VENLAFAXINE Drug Patent Profile
✉ Email this page to a colleague
When do Venlafaxine patents expire, and what generic alternatives are available?
Venlafaxine is a drug marketed by Almatica, Alembic, Anchen Pharms, Annora Pharma, Aurobindo Pharma Ltd, Dr Reddys Labs Ltd, Granules, Inventia Hlthcare, Macleods Pharms Ltd, Medicap Labs, Nostrum Pharms Llc, Orbion Pharms, Pharmobedient, Sciegen Pharms, Teva, Torrent, Valeant Pharms North, Yichang Humanwell, Zydus Pharms Usa Inc, Abon Pharms Llc, Adaptis, Ajanta Pharma Ltd, Alkem Labs Ltd, Ascent Pharms Inc, Cadila Pharms Ltd, Dexcel, Mpp Pharma, Osmotica Pharm Us, Pharmadax, Somerset Theraps Llc, Sun Pharm, Unique, Zydus Pharms, Alembic Pharms Ltd, Amneal Pharms, Aurobindo Pharma, Chartwell Rx, Heritage, Pliva Hrvatska Doo, Prinston Inc, Sun Pharm Inds Inc, Yaopharma Co Ltd, and Zydus Pharms Usa. and is included in forty-eight NDAs. There is one patent protecting this drug.
The generic ingredient in VENLAFAXINE is venlafaxine hydrochloride. There are seventy-one drug master file entries for this compound. Sixty-two suppliers are listed for this compound. Additional details are available on the venlafaxine hydrochloride profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Venlafaxine
A generic version of VENLAFAXINE was approved as venlafaxine hydrochloride by TEVA on August 3rd, 2006.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for VENLAFAXINE?
- What are the global sales for VENLAFAXINE?
- What is Average Wholesale Price for VENLAFAXINE?
Summary for VENLAFAXINE
| US Patents: | 0 |
| Applicants: | 43 |
| NDAs: | 48 |
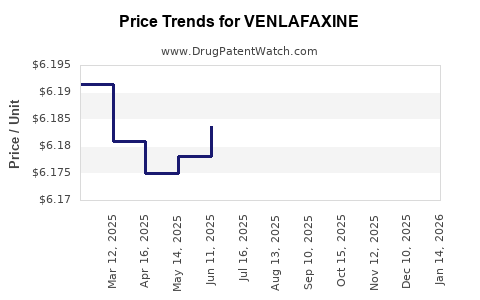
| Drug Prices: | Drug price information for VENLAFAXINE |
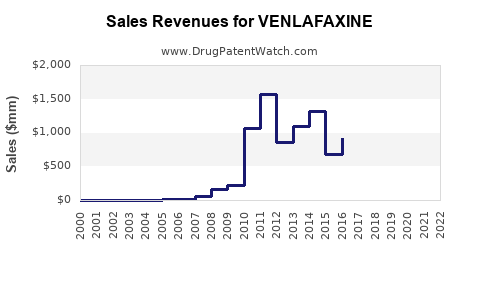
| Drug Sales Revenues: | Drug sales revenues for VENLAFAXINE |
| What excipients (inactive ingredients) are in VENLAFAXINE? | VENLAFAXINE excipients list |
| DailyMed Link: | VENLAFAXINE at DailyMed |
US Patents and Regulatory Information for VENLAFAXINE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cadila Pharms Ltd | VENLAFAXINE HYDROCHLORIDE | venlafaxine hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 211323-002 | Aug 29, 2019 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Zydus Pharms | VENLAFAXINE HYDROCHLORIDE | venlafaxine hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 215622-004 | Aug 30, 2022 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Teva | VENLAFAXINE HYDROCHLORIDE | venlafaxine hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 076565-001 | Jun 28, 2010 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Orbion Pharms | VENLAFAXINE HYDROCHLORIDE | venlafaxine hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 091123-001 | Jul 11, 2011 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |