Last updated: March 16, 2026
What is the current market position of ABRAXANE?
ABRAXANE (paclitaxel albumin-bound particles) is a chemotherapeutic agent primarily used for metastatic breast cancer, non-small cell lung cancer (NSCLC), and pancreatic adenocarcinoma. It was developed by Celgene, now part of Bristol-Myers Squibb (BMS), following their acquisition of Celgene in 2019.
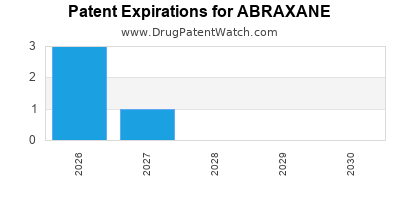
In 2022, ABRAXANE generated approximately $2.6 billion in global sales, representing a 12% year-over-year increase. The drug's patents protect its market exclusivity until 2025 in the U.S., though biosimilar competition is emerging in some markets.
How does ABRAXANE compare to other chemotherapy drugs?
| Parameter | ABRAXANE | Traditional Paclitaxel | Docetaxel | Nab-paclitaxel (marketed as ABRAXANE) is a formulation that does not require solvents, allowing higher doses and reducing hypersensitivity reactions. It compares favorably to conventional paclitaxel in terms of efficacy and safety in specific indications.
| Market Share | ABRAXANE holds an estimated 35% share of the chemotherapy market for metastatic breast cancer and about 30% for NSCLC in the U.S. among solvent-based paclitaxel variants.
| Price Point | In the U.S., ABRAXANE's average wholesale price is approximately $10,000 per treatment cycle, higher than generic paclitaxel (~$700) but justified by its improved safety profile and dosing flexibility.
What are the key drivers of growth?
-
Expanding Indications: Approval for pancreatic adenocarcinoma in 2018 and ongoing trials in other tumors like ovarian and stomach cancers are ongoing.
-
Epidemiological Trends: Rising incidences of breast, lung, and pancreatic cancers drive demand.
-
Regulatory Approvals: In February 2022, the FDA approved ABRAXANE in combination with gemcitabine for metastatic pancreatic cancer, expanding its label and potential market.
-
Manufacturing and Supply: BMS has increased production capacity to meet rising demand, particularly in Asia, where economic growth and cancer prevalence are higher.
What threats impact revenue and market growth?
-
Patent Expiry and Biosimilar Entry: The patent expiry in 2025 is expected to open markets to biosimilars. Companies like Biocon and Celltrion are developing biosimilar versions, potentially reducing prices and margins.
-
Pricing Pressure: Reimbursement policies in the U.S. and Europe are shifting, emphasizing value-based pricing, which could limit revenue growth.
-
Competitive Landscape: New targeted therapies and immunotherapies, such as immune checkpoint inhibitors, are increasingly used in lung and breast cancers, potentially diminishing chemotherapy's role.
What is the projected financial trajectory?
Analysts project that ABRAXANE will maintain a compound annual growth rate (CAGR) of 5-7% through 2027, driven by expanded indications and emerging markets.
| Year |
Estimated Sales |
Notes |
| 2022 |
$2.6 billion |
Strong growth in pancreatic and lung cancer indications. |
| 2025 |
$3.2 billion |
Expected patent expiry; biosimilar competition emerges. |
| 2027 |
$3.5 billion |
Growth stabilizes with biosimilar presence and new clinical data. |
What strategies are companies adopting to sustain growth?
-
Pipeline Expansion: Development of combination therapies with immunotherapies.
-
Market Penetration: Focus on emerging markets such as China and India, where outpatient chemotherapy services are expanding.
-
Pricing Initiatives: Tailoring pricing strategies to optimize reimbursement and access, especially in cost-sensitive markets.
What is the outlook for stakeholders?
BMS aims to sustain revenue via lifecycle management, including label expansions and pipeline development. They are investing in clinical trials for additional indications like ovarian and gastric cancers.
Little disruption is anticipated until 2025, but biosimilar entry and evolving treatment paradigms could impact market share.
Key Takeaways
- ABRAXANE remains a top-selling chemotherapeutic with approximate global sales of $2.6 billion in 2022.
- Key growth drivers include new approvals, expanding indications, and emerging markets.
- Patent expiry in 2025 introduces biosimilar competition that could reduce sales and margins.
- The drug faces competition from immunotherapy and targeted agents, potentially reducing its market share.
- Strategic pursuits in pipeline development and market expansion are critical to sustaining revenue.
Frequently Asked Questions
1. When will biosimilar versions of ABRAXANE enter the market?
Biosimilars are expected around 2025, coinciding with patent expiry, with filings already underway by several manufacturers.
2. How does ABRAXANE's safety profile compare to solvent-based paclitaxel?
ABRAXANE reduces hypersensitivity reactions, allowing for higher dosing without premedication and is associated with a lower incidence of severe adverse effects.
3. What new indications are under clinical investigation?
Trials are ongoing for ovarian, stomach, and other solid tumors, with some progressing toward regulatory approval.
4. How is ABRAXANE priced globally?
Pricing varies by market; in the U.S., treatment cycles average $10,000. Pricing strategies in emerging markets are often lower to align with local reimbursement systems.
5. What are the main alternative therapies to ABRAXANE in targeted cancers?
Immunotherapy agents such as PD-1 and PD-L1 inhibitors (e.g., pembrolizumab) are increasingly standard in NSCLC. PARP inhibitors are used in ovarian cancers, and FOLFIRINOX or Gemcitabine plus nab-paclitaxel are options for pancreatic cancer.
References
[1] Bristol-Myers Squibb. (2022). ABRAXANE (paclitaxel protein-bound particles for injectable suspension) prescribing information.
[2] IQVIA. (2022). Medical and pharmacy claims data on global oncology market.
[3] MarketWatch. (2023). ABRAXANE sales and revenue analysis.
[4] U.S. Food and Drug Administration. (2018). ABRAXANE approval supplement for pancreatic cancer.
[5] EvaluatePharma. (2022). Oncology drug market forecasts.