ZELBORAF Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Zelboraf, and when can generic versions of Zelboraf launch?
Zelboraf is a drug marketed by Hoffmann La Roche and is included in one NDA. There are six patents protecting this drug.
This drug has one hundred and ninety-five patent family members in forty-six countries.
The generic ingredient in ZELBORAF is vemurafenib. One supplier is listed for this compound. Additional details are available on the vemurafenib profile page.
DrugPatentWatch® Generic Entry Outlook for Zelboraf
Zelboraf was eligible for patent challenges on August 17, 2015.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be June 6, 2032. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ZELBORAF?
- What are the global sales for ZELBORAF?
- What is Average Wholesale Price for ZELBORAF?
Summary for ZELBORAF
| International Patents: | 195 |
| US Patents: | 6 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 96 |
| Clinical Trials: | 53 |
| Patent Applications: | 5,540 |
| Drug Prices: | Drug price information for ZELBORAF |
| What excipients (inactive ingredients) are in ZELBORAF? | ZELBORAF excipients list |
| DailyMed Link: | ZELBORAF at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for ZELBORAF
Generic Entry Date for ZELBORAF*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for ZELBORAF
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Cancer Research UK | Phase 2/Phase 3 |
| Royal Marsden NHS Foundation Trust | Phase 2/Phase 3 |
| University of Manchester | Phase 2/Phase 3 |
Pharmacology for ZELBORAF
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Cytochrome P450 1A2 Inhibitors P-Glycoprotein Inhibitors Protein Kinase Inhibitors |
US Patents and Regulatory Information for ZELBORAF
ZELBORAF is protected by seven US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of ZELBORAF is ⤷ Start Trial.
This potential generic entry date is based on patent 9,447,089.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hoffmann La Roche | ZELBORAF | vemurafenib | TABLET;ORAL | 202429-001 | Aug 17, 2011 | RX | Yes | Yes | 8,470,818 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Hoffmann La Roche | ZELBORAF | vemurafenib | TABLET;ORAL | 202429-001 | Aug 17, 2011 | RX | Yes | Yes | 7,504,509 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Hoffmann La Roche | ZELBORAF | vemurafenib | TABLET;ORAL | 202429-001 | Aug 17, 2011 | RX | Yes | Yes | 9,447,089 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Hoffmann La Roche | ZELBORAF | vemurafenib | TABLET;ORAL | 202429-001 | Aug 17, 2011 | RX | Yes | Yes | 8,143,271 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Hoffmann La Roche | ZELBORAF | vemurafenib | TABLET;ORAL | 202429-001 | Aug 17, 2011 | RX | Yes | Yes | 8,741,920 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for ZELBORAF
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Roche Registration GmbH | Zelboraf | vemurafenib | EMEA/H/C/002409Vemurafenib is indicated in monotherapy for the treatment of adult patients with BRAF-V600-mutation-positive unresectable or metastatic melanoma., | Authorised | no | no | no | 2012-02-17 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
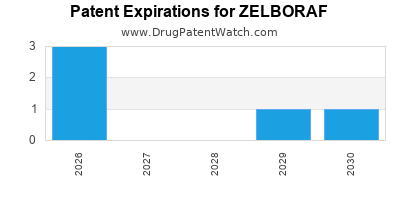
International Patents for ZELBORAF
When does loss-of-exclusivity occur for ZELBORAF?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 8033
Estimated Expiration: ⤷ Start Trial
Patent: 1037
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 10232670
Estimated Expiration: ⤷ Start Trial
Patent: 10318049
Estimated Expiration: ⤷ Start Trial
Patent: 15238857
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2012009609
Estimated Expiration: ⤷ Start Trial
Patent: 2020005420
Estimated Expiration: ⤷ Start Trial
Patent: 1008709
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 38573
Estimated Expiration: ⤷ Start Trial
Patent: 78693
Estimated Expiration: ⤷ Start Trial
China
Patent: 2361870
Estimated Expiration: ⤷ Start Trial
Patent: 2596953
Estimated Expiration: ⤷ Start Trial
Patent: 5237530
Estimated Expiration: ⤷ Start Trial
Patent: 0269838
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 10296
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 110420
Estimated Expiration: ⤷ Start Trial
Patent: 170089
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0151156
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 16983
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 14356
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 011000291
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 11011282
Estimated Expiration: ⤷ Start Trial
El Salvador
Patent: 11004004
Patent: COMPOSICIONES DEL ACIDO PROPANO-1-SULFONICO {3-[5-(4-CLORO-FENIL)-1H-PIRROLO [2,3-B]-PIRIDINA-3-CARBONIL]-2,4-DIFLUORO-FENIL}-AMIDA Y EL USO DE LAS MISMAS
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 2924
Estimated Expiration: ⤷ Start Trial
Patent: 1116
Estimated Expiration: ⤷ Start Trial
Patent: 1190098
Estimated Expiration: ⤷ Start Trial
Patent: 1591240
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 14356
Estimated Expiration: ⤷ Start Trial
Patent: 99138
Estimated Expiration: ⤷ Start Trial
Patent: 55180
Estimated Expiration: ⤷ Start Trial
Honduras
Patent: 11002147
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 68590
Estimated Expiration: ⤷ Start Trial
Patent: 17195
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 27598
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 4328
Estimated Expiration: ⤷ Start Trial
Patent: 1336
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 11942
Estimated Expiration: ⤷ Start Trial
Patent: 12522791
Estimated Expiration: ⤷ Start Trial
Patent: 13510813
Estimated Expiration: ⤷ Start Trial
Jordan
Patent: 56
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 0737
Patent: PROPANE-I-SULFONIC ACID {3- [5-(4-CHLORO-PHENYL) -1H -PYRROLO [2, 3-B] PYRIDINE-3-CARBONYL] -2,4 DIFLUORO-PHENYL} - AMIDE COMPOSITIONS AND USES THEREOF
Estimated Expiration: ⤷ Start Trial
Patent: 2424
Patent: PROPANE- I-SULFONIC ACID {3- (4-CHLORO-PHENYL)-1H-PYRROLO [2, 3-B] PYRIDINE-3-CARCONYL] -2, 4-DIFLUORO-PHENYL} -AMIDE COMPOSITIONS AND USES THEREOF
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 9923
Patent: COMPOSICIONES DEL ÁCIDO PROPANO-1-SULFÓNICO {3-[5-(4-CLORO-FENIL)- 1H-PIRROLO [2,3-B]-PIRIDINA-3-CARBONIL]-2,4-DIFLUORO-FENIL]-AMIDA Y EL USO DE LAS MISMAS. (PROPANE- I-SULFONIC ACID {3- [5- (4 -CHLORO-PHENYL) -1H-PYRROLO [2, 3-B] PYRIDINE-3-CARBONYL] -2, 4-DIFLUORO-PHENY L } -AMIDE COMPOSITIONS AND USES THEREOF.)
Estimated Expiration: ⤷ Start Trial
Patent: 11008303
Estimated Expiration: ⤷ Start Trial
Patent: 12005224
Patent: NUEVO PROCESO PARA LA MANUFACTURA DE PREPARACIONES FARMACEUTICAS. (PROPANE-I-SULFONIC ACID {3-[5-(4-CHLORO-PHENYL)-1H-PYRROLO[2,3-B] PYRIDINE-3-CARBONYL]-2,4-DIFLUORO-PHENYL}-AMIDE COMPOSITIONS AND USES THEREOF.)
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 028
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 4398
Patent: Propane-1-sulfonic acid (3-[5-(4-chloro-phenyl)-1h-pyrrol [2, 3-b] pyridine-3-carbonyl]-2,4-difluoro-phenyl} -amide compositions and uses thereof
Estimated Expiration: ⤷ Start Trial
Nicaragua
Patent: 1100161
Patent: COMPOSICIONES DEL ÁCIDO PROPANO - 1 - SULFÓNICO { 3 - [5 - (4 - CLORO - FENIL) - 1H - PIRROLO [2, 3-b] - PIRIDINA - 3 - CARBONIL] - 2, 4 - DIFLUORO - FENIL} - AMIDA Y EL USO DE LAS MISMAS
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 120876
Patent: COMPOSICIONES DEL ACIDO PROPANO-1-SULFONICO{3-[5-(4-CLORO-FENIL)-1H-PIRROLO[2,3-B]-PIRIDINA-3-CARBONIL]-2,4-DIFLUORO-FENIL}-AMIDA
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 14356
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 14356
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 12123958
Patent: КОМПОЗИЦИИ {3-[5-(4-ХЛОРФЕНИЛ)-1Н-ПИРРОЛО[2, 3]ПИРИДИН-3-КАРБОНИЛ]-2,4-ДИФТОРФЕНИЛ}АМИДА ПРОПАН-1-СУЛЬФОНОВОЙ КИСЛОТЫ И ИХ ПРИМЕНЕНИЕ
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01500302
Patent: COMPOSIZIONI DI {3-[5-(4-CLORO-FENIL)-1H-PIRROLO[2,3-B]PIRIDIN-3-CARBONIL]-2,4-DIFLUORO-FENIL}-AMMIDE DELL'ACIDO PROPAN-1-SOLFONICO E LORO USI
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 3178
Patent: PROPANE- I-SULFONIC ACID {3- [5- (4 -CHLORO-PHENYL) -1H-PYRROLO [2, 3-B] PYRIDINE-3-CARBONYL] -2, 4-DIFLUORO-PHENY L } -AMIDE COMPOSITIONS AND USES THEREOF
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 14356
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1202937
Patent: PROPANE-I-SULFONIC ACID {3-[5-(4-CHLORO-PHENYL)-1H-PYRROLO[2,3-B]PYRIDINE-3-CARBONYL]-2,4-DIFLUORO-PHENYL}-AMIDE COMPOSITIONS AND USES THEREOF
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1739994
Estimated Expiration: ⤷ Start Trial
Patent: 120006006
Estimated Expiration: ⤷ Start Trial
Patent: 120101439
Estimated Expiration: ⤷ Start Trial
Patent: 170058465
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 52386
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1040179
Patent: Compositions and uses therof
Estimated Expiration: ⤷ Start Trial
Patent: 04719
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 11000436
Patent: PROPANE- I-SULFONIC ACID {3- [5- (4 -CHLORO-PHENYL) -1H-PYRROLO [2, 3-B] PYRIDINE-3-CARBONYL] -2, 4-DIFLUORO-PHENY L } -AMIDE COMPOSITIONS AND USES THEREOF
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 8842
Patent: ТВЕРДА ДИСПЕРСІЯ, СПОСІБ ЇЇ ОДЕРЖАННЯ, А ТАКОЖ КОМПОЗИЦІЯ І ЛІКАРСЬКА ФОРМА, ЩО ЇЇ МІСТЯТЬ
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 540
Patent: COMPOSICIONES QUE INCLUYEN COMPUESTOS QUE CONTIENEN LA {3-[5-(4-CLORO-FENIL)-1H-PIRROLO[2,3-B]PIRIDINA-3-CARBONIL]-2,4-DIFLUOR-FENIL}-AMIDA DEL ÁCIDO PROPANO-1-SULFÓNICO Y MÉTODOS PARA FABRICAR ESTAS COMPOSICIONES
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ZELBORAF around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Costa Rica | 9677 | DERIVADOS DE PIRROLO(2,3-B) PIRIDINA COMO INHIBIDORES DE PROTEINA CINASA | ⤷ Start Trial |
| Japan | 2013510813 | ⤷ Start Trial | |
| Ecuador | SP11011282 | ⤷ Start Trial | |
| China | 102361870 | ⤷ Start Trial | |
| Hungary | E027370 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ZELBORAF
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1893612 | SPC/GB12/021 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: VEMURAFENIB AND PHARMACEUTICALLY ACCEPTABLE SALTS; REGISTERED: UK EU/1/12/751/001 20120221 |
| 1893612 | 122 5012-2012 | Slovakia | ⤷ Start Trial | FIRST REGISTRATION NO/DATE: EU/1/12/751/001, 20120217 |
| 1893612 | 132012902073472 | Italy | ⤷ Start Trial | PRODUCT NAME: VEMURAFENIB(ZELBORAF); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/12/751/001, 20120221 |
| 1893612 | 28/2012 | Austria | ⤷ Start Trial | PRODUCT NAME: VEMURAFENIB UND PHARMAZEUTISCH VERTRAEGLICHE SALZE DAVON; REGISTRATION NO/DATE: EU/1/12/751/001 (MITTEILUNG) 20120221 |
| 1893612 | 92035 | Luxembourg | ⤷ Start Trial | 92035, EXPIRES: 20270217 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for ZELBORAF
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.