Last updated: January 26, 2026
Summary
Bristol-Myers Squibb (BMS) stands as a leading global biopharmaceutical company with a focus on innovative medicines in oncology, immunology, cardiovascular, and fibrotic diseases. This report analyzes BMS's market position, key strengths, competitive differentiators, and strategic outlook within the dynamic pharmaceutical landscape. BMS's recent acquisitions, pipeline robustness, and focus on immuno-oncology position it as a formidable contender. The report offers a data-driven, comprehensive overview to inform stakeholders on BMS's strategic direction and competitive edge.
What Is Bristol-Myers Squibb’s Current Market Position?
Market Capitalization and Revenue
| Metric |
Value (2022) |
Source |
| Market Capitalization |
~$150 billion |
Financial Times (2022) |
| 2022 Revenue |
$46.4 billion |
BMS Annual Report (2022) |
Market Domination:
BMS ranks among top-tier global pharmaceutical firms, particularly excelling in immuno-oncology with its blockbuster drug Opdivo (nivolumab), a PD-1 checkpoint inhibitor. Its portfolio's diversification across oncology, immunology, and hematology bolsters resilience amid competitive pressures.
Revenue Breakdown by Segment
| Segment |
Percentage of Total Revenue |
Key Products |
Notes |
| Oncology |
65% |
Opdivo, Yervoy, Camzyos |
Primary growth driver, with a focus on immunotherapies |
| Hematology |
15% |
Abecma, Breyanzi, Inrebic |
Blood cancer treatments, accelerated growth |
| Cardiovascular & Fibrosis |
10% |
Orencia, Zeposia |
Diversified, less dominant |
| Immunology & Other |
10% |
Orencia, other pipeline components |
Moderate contribution, growth potential |
Global Footprint & Market Penetration
- Geographic Revenue Breakdown (2022):
| Region |
Revenue Share |
Notable Markets |
| U.S. |
55% |
Leading in oncology, strong reimbursement systems |
| Europe |
25% |
Robust adoption, competitive biosimilars |
| Asia-Pacific |
15% |
Emerging markets, pipeline expansion opportunities |
| Rest of World |
5% |
Developing markets, emerging healthcare infrastructure |
Competitive Positioning
| Competitor |
Market Share (Top Oncology Biotechs, 2022) |
Key Differentiators |
| Roche |
20% |
Leadership in biosimilars and diagnostics |
| Merck & Co. |
14% |
Strong oncology pipeline, immunotherapies |
| Novartis |
12% |
Generics and biosimilars, diversified R&D |
| Pfizer |
11% |
Broad portfolio, recent pipeline boosts |
| Bristol-Myers Squibb |
10-12% (est.) |
Focused leadership in immuno-oncology |
(Source: IQVIA Institute, 2022)
What Are Bristol-Myers Squibb’s Key Strengths?
Robust Oncology and Immunotherapy Portfolio
- Opdivo (nivolumab): Approved in over 65 indications globally, with annual sales exceeding $9 billion in 2022. Its versatility across lung, melanoma, renal, and other cancers consolidates BMS’s leadership.
- Yervoy (ipilimumab): Complementary immune checkpoint inhibitor, increasing combination therapy opportunities.
- Pipeline Robustness: Over 50 ongoing clinical trials targeting multiple tumor types and resistance mechanisms.
Strategic Acquisitions and Collaborations
- Celgene Acquisition (2019): A $74 billion deal, adding:
- Hematology portfolio (Revlimid, Pomalyst)
- Cancer cell therapy units (Abecma)
- Enhances pipeline capacity and market penetration.
- Partnerships: Collaborations with industry giants like Pfizer, AstraZeneca, and BeiGene bolster R&D and market access.
Innovative R&D Focus
- Emphasis on immuno-oncology, cell therapies, and precision medicine.
- Heavy investment (~$4 billion annually) in R&D (2022 figures), leading to:
- 15+ FDA-approved therapies since 2014.
- A pipeline targeting solid tumors, immunology, and fibrosis.
Financial Strength and Operating Efficiency
- Solid revenue growth (compound annual growth rate of ~12% from 2018-2022).
- Operating margins ~33%, supporting sustained investment in innovation and acquisitions.
Regulatory and Market Access Strategies
- Proactive engagement with health authorities (FDA, EMA).
- Focus on personalized medicine pathways reducing developmental risk.
- Payer negotiations to optimize reimbursement rates.
What Are the Strategic Opportunities & Challenges Facing Bristol-Myers Squibb?
Opportunities
| Opportunity Area |
Strategic Moves & Impacts |
| Expansion in Cell & Gene Therapy |
Continued pipeline development, leveraging Celgene assets, entering new indications |
| Digital & Data Transformation |
Use of real-world data and AI to refine clinical trials and personalize therapy |
| Emerging Market Penetration |
Tailored pricing, local partnerships to grow revenue in Asia-Pacific and LATAM |
| Focus on Rare and Orphan Diseases |
Diversification, reduced competition, and higher pricing potential |
Challenges
| Challenge |
Implication |
| Biosimilar Competition |
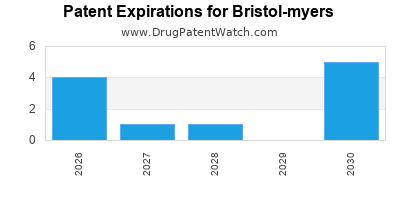
Patent expirations (e.g., Revlimid 2026) threaten revenue streams |
| Pricing & Reimbursement Pressures |
Especially in the U.S. and Europe, impacting profit margins |
| Pipeline Risks |
Clinical trial failures could diminish expected growth |
| Market Saturation |
Intense competition, especially in immuno-oncology, may limit growth |
| Regulatory Hurdles |
Navigating complex global approval processes can delay product launches |
Comparison with Key Competitors
| Aspect |
Bristol-Myers Squibb |
Roche |
Merck & Co. |
Novartis |
Pfizer |
| Focus Areas |
Oncology, immunology |
Diagnostics, biosimilars |
Oncology, vaccines |
Diversified |
Broad portfolio |
| Dominant Products |
Opdivo, Yervoy |
Herceptin, Avastin |
Keytruda |
Cosentyx |
Paxlovid, vaccines |
| R&D Spend (2022) |
~$4 billion |
$13 billion |
$7 billion |
$9.9 billion |
$13 billion |
| Pipeline Strength |
Oncology, cell therapy |
Oncology, rare diseases |
Oncology, vaccines |
Rare diseases, biosimilars |
Oncology, antivirals |
(Source: IQVIA, 2022; Company Reports 2022)
Key Strategic Insights
- Deepening Immuno-oncology Leadership: BMS’s top tier position hinges critically on Opdivo’s continued innovation, expanding indications, and combination therapies.
- Pipeline Diversification: Emphasize cell therapies, fibrosis, and rare diseases to offset biosimilar erosion.
- Market Expansion: Target high-growth regions with tailored strategies, integrating local partnerships.
- Innovation and Digital Utilization: Leverage AI and real-world evidence to accelerate R&D, improve efficiencies, and facilitate personalized medicine.
Deep Dive: BMS’s Pipeline and R&D Focus
| Key Areas |
Description |
Major Candidates |
Development Stage |
| Solid Tumors |
Lung, melanoma, colorectal |
Libtayo (cemiplimab), Opdivo combos |
Multiple Phase III & FDA approvals |
| Hematologic Malignancies |
Multiple myeloma, lymphoma |
Abecma, Breyanzi |
Approved/regulatory filings |
| Fibrosis & Autoimmune |
NASH, idiopathic pulmonary fibrosis |
Clinical trials underway |
Early-stage development |
| Cell & Gene Therapy |
Personalized cell therapies |
Multiple candidates in early phases |
Experimental, high risk/high reward |
Frequently Asked Questions (FAQs)
1. How does Bristol-Myers Squibb differentiate itself in the immuno-oncology market?
BMS leverages its extensive portfolio of immunotherapies, notably Opdivo and Yervoy, with broad indication approvals and combination strategies. Its strategic acquisitions, including Celgene, have bolstered its pipeline with cell therapies and hematologic oncology agents.
2. What impact will biosimilar competition have on BMS’s revenue streams?
Pipeline reliance on blockbuster drugs like Revlimid (biosimilar coming in 2026) poses risk. BMS actively diversifies its portfolio towards novel therapies and cell-based treatments to mitigate biosimilar erosion.
3. How is BMS planning to expand in emerging markets?
The company employs region-specific pricing strategies and forms local partnerships to penetrate markets, particularly in Asia-Pacific, where growing healthcare infrastructure offers growth potential.
4. What are BMS’s key R&D priorities moving forward?
Focus areas include immuno-oncology, cell and gene therapies, fibrosis, and precision medicine. The emphasis on combination therapies and digital health integration aims to sustain innovation leadership.
5. How does Bristol-Myers Squibb compare to its top competitors?
BMS maintains a specialized focus on immuno-oncology and cell therapies, while competitors like Roche lead in diagnostics and biosimilars, and Merck & Co. has a broader oncology pipeline. BMS’s strategic acquisitions have uniquely strengthened its position in hematology and immunotherapies.
Key Takeaways
- Bristol-Myers Squibb is a robust player in immuno-oncology, with a diversified pipeline and strategic acquisitions like Celgene underpinning its growth.
- The company’s revenue is heavily concentrated in oncology, particularly with Opdivo, which surpasses $9 billion annually.
- BMS faces upcoming patent expirations and biosimilar competition, necessitating pipeline diversification.
- Strategic focus areas include advancing cell therapies, expanding in emerging markets, and integrating digital innovations.
- Competition remains fierce, with Roche and Merck leading in biosimilars and broad oncology portfolios, respectively.
Actionable Insights for Stakeholders
- Investors should monitor pipeline advancements, especially in cell and gene therapy areas, which promise future growth.
- Business partners can leverage BMS’s focus on immuno-oncology for collaborative development opportunities.
- Regulators and policymakers should engage with BMS’s innovation strategies, as they drive advances in personalized medicine and oncology care.
References
- Bristol-Myers Squibb Annual Report 2022.
- IQVIA Institute, "The Global Oncology Market," 2022.
- Financial Times, "Top Pharmaceutical Conglomerates," 2022.
- Company press releases and clinical trial registries (clinicaltrials.gov).
- Market analytics reports, EvaluatePharma, 2022.