MERCK SHARP DOHME Company Profile
✉ Email this page to a colleague
What is the competitive landscape for MERCK SHARP DOHME, and what generic alternatives to MERCK SHARP DOHME drugs are available?
MERCK SHARP DOHME has twenty-three approved drugs.
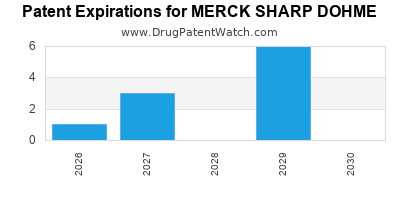
There are twenty-five US patents protecting MERCK SHARP DOHME drugs.
There are six hundred and ninety-four patent family members on MERCK SHARP DOHME drugs in sixty-three countries and one hundred and twenty-seven supplementary protection certificates in nineteen countries.
Summary for MERCK SHARP DOHME
| International Patents: | 694 |
| US Patents: | 25 |
| Tradenames: | 19 |
| Ingredients: | 19 |
| NDAs: | 23 |
Drugs and US Patents for MERCK SHARP DOHME
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Merck Sharp Dohme | DUTREBIS | lamivudine; raltegravir potassium | TABLET;ORAL | 206510-001 | Feb 6, 2015 | DISCN | No | No | 7,754,731*PED | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Merck Sharp Dohme | VERQUVO | vericiguat | TABLET;ORAL | 214377-002 | Jan 19, 2021 | RX | Yes | No | 8,921,377 | ⤷ Sign Up | ⤷ Sign Up | ||||
| Merck Sharp Dohme | TEMODAR | temozolomide | CAPSULE;ORAL | 021029-005 | Oct 19, 2006 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for MERCK SHARP DOHME
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Merck Sharp Dohme | REBETOL | ribavirin | CAPSULE;ORAL | 020903-002 | Jul 25, 2001 | 5,914,128*PED | ⤷ Sign Up |
| Merck Sharp Dohme | TEMODAR | temozolomide | POWDER;INTRAVENOUS | 022277-001 | Feb 27, 2009 | 6,987,108 | ⤷ Sign Up |
| Merck Sharp Dohme | JUVISYNC | simvastatin; sitagliptin phosphate | TABLET;ORAL | 202343-005 | Sep 18, 2012 | 6,303,661 | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for MERCK SHARP DOHME drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Oral Suspension | 40 mg/mL | ➤ Subscribe | 2011-02-28 |
| ➤ Subscribe | Injection | 18 mg/mL, 16.7 mL vials | ➤ Subscribe | 2015-11-24 |
| ➤ Subscribe | Orally Disintegrating Tablets | 2.5 mg and 5 mg | ➤ Subscribe | 2006-06-21 |
| ➤ Subscribe | Tablets | 100 mg/10 mg and 100 mg/40 mg | ➤ Subscribe | 2012-06-19 |
| ➤ Subscribe | Tablets | 50 mg/10 mg, 50 mg/20 mg, and 50 mg/40 mg | ➤ Subscribe | 2012-11-06 |
| ➤ Subscribe | Capsules | 140 mg and 180 mg | ➤ Subscribe | 2008-03-24 |
| ➤ Subscribe | Delayed-release Tablets | 100 mg | ➤ Subscribe | 2014-06-16 |
| ➤ Subscribe | Tablets | 5 mg | ➤ Subscribe | 2006-06-21 |
| ➤ Subscribe | Tablets | 25 mg, 50 mg and 100 mg | ➤ Subscribe | 2010-10-18 |
| ➤ Subscribe | Tablets | 100 mg/10 mg and 100 mg/40 mg | ➤ Subscribe | 2012-06-25 |
International Patents for MERCK SHARP DOHME Drugs
Supplementary Protection Certificates for MERCK SHARP DOHME Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1412357 | 50/2008 | Austria | ⤷ Sign Up | PRODUCT NAME: SITAGLIPTIN, GEGEBENENFALLS IN DER FORM EINES PHARMAZEUTISCH ANNEHMBAREN SALZES, INSBESONDERE ALS MONOPHOSPHAT, UND METFORMIN, GEGEBENENFALLS IN FORM EINES PHARMAZEUTISCH ANNEHMBAREN SALZES, INSBESONDERE DES HYDROCHLORIDS; NAT. REGISTRATION NO/DATE: EU/1/08/455/001-014, EU/1/08/456/001-014, EU/1/08/457/001-014 20080716; FIRST REGISTRATION: CH 58450 01-03 20080408 |
| 1622880 | 28/2018 | Austria | ⤷ Sign Up | PRODUCT NAME: LETERMOVIR ODER DESSEN SALZ, SOLVAT ODER SOLVAT DES SALZES; NAT. REGISTRATION NO/DATE: EU/1/17/1245/001 - EU/1/17/1245/004 (MITTEILUNG) 20180110; FIRST REGISTRATION: EU EU/1/17/1245/001 - EU/1/17/1245/004 20180110 |
| 1412357 | SPC/GB08/040 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: SITAGLIPTIN OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, IN PARTICULAR THE MONOPHOSPHATE, PLUS METFORMIN OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, IN PARTICULAR THE HYDROCHLORIDE.; REGISTERED: CH 58450 01-03 20080408; UK EU/1/08/455/001 20080716; UK EU/1/08/455/002 20080716; UK EU/1/08/455/003 20080716; UK EU/1/08/455/004 20080716; UK EU/1/08/455/005 20080716; UK EU/1/08/455/006 20080716; UK EU/1/08/455/007 20080716; UK EU/1/08/455/008 20080716; UK EU/1/08/455/009 20080716; UK EU/1/08/455/010 20080716; UK EU/1/08/455/011 20080716; UK EU/1/08/455/012 20080716; UK EU/1/08/455/013 20080716; UK EU/1/08/455/014 20080716 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.