Share This Page
Drug Sales Trends for VENLAFAXINE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for VENLAFAXINE (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
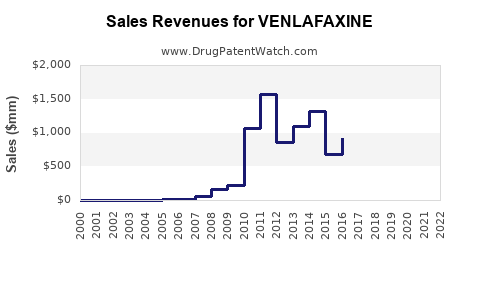
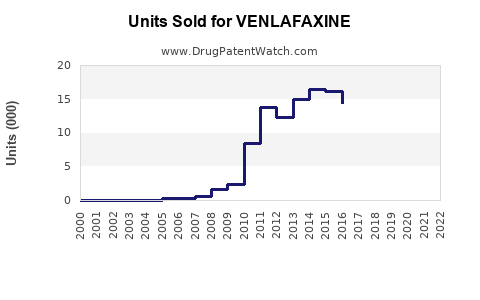
Annual Sales Revenues and Units Sold for VENLAFAXINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| VENLAFAXINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| VENLAFAXINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| VENLAFAXINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Venlafaxine Market Analysis and Sales Projections
Venlafaxine, a serotonin-norepinephrine reuptake inhibitor (SNRI) primarily used to treat depression and anxiety disorders, faces a mature and competitive market. Generic competition has significantly impacted originator sales, with robust sales driven by established efficacy and broad therapeutic indications. The market is characterized by stable demand, with incremental growth expected from emerging markets and the potential for new delivery systems or formulations.
What is the Current Market Landscape for Venlafaxine?
The global market for venlafaxine is substantial, with total sales estimated to be in the billions of dollars annually. The dominant force in this market is generic venlafaxine, which entered widespread availability following the patent expiration of the originator product, Effexor XR, in the United States in 2010. [1] This has led to significant price erosion and a fragmented manufacturer landscape.
Key market segments include immediate-release (IR) and extended-release (XR) formulations. The XR formulation, due to its simplified dosing regimen and potentially improved tolerability, accounts for a significant portion of the market share.
The primary therapeutic indications driving venlafaxine sales are:
- Major Depressive Disorder (MDD)
- Generalized Anxiety Disorder (GAD)
- Social Anxiety Disorder (SAD)
- Panic Disorder (PD)
The prevalence of these conditions globally, particularly depression and anxiety, provides a consistent demand for effective treatment options.
Major Geographic Markets
North America, particularly the United States, remains the largest market for venlafaxine due to high healthcare spending, established diagnostic rates for mental health conditions, and a well-developed pharmaceutical distribution network. Europe, with its sizable populations and robust healthcare systems in countries like Germany, the UK, France, and Spain, represents the second-largest market. Asia-Pacific is an emerging growth market, driven by increasing awareness of mental health issues, improving healthcare infrastructure, and a growing middle class in countries such as China and India. [2]
Key Market Drivers
- High Prevalence of Mental Health Disorders: The persistent and rising incidence of depression and anxiety disorders globally fuels demand for antidepressants like venlafaxine.
- Established Efficacy and Safety Profile: Venlafaxine has a long history of clinical use, with a well-documented efficacy and safety profile, making it a trusted option for healthcare providers and patients.
- Cost-Effectiveness of Generics: The availability of affordable generic venlafaxine makes it accessible to a broader patient population, especially in markets with price-sensitive healthcare systems.
- Broad Label Indications: Venlafaxine's effectiveness across multiple psychiatric conditions expands its patient base.
Market Restraints
- Intense Generic Competition: The high number of generic manufacturers leads to significant price pressure, limiting revenue growth for individual companies.
- Development of Newer Antidepressants: The emergence of newer drug classes with potentially different side-effect profiles or novel mechanisms of action can divert market share from established drugs like venlafaxine.
- Side Effect Profile: While generally well-tolerated, venlafaxine can cause side effects such as nausea, insomnia, and sexual dysfunction, which can lead to patient non-adherence or switching to alternative medications.
- Regulatory Scrutiny: Like all pharmaceuticals, venlafaxine is subject to ongoing regulatory oversight and potential post-market studies.
What are the Sales Projections for Venlafaxine?
Sales projections for venlafaxine are largely influenced by the dynamics of the generic market and the sustained demand for its therapeutic indications. The market is expected to exhibit modest growth, with a compound annual growth rate (CAGR) typically ranging from 2% to 4% over the next five to seven years. [3]
Global Sales Estimates
- Current Market Size: Estimated to be between $2.5 billion and $3.0 billion globally in recent years. [4]
- Projected Market Size (2028): Expected to reach $3.0 billion to $3.8 billion, reflecting the moderate growth trajectory. [4]
Regional Sales Projections
- North America: Expected to maintain its leading position but with slower growth, likely in the 1% to 3% CAGR range, due to market saturation and competitive pricing.
- Europe: Projected to grow at a slightly higher rate than North America, around 2% to 4% CAGR, driven by an aging population and increasing mental health awareness.
- Asia-Pacific: Anticipated to be the fastest-growing region, with a CAGR of 4% to 6%, fueled by expanding healthcare access, rising disposable incomes, and destigmatization of mental health.
- Latin America and Middle East & Africa: These markets are expected to show growth in the 3% to 5% CAGR range, driven by improving healthcare infrastructure and increasing adoption of generic pharmaceuticals.
Factors Influencing Projections
- Generic Pricing Trends: Continued price erosion due to competition will cap absolute revenue growth, even if unit sales increase.
- Market Penetration in Emerging Economies: The primary driver for higher growth rates will be increased access and prescription of venlafaxine in developing countries.
- Competition from Biosimil/Generic Antidepressants: The introduction of new generic antidepressants or alternative treatment modalities could impact venlafaxine's market share.
- Clinical Practice Guidelines: Updates to treatment guidelines that favor or disfavor venlafaxine for specific patient profiles will influence prescribing patterns.
What is the Competitive Landscape for Venlafaxine Manufacturers?
The competitive landscape for venlafaxine is highly fragmented, dominated by generic pharmaceutical manufacturers. The original innovator, Pfizer, with its Effexor XR brand, has seen its market share drastically diminish due to patent expiries.
Major Generic Manufacturers
Numerous pharmaceutical companies globally manufacture and market generic venlafaxine. Some of the prominent players include:
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Mylan N.V. (now part of Viatris)
- Aurobindo Pharma Ltd.
- Lupin Ltd.
- Gland Pharma Ltd.
- Torrent Pharmaceuticals Ltd.
These companies compete primarily on price, supply chain efficiency, and market reach. The ability to secure regulatory approvals in key markets and maintain consistent product quality are critical for success.
Brand vs. Generic Dynamics
The vast majority of venlafaxine sales are accounted for by generic products. The price difference between branded Effexor XR and its generic equivalents is substantial, often exceeding 80-90%, making generics the preferred choice for most healthcare systems and patients. [5]
Strategies of Key Players
- Cost Leadership: Generic manufacturers focus on optimizing production costs to offer the lowest possible prices.
- Supply Chain Management: Ensuring a reliable and efficient supply chain is crucial for meeting market demand and securing contracts with distributors and pharmacies.
- Regulatory Expertise: Navigating complex regulatory pathways in different countries to obtain marketing authorizations is a key competitive advantage.
- Market Access and Distribution: Establishing strong distribution networks and securing preferred provider status with pharmacy benefit managers (PBMs) and national health services are vital.
- Product Portfolio Diversification: While venlafaxine is a significant product, companies often leverage their manufacturing capabilities for a broader range of generics.
Barriers to Entry
While the market is crowded, barriers to entry still exist:
- Regulatory Hurdles: Obtaining approvals from agencies like the FDA and EMA is time-consuming and costly.
- Capital Investment: Establishing manufacturing facilities that meet Good Manufacturing Practice (GMP) standards requires significant capital.
- Intellectual Property: While the primary patents for venlafaxine have expired, there can be secondary patents related to specific formulations or manufacturing processes that could pose challenges.
- Established Relationships: Long-standing relationships between existing manufacturers and distributors create an entrenched market.
What is the Regulatory Status and Patent Landscape of Venlafaxine?
The patent landscape for venlafaxine is largely characterized by expired primary patents, opening the door for extensive generic competition.
Key Patent Expirations
The most significant patent for the extended-release formulation of venlafaxine, Effexor XR (U.S. Patent No. 4,775,524), expired in the United States in 2010. [1] Similar patent expiries occurred in other major markets shortly thereafter.
Current Patent Activity
While primary composition of matter patents have expired, companies may pursue patents related to:
- New Formulations: Development of novel drug delivery systems, such as improved extended-release mechanisms, orally disintegrating tablets, or injectable forms.
- Manufacturing Processes: Patented methods for more efficient or cost-effective synthesis of venlafaxine or its intermediates.
- New Indications: While unlikely for a well-established drug, research into novel therapeutic uses could theoretically lead to new patent filings, though success in this area for venlafaxine is improbable.
However, the majority of patent litigation in the venlafaxine space has focused on challenging or defending secondary patents, primarily by generic manufacturers seeking to enter the market.
Regulatory Approvals
Venlafaxine is approved for marketing in virtually all major countries by their respective regulatory authorities. In the United States, it is approved by the Food and Drug Administration (FDA). In Europe, it is approved by the European Medicines Agency (EMA) and subsequently by national competent authorities.
- FDA Approval History: Venlafaxine hydrochloride (immediate-release) was first approved in 1993. The extended-release formulation (Effexor XR) was approved in 1997.
- EMA Approval: Similar approval timelines exist across the European Union member states.
Post-Market Surveillance and Generic Approvals
Regulatory agencies continue to monitor the safety and efficacy of venlafaxine. Generic manufacturers must demonstrate bioequivalence to the reference listed drug (RLD) to gain approval. The FDA utilizes the Abbreviated New Drug Application (ANDA) pathway for generic drug approvals. [6]
Implications for R&D and Investment
- Limited Opportunities for Novel Drug Discovery: The expired patents mean that true novel drug discovery for venlafaxine itself is unlikely to yield significant commercial returns, as the market is dominated by generics.
- Focus on Formulation and Delivery Systems: Innovation efforts are likely to be concentrated on developing improved formulations that offer patient benefits such as better adherence, reduced side effects, or alternative routes of administration. These may be patentable.
- Investment in Generic Manufacturing: Investment opportunities exist in companies that can efficiently and cost-effectively manufacture high-quality generic venlafaxine, particularly for emerging markets.
- Navigating Secondary Patents: Companies looking to enter the market or launch new formulations must carefully analyze the existing patent landscape to avoid infringement.
Key Takeaways
- The venlafaxine market is mature and dominated by generic competition, leading to significant price erosion.
- Global sales are estimated between $2.5-$3.0 billion annually, with projections indicating modest growth of 2-4% CAGR, reaching $3.0-$3.8 billion by 2028.
- North America and Europe are established markets, while the Asia-Pacific region presents the highest growth potential.
- Key market drivers include the high prevalence of mental health disorders and venlafaxine's established efficacy.
- Intense generic competition and the availability of newer antidepressants act as market restraints.
- The competitive landscape is fragmented, with numerous generic manufacturers vying for market share based on cost leadership and supply chain efficiency.
- Primary patents for venlafaxine have expired, with ongoing patent activity focused on secondary aspects like new formulations and manufacturing processes.
Frequently Asked Questions
1. What is the primary reason for the decline in sales of branded Effexor XR?
The primary reason for the decline in sales of branded Effexor XR is the expiration of its key patents, which allowed numerous generic manufacturers to enter the market with significantly lower-priced versions of the drug.
2. Are there any new clinical trials ongoing for venlafaxine that could impact its market?
While large-scale clinical trials for new indications are unlikely for a drug of this age, ongoing research may focus on specific patient subgroups, comparative effectiveness studies against newer antidepressants, or studies related to its use in combination therapies. Significant new therapeutic indications are not anticipated.
3. How does the cost-effectiveness of generic venlafaxine compare to newer antidepressants?
Generic venlafaxine is substantially more cost-effective than newer antidepressants, particularly novel compounds with unique mechanisms of action. This cost advantage is a critical factor in its continued widespread use, especially in public healthcare systems and for price-sensitive patient populations.
4. What are the main side effects of venlafaxine that patients should be aware of?
Common side effects of venlafaxine include nausea, dry mouth, dizziness, insomnia, somnolence, sweating, constipation, and sexual dysfunction. The extended-release formulation is designed to mitigate some of these by providing a more stable drug level in the bloodstream.
5. What is the outlook for venlafaxine in emerging markets over the next five years?
The outlook for venlafaxine in emerging markets is positive, with projected growth rates higher than in developed regions. This growth will be driven by increasing diagnosis rates of mental health conditions, expanding healthcare access, a growing middle class, and the affordability of generic venlafaxine.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Website - Specific page may vary, search for "Orange Book"]
[2] Grand View Research. (2023). Venlafaxine Market Size, Share & Trends Analysis Report. [Report Summary - Access may require subscription]
[3] Mordor Intelligence. (2023). Venlafaxine Market - Growth, Trends, COVID-19 Impact, and Forecasts (2023 - 2028). [Report Summary - Access may require subscription]
[4] Market Research Future. (2023). Venlafaxine Market Report. [Report Summary - Access may require subscription]
[5] GoodRx. (n.d.). Venlafaxine Prices, Coupons & Savings Tips. Retrieved from [GoodRx Website - Specific page may vary, search for "venlafaxine"]
[6] U.S. Food & Drug Administration. (n.d.). Abbreviated New Drug Applications (ANDAs) for Generic Drugs. Retrieved from [FDA Website - Specific page may vary, search for "ANDA generic drugs"]
More… ↓
