Hoffmann La Roche Company Profile
✉ Email this page to a colleague
What is the competitive landscape for HOFFMANN LA ROCHE, and when can generic versions of HOFFMANN LA ROCHE drugs launch?
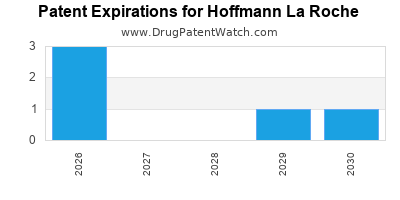
HOFFMANN LA ROCHE has ten approved drugs.
There are six US patents protecting HOFFMANN LA ROCHE drugs.
There are one hundred and eighty patent family members on HOFFMANN LA ROCHE drugs in forty-six countries and twenty-three supplementary protection certificates in seventeen countries.
Summary for Hoffmann La Roche
| International Patents: | 180 |
| US Patents: | 6 |
| Tradenames: | 9 |
| Ingredients: | 8 |
| NDAs: | 10 |
Drugs and US Patents for Hoffmann La Roche
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hoffmann La Roche | ZELBORAF | vemurafenib | TABLET;ORAL | 202429-001 | Aug 17, 2011 | RX | Yes | Yes | 8,143,271 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Hoffmann La Roche | ACCUTANE | isotretinoin | CAPSULE;ORAL | 018662-002 | May 7, 1982 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Hoffmann La Roche | ROCEPHIN | ceftriaxone sodium | INJECTABLE;INJECTION | 063239-002 | Aug 13, 1993 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Hoffmann La Roche | ROCEPHIN | ceftriaxone sodium | INJECTABLE;INTRAMUSCULAR, INTRAVENOUS | 050585-003 | Dec 21, 1984 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Hoffmann La Roche
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Hoffmann La Roche | BONIVA | ibandronate sodium | TABLET;ORAL | 021455-002 | Mar 24, 2005 | 7,718,634 | ⤷ Try a Trial |
| Hoffmann La Roche | ACCUTANE | isotretinoin | CAPSULE;ORAL | 018662-003 | May 7, 1982 | 4,322,438 | ⤷ Try a Trial |
| Hoffmann La Roche | ACCUTANE | isotretinoin | CAPSULE;ORAL | 018662-004 | Mar 28, 1983 | 4,464,394*PED | ⤷ Try a Trial |
| Hoffmann La Roche | ACCUTANE | isotretinoin | CAPSULE;ORAL | 018662-002 | May 7, 1982 | 4,464,394*PED | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for HOFFMANN LA ROCHE drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 2.5 mg and 150 mg | ➤ Subscribe | 2007-05-16 |
| ➤ Subscribe | Injection | 1 mg/mL, 3 mL Vial | ➤ Subscribe | 2007-08-31 |
International Patents for Hoffmann La Roche Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Mexico | 349923 | ⤷ Try a Trial |
| Japan | 2008545652 | ⤷ Try a Trial |
| China | 110269838 | ⤷ Try a Trial |
| Ecuador | SP077985 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Hoffmann La Roche Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1893612 | C300534 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: VEMURAFENIB ALSMEDE FARMACEUTISCH AANVAARDBARE ZOUTEN DAARVAN; REGISTRATION NO/DATE: EU/1/12/751/001 20120217 |
| 1893612 | PA2012010,C1893612 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: VEMURAFENIBUM; REGISTRATION NO/DATE: EU/1/12/751/001 20120217 |
| 1893612 | 2012C/028 | Belgium | ⤷ Try a Trial | PRODUCT NAME: VERMURAFENIB ET SES SELS PHARMACEUTIQUEMENT ACCEPTABLES, SOUS TOUTES FORMES COUVERTES PAR LE BREVET DE BASE; AUTHORISATION NUMBER AND DATE: EU/1/12/751/001 20120221 |
| 1893612 | 1290026-2 | Sweden | ⤷ Try a Trial | PRODUCT NAME: VEMURAFENIB OCH FARMACEUTISKT GODTAGBARA SALTER DAERAV; REG. NO/DATE: EU/1/12/751/001 20120217 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.