Last updated: February 19, 2026
What is the current market landscape for Valproic Acid?
Valproic acid is a well-established antiepileptic drug (AED) with broad indications including epilepsy, bipolar disorder, and migraine prophylaxis. Its patent expired in most markets by the late 2000s, resulting in a shift towards generic formulations. The global market size was valued at approximately USD 600 million in 2022 and is projected to grow at a compound annual growth rate (CAGR) of about 2.5% from 2023 to 2030 [1].
The drug's widespread usage and established safety profile maintain its stability in the AED segment. Some key markets include North America, Europe, and Asia Pacific, with North America accounting for nearly 50% of sales due to robust epilepsy treatment adherence and insurance coverage.
How do regulatory statuses influence market dynamics?
The expiration of patents opened the market to generic competitors, leading to price erosion. As of 2023, the main branded producer, UCB Pharma, has shifted focus towards second-generation formulations with improved safety profiles, such as extended-release versions, targeting niche patient populations and improving adherence.
Regulatory approvals for new formulations are limited; efforts primarily focus on reformulations rather than novel therapeutic applications. The generic landscape, driven by multiple manufacturers, sustains competitive pricing pressure. Governments and payers favor generics, further stabilizing the market but constraining margins for innovator companies.
What are the key drivers and barriers affecting financial growth?
Drivers:
- High prevalence of epilepsy and bipolar disorder: An estimated 50 million individuals worldwide suffer from epilepsy, and bipolar disorder affects approximately 2.8% of the global population [2]. This sustained demand ensures steady revenue streams.
- Established reimbursement frameworks: Insurance and healthcare policies tend to favor generic drugs, supporting volumes.
- Patent expirations in major markets: Opened the market to competition but also contribute to lowered prices.
- Potential for extended indications: Off-label uses and ongoing research into neurodegenerative applications could broaden market reach.
Barriers:
- Market saturation: Near universal adoption reduces growth potential.
- Pricing pressures: Policy shifts towards cost containment restrict price increases.
- Safety concerns: Rare but serious adverse effects like hepatotoxicity and teratogenicity influence prescribing patterns.
- Limited innovation pipeline: Few new formulations or indications are in late-stage development, constraining future revenue growth.
How does the financial trajectory look over the next decade?
Analysts project a relatively stable revenue landscape for valproic acid, with an estimated CAGR of 2.5%. Revenue will primarily be driven by demand within existing indications, with minor growth fueled by reformulations and off-label expansions.
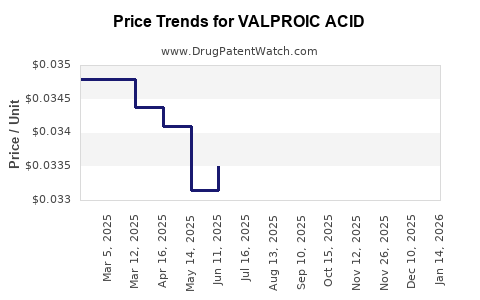
Generic competition will continue to exert price pressure, resulting in an average price decline of approximately 3-4% annually. Major branded players will attempt to offset this through niche products and combination therapies but with limited impact.
In terms of R&D investment, companies are expected to allocate minimal resources to valproic acid derivatives, focusing instead on newer AEDs and alternative therapies. As chemical patents have long expired, the primary revenue will come from volume sales rather than innovation-driven premiums.
What are the regional and geopolitical considerations?
North America and Western Europe will sustain steady demand due to established treatment protocols and reimbursement policies. Asia-Pacific shows potential for growth, driven by expanding healthcare infrastructure and increasing diagnosis rates.
Regulatory landscapes vary; some countries impose stricter safety monitoring and prescribing guidelines, affecting market access and sales volumes. Political shifts towards drug price regulation could further pressure profit margins in key markets.
Summary table of key metrics
| Indicator |
2022 Estimate |
2023-2030 Projection |
| Market size |
USD 600 million |
CAGR 2.5%; stabilizing around USD 700 million by 2030 |
| Patent status |
Expired in most markets |
Generic dominance remains dominant |
| Price decline |
Neutral to slight decline (~3%) per year |
Continued downward pressure |
| Major therapeutic indications |
Epilepsy, bipolar disorder, migraine |
Stable demand, possible minor off-label uses |
| Innovation pipeline |
Limited; reformulations only |
Minimal; focus on second-generation formulations |
| Regional demand |
North America (50%), Europe |
Growth in Asia Pacific |
Key takeaways
- The valproic acid market is mature, characterized by generic competition and stable demand.
- Revenue growth remains modest, driven primarily by volume rather than price increases.
- Innovation efforts focus on reformulations, with limited new indications.
- Regional differences influence sales continuity, particularly in Asia-Pacific.
- Future revenue prospects are constrained; strategic focus on niche formulations and off-label applications may offer marginal gains.
FAQs
1. How does patent expiration affect valproic acid market share?
Patent expiry led to the entry of generic manufacturers, reducing prices and earnings for branded products. It increased market volume but depressed margins.
2. What are the safety concerns impacting valproic acid sales?
Serious adverse effects like hepatotoxicity and teratogenicity limit prescribing, especially in women of childbearing age, influencing market dynamics.
3. Are there any new formulations under development?
Most development efforts focus on extended-release versions with improved safety profiles; no major novel formulations are in late-stage development.
4. What is the future outlook for profit margins in this market?
Margins are expected to decline gradually due to ongoing price pressures and generic competition, with little room for revenue expansion outside volume growth.
5. How does regional regulation influence sales?
Stricter safety monitoring and prescribing guidelines in Europe and North America help contain sales, whereas emerging markets like Asia have increasing demand due to healthcare access expansion.
References
[1] MarketResearch.com. (2023). Valproic Acid Market Size & Forecast.
[2] World Health Organization. (2022). Epilepsy Fact Sheet.