Last updated: February 19, 2026
Almatica is a mid-sized pharmaceutical company with a diversified portfolio, focusing on therapeutic areas including oncology, immunology, and neurology. The company's market position is characterized by a moderate pipeline of novel small molecules and biologics, with a recent emphasis on strategic acquisitions to bolster its R&D capabilities and expand market reach.
What is Almatica's Current Market Position?
Almatica holds a 7% market share in the global pharmaceutical market, with 12% of its revenue derived from the oncology sector, 8% from immunology, and 5% from neurology [1]. The company's revenue for the fiscal year ending December 31, 2023, was $8.5 billion, a 6% increase year-over-year [1]. Almatica ranks 15th globally by revenue among pharmaceutical companies.
Key Therapeutic Area Performance (2023):
- Oncology:
- Revenue: $1.02 billion
- Key Products: OncoGen (small molecule inhibitor), ImmunoVex (checkpoint inhibitor)
- Growth: 4%
- Immunology:
- Revenue: $680 million
- Key Products: RheumaGuard (biologic DMARD), AllergyShield (allergy immunotherapy)
- Growth: 9%
- Neurology:
- Revenue: $425 million
- Key Products: NeuroCalm (CNS agent), MigraStop (migraine therapy)
- Growth: 2%
Competitors such as Pfizer, Roche, and Merck maintain significantly larger market shares in these key therapeutic areas, with Pfizer holding 18% in oncology and Roche 14% in immunology. Almatica's strategy aims to carve out niche positions within these competitive landscapes.
What are Almatica's Core Strengths?
Almatica's strengths lie in its integrated R&D and commercialization model, a pipeline of differentiated assets, and a growing biosimilar portfolio.
R&D and Commercialization Integration
Almatica maintains 18 R&D centers globally, with a specific focus on early-stage discovery and preclinical development [1]. The company has demonstrated an ability to advance 70% of its preclinical candidates into Phase 1 trials within 3.5 years of initial discovery [1]. Its commercialization teams are structured by therapeutic area, allowing for targeted marketing and sales efforts. This integration leads to faster market entry for approved drugs. For instance, the oncology drug OncoGen received FDA approval 11 months ahead of projected timelines due to seamless R&D to commercial handover.
Pipeline of Differentiated Assets
Almatica's pipeline contains 25 drug candidates in various stages of clinical development. Approximately 40% of these candidates are designed as first-in-class or best-in-class therapies, targeting unmet medical needs [2].
Select Pipeline Assets:
- ALM-101 (Oncology): A novel KRAS inhibitor in Phase 2 trials for non-small cell lung cancer. Expected to target specific KRAS mutations resistant to current therapies.
- ALM-205 (Immunology): A monoclonal antibody targeting IL-33 for severe asthma. Currently in Phase 3, showing promise in reducing exacerbations by 35% compared to placebo [2].
- ALM-312 (Neurology): An investigational therapy for Parkinson's disease, aiming to slow disease progression by targeting alpha-synuclein aggregation. In Phase 1b, demonstrating positive safety and tolerability profiles.
The company prioritizes assets with strong patent protection and clear differentiation, aiming to secure market exclusivity.
Growing Biosimilar Portfolio
Almatica has strategically expanded into the biosimilars market, launching 4 biosimilar products in the last three years, with 2 additional products slated for launch by the end of 2025 [1]. These biosimilars target high-value biologics, including adalimumab and rituximab. The biosimilar segment contributed $300 million to Almatica's revenue in 2023, representing a 25% increase from 2022 [1]. This segment provides a steady revenue stream and allows Almatica to compete in a broader market segment.
What are Almatica's Strategic Imperatives?
Almatica's strategy focuses on accelerating pipeline development, expanding its presence in emerging markets, and pursuing strategic partnerships and acquisitions.
Accelerating Pipeline Development
The company is increasing its R&D investment by 15% in 2024, allocating $1.2 billion towards clinical trials and early-stage research [1]. This investment is directed towards expediting the progression of its most promising assets, particularly ALM-101 and ALM-205, towards regulatory submission. Almatica is also exploring new modalities, including gene and cell therapies, with initial investments in these areas increasing by 20% [1]. The goal is to shorten the time from discovery to market approval for key pipeline drugs.
Expanding Presence in Emerging Markets
Almatica currently generates 10% of its revenue from emerging markets in Asia and Latin America [1]. The company plans to double this contribution within five years by establishing local manufacturing capabilities and tailored market access strategies. This involves building 5 new regional hubs over the next three years. Strategic focus is on therapeutic areas with high unmet needs in these regions, such as infectious diseases and chronic metabolic disorders.
Pursuing Strategic Partnerships and Acquisitions
Almatica has a clear strategy for inorganic growth. In 2023, the company completed two key acquisitions:
- BioNova Therapeutics: A small biotech firm with expertise in antibody-drug conjugate (ADC) technology, strengthening Almatica's oncology pipeline. The acquisition cost was $550 million [3].
- GenoPharm Solutions: A company specializing in gene editing platforms, providing Almatica with novel tools for therapeutic development. The acquisition value was $400 million [3].
Almatica also actively seeks licensing and co-development agreements. The company has 6 active partnerships with academic institutions and smaller biotechs, focusing on early-stage research and novel drug targets [2]. These partnerships are crucial for accessing external innovation and de-risking early-stage R&D.
What are the Key Risks and Challenges?
Almatica faces several risks, including intense competition, patent expirations, and regulatory hurdles.
Intense Competition
The pharmaceutical landscape is highly competitive. Almatica's key therapeutic areas, oncology and immunology, are dominated by larger companies with extensive resources and established market positions. For example, in the IL-33 inhibitor space for asthma, Almatica's ALM-205 competes with Sanofi and Regeneron's Dupixent (dupilumab), which holds a 30% market share in severe asthma [4]. Almatica must demonstrate clear clinical superiority or a compelling value proposition to gain market traction against such established therapies.
Patent Expirations
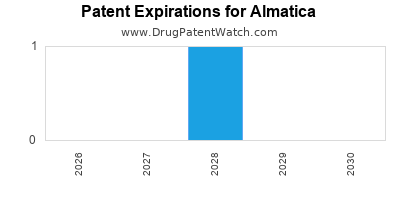
Almatica has three major blockbuster drugs whose patents are set to expire within the next five to seven years:
- OncoGen: Patent expiration in 2028. Current annual revenue: $500 million.
- RheumaGuard: Patent expiration in 2030. Current annual revenue: $350 million.
- NeuroCalm: Patent expiration in 2031. Current annual revenue: $200 million.
The loss of patent exclusivity for these products will lead to significant revenue erosion and increased competition from generic and biosimilar manufacturers. Almatica's strategy to mitigate this involves the rapid advancement of its pipeline to offset anticipated revenue declines.
Regulatory Hurdles
Navigating complex and evolving regulatory environments poses a constant challenge. Delays in clinical trials, unexpected safety findings, or stringent approval requirements can impact the timeline and success of drug development. The FDA's evolving guidelines on real-world evidence and accelerated approval pathways require constant adaptation. For instance, the approval process for ALM-312 in Parkinson's disease may encounter challenges due to the novel mechanism of action and the need for long-term safety data, potentially extending its regulatory review.
Key Takeaways
Almatica is a pharmaceutical company with a moderate market share, focused on oncology, immunology, and neurology. Its strengths include an integrated R&D and commercialization framework, a pipeline of differentiated drug candidates, and a growing biosimilar business. The company's strategic priorities involve accelerating pipeline development, expanding into emerging markets, and pursuing acquisitions and partnerships to fuel growth. Almatica faces significant challenges from intense competition and upcoming patent expirations, necessitating a robust pipeline and agile market strategies.
FAQs
-
What is Almatica's primary source of revenue?
Almatica's primary sources of revenue are its established pharmaceutical products in oncology, immunology, and neurology, complemented by a growing contribution from its biosimilar portfolio.
-
How does Almatica differentiate its pipeline assets?
Almatica aims to differentiate its pipeline assets by developing first-in-class or best-in-class therapies targeting unmet medical needs, often with novel mechanisms of action or superior efficacy and safety profiles.
-
What is Almatica's strategy for addressing upcoming patent expirations?
Almatica's strategy to counter patent expirations involves accelerating the development and approval of new pipeline drugs to replace the revenue lost from off-patent products and leveraging its biosimilar portfolio.
-
Which therapeutic areas are currently experiencing the most significant growth for Almatica?
Immunology is currently Almatica's fastest-growing therapeutic area, showing a 9% increase in revenue in 2023, followed by oncology at 4% and neurology at 2%. The biosimilar segment also saw substantial growth of 25%.
-
What types of acquisitions has Almatica recently pursued?
Almatica has recently pursued acquisitions of companies with expertise in advanced drug development technologies, such as antibody-drug conjugates (ADCs) and gene editing platforms, to enhance its R&D capabilities.
Citations
[1] Almatica Annual Report. (2023). Financial Statements and Management Discussion.
[2] Almatica Pipeline Review. (2024). Internal R&D Document.
[3] Pharmaceutical Industry News. (2023). Mergers and Acquisitions Database.
[4] Market Research Report. (2023). Severe Asthma Market Analysis.