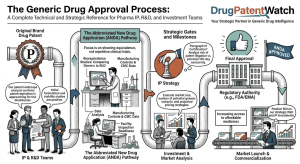
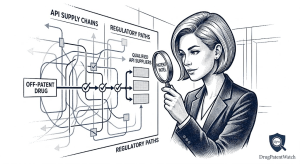
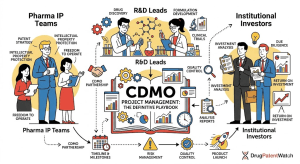
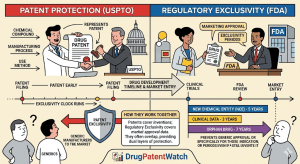
Drug Market Entry Intelligence: The Definitive Guide for Pharma Strategists, IP Teams, and Institutional Investors
1. The $2.8 Trillion Arithmetic: Market Structure and Strategic Stakes The global pharmaceutical market reached approximately $1.6 trillion in 2024 […]