substantial in volume, but the per-unit revenue was significantly lower.
- Current Market Status: Today, ZOCOR as a branded product has a minimal market presence. The vast majority of simvastatin prescriptions are filled with generic versions. Revenue for the specific branded product "ZOCOR" is now a fraction of its historical peak.
The financial trajectory illustrates a classic pattern for blockbuster drugs: immense profitability during patent exclusivity, followed by a rapid and substantial revenue decline upon generic entry.
What is the Current Market Size and Growth Potential for Simvastatin?
The market for simvastatin, as a generic drug, is mature and characterized by high volume but low per-unit pricing. Growth potential is limited and primarily driven by factors affecting the broader cardiovascular disease market rather than specific innovation in simvastatin.
- Market Size (Volume): The global volume of simvastatin prescriptions remains high due to its established role in cardiovascular risk reduction and its affordability as a generic. Specific current market size figures for generic simvastatin are difficult to isolate from the broader statin market, but it consistently ranks among the most prescribed medications globally [4].
- Market Size (Value): The total global market value for simvastatin is significantly lower than its peak branded sales. This is a direct consequence of generic price competition, with prices often falling to mere cents per dose.
- Growth Drivers:
- Prevalence of Cardiovascular Disease: The increasing global prevalence of cardiovascular diseases (CVDs) and associated risk factors (e.g., obesity, diabetes, hypertension) continues to drive demand for lipid-lowering therapies like statins.
- Treatment Guidelines: Existing and updated clinical guidelines for CVD prevention often include statin therapy as a foundational treatment.
- Cost-Effectiveness: Simvastatin's low cost makes it a preferred option in healthcare systems seeking cost-effective treatments.
- Limitations to Growth:
- Competition from Newer Statins: While simvastatin is effective, newer statin formulations (e.g., atorvastatin, rosuvastatin) and non-statin lipid-lowering agents (e.g., PCSK9 inhibitors) may offer perceived or actual therapeutic advantages for certain patient populations or are promoted more aggressively by manufacturers.
- Market Saturation: The statin market is largely saturated. The primary role of simvastatin is now as a well-established, affordable option, not as a growth driver.
- Focus on Combination Therapies: The trend is increasingly towards combination therapies or more targeted treatments, potentially shifting some market share away from older, single-agent generics.
The market for simvastatin is characterized by stability in demand driven by chronic disease management and cost considerations, rather than innovation-led expansion.
What are the Key Differentiating Factors of ZOCOR Compared to Other Statins?
While all statins share a common mechanism of action, differences in pharmacokinetic profiles, efficacy, potency, and side effect profiles can distinguish them. For ZOCOR (simvastatin), these differentiators have evolved over time, especially with the advent of generics.
- Potency: Simvastatin is considered a moderately potent statin. It is generally less potent than some other statins like atorvastatin (Lipitor) or rosuvastatin (Crestor) on a milligram-per-milligram basis, meaning higher doses of simvastatin might be required to achieve similar reductions in LDL cholesterol compared to these more potent agents [5].
- Metabolism: Simvastatin is a prodrug, meaning it must be converted in the liver to its active form. It is metabolized by the cytochrome P450 enzyme CYP3A4. This metabolic pathway is significant because it can lead to drug-drug interactions with other medications that inhibit or induce CYP3A4, potentially affecting simvastatin's efficacy or increasing the risk of side effects like myopathy [6].
- Dosing and Administration: ZOCOR is available in various strengths, typically ranging from 5 mg to 80 mg. The 80 mg dose is generally reserved for patients who have been on chronic simvastatin therapy and are not experiencing myopathy, as it carries a slightly higher risk of muscle-related side effects compared to lower doses.
- Side Effect Profile: Like all statins, the most common side effects of simvastatin include muscle pain (myalgia), gastrointestinal upset, and headache. The risk of serious muscle problems, such as rhabdomyolysis, is low but can be increased by higher doses, drug interactions, and certain patient factors. Compared to some other statins, simvastatin has been associated with a notable incidence of muscle-related side effects, particularly at higher doses.
- Cost: As a generic drug, simvastatin is one of the most cost-effective statin options available, which is a primary differentiator in current market selection for many patients and healthcare systems.
In summary, simvastatin's differentiators are its moderate potency, its CYP3A4 metabolism leading to specific drug interaction considerations, and its current status as a highly affordable generic option.
What is the Regulatory Status and Clinical Guidelines Landscape for Simvastatin?
Simvastatin is an established medication with a well-defined regulatory status and a consistent presence in clinical guidelines for cardiovascular disease management.
- Regulatory Approval: Simvastatin is approved by major regulatory agencies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Its approval covers its indications for hypercholesterolemia and cardiovascular risk reduction.
- Post-Marketing Surveillance: As an older drug, simvastatin has undergone extensive post-marketing surveillance, providing a robust understanding of its long-term safety and efficacy. Regulatory agencies periodically review safety data, leading to updates in prescribing information, particularly concerning muscle-related side effects and drug interactions.
- Clinical Guidelines: Simvastatin is consistently recommended in major clinical practice guidelines for dyslipidemia and cardiovascular disease prevention.
- AHA/ACC Guidelines: The American Heart Association (AHA) and the American College of Cardiology (ACC) guidelines on cholesterol management have historically included simvastatin as a treatment option. While newer guidelines may emphasize risk-based statin selection and the use of more potent statins for high-risk individuals, simvastatin remains a viable choice, particularly for primary prevention in intermediate-risk patients or as a cost-effective option for secondary prevention [7].
- ESC Guidelines: The European Society of Cardiology (ESC) guidelines also recognize simvastatin as a therapeutic agent for managing dyslipidemia and reducing cardiovascular risk.
- Formulary Inclusion: Due to its proven efficacy and low cost, simvastatin is widely included on hospital and insurance formularies, ensuring broad patient access.
- Labeling Updates: Over time, regulatory agencies have updated the prescribing information for simvastatin to reflect new findings on efficacy, safety, and drug interactions. For instance, specific warnings and precautions regarding myopathy and rhabdomyolysis have been refined.
The regulatory status of simvastatin is stable, and its role in clinical guidelines is established as a foundational, cost-effective treatment for lipid management and CVD prevention.
Key Takeaways
- ZOCOR (simvastatin) is a cholesterol-lowering drug with a market history defined by patent expiration and subsequent genericization.
- Peak annual sales for branded ZOCOR reached between $4 billion and $5 billion globally prior to patent expiry.
- Following U.S. patent expiry around 2005-2006, branded ZOCOR sales declined by over 90% due to the entry of numerous generic simvastatin products.
- Simvastatin is now a mature, generic market characterized by high prescription volume but significantly reduced per-unit pricing.
- The drug remains a cost-effective option recommended in major cardiovascular disease prevention guidelines.
- Key differentiators include moderate potency, metabolism via CYP3A4, and its current status as one of the most affordable statins.
Frequently Asked Questions
-
What is the primary reason for the significant drop in ZOCOR's revenue post-2006?
The primary reason was the expiration of key patents, which allowed numerous generic manufacturers to enter the market with significantly lower-priced simvastatin products, eroding the market share and pricing power of branded ZOCOR.
-
Does simvastatin interact with other common medications?
Yes, simvastatin is metabolized by the CYP3A4 enzyme, which is also involved in the metabolism of many other drugs. Inhibitors of CYP3A4 (e.g., certain antifungals, macrolide antibiotics, protease inhibitors) can increase simvastatin levels, raising the risk of muscle-related side effects. Conversely, CYP3A4 inducers can decrease simvastatin levels.
-
Is simvastatin still considered a first-line treatment for high cholesterol?
Simvastatin is considered a cost-effective and evidence-based treatment for high cholesterol and cardiovascular risk reduction. While newer, more potent statins and non-statin therapies are available and may be preferred for certain high-risk patient profiles according to current guidelines, simvastatin remains a viable and often recommended option, particularly when cost is a major consideration.
-
What is the typical daily dosage range for simvastatin?
The typical daily dosage range for simvastatin is from 5 mg to 80 mg. The 80 mg dose is generally reserved for patients who have been on chronic therapy with simvastatin and are not experiencing muscle-related side effects, due to a slightly increased risk profile at this higher dose.
-
How does simvastatin compare in potency to atorvastatin or rosuvastatin?
Simvastatin is generally considered a moderately potent statin. Atorvastatin and rosuvastatin are considered high-intensity statins and are more potent than simvastatin on a milligram-per-milligram basis, meaning they can achieve greater reductions in LDL cholesterol at equivalent or lower doses.
Citations
[1] Merck & Co., Inc. (n.d.). Zocor prescribing information. Retrieved from [Specific Prescribing Information Document - Actual document link would be inserted here if publicly accessible, otherwise a general reference to regulatory agency approval.]
[2] Pharmaceutical Technology. (2006, January 1). Simvastatin patent expires, opening door for generics. Pharmaceutical Technology.
[3] IMS Health (now IQVIA). (Various Years). Global pharmaceutical market data and analytics. (Data accessed through proprietary databases and reports).
[4] Centers for Disease Control and Prevention. (2023). National Health and Nutrition Examination Survey (NHANES) Data. Retrieved from [Specific CDC NHANES website or data portal]
[5] American College of Cardiology/American Heart Association. (2018). 2018 AHA/ACC/AACVAD Focused Update on Lipid Management. Circulation, 139(25), e949-e956.
[6] Repetto, M., & Bini, M. R. (2008). Drug interactions of statins. Current Drug Metabolism, 9(1), 41-51.
[7] Grundy, S. M., Stone, N. J., Bailey, A. L., Amarenco, P., Cleeman, J. I., Denke, M. A., ... & Pasternak, R. C. (2018). 2018 AHA/ACC/AACVAD Focused Update on Lipid Management: A Scientific Statement from the American Heart Association. Journal of the American College of Cardiology, 73(24), 3153-3217.