VAGIFEM Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Vagifem, and when can generic versions of Vagifem launch?
Vagifem is a drug marketed by Novo Nordisk Inc and is included in one NDA.
The generic ingredient in VAGIFEM is estradiol. There are seventy-five drug master file entries for this compound. Forty-six suppliers are listed for this compound. Additional details are available on the estradiol profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Vagifem
A generic version of VAGIFEM was approved as estradiol by BARR LABS INC on October 22nd, 1997.
Summary for VAGIFEM
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 109 |
| Clinical Trials: | 12 |
| Patent Applications: | 6,715 |
| Formulation / Manufacturing: | see details |
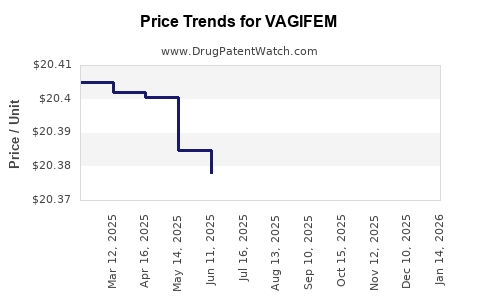
| Drug Prices: | Drug price information for VAGIFEM |
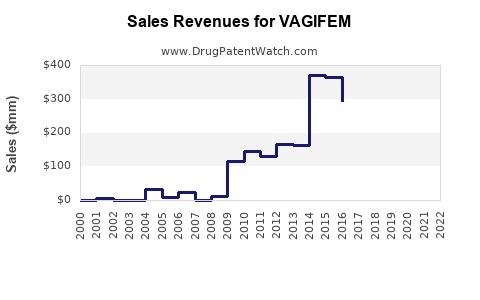
| Drug Sales Revenues: | Drug sales revenues for VAGIFEM |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for VAGIFEM |
| What excipients (inactive ingredients) are in VAGIFEM? | VAGIFEM excipients list |
| DailyMed Link: | VAGIFEM at DailyMed |
Recent Clinical Trials for VAGIFEM
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Angelica Lindén Hirschberg | Phase 4 |
| University Hospitals Cleveland Medical Center | Phase 4 |
| University of Minnesota, MN | Phase 3 |
Pharmacology for VAGIFEM
| Drug Class | Estrogen |
| Mechanism of Action | Estrogen Receptor Agonists |
Anatomical Therapeutic Chemical (ATC) Classes for VAGIFEM
US Patents and Regulatory Information for VAGIFEM
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Novo Nordisk Inc | VAGIFEM | estradiol | TABLET;VAGINAL | 020908-002 | Nov 25, 2009 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Novo Nordisk Inc | VAGIFEM | estradiol | TABLET;VAGINAL | 020908-001 | Mar 26, 1999 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for VAGIFEM
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Novo Nordisk Inc | VAGIFEM | estradiol | TABLET;VAGINAL | 020908-002 | Nov 25, 2009 | ⤷ Try a Trial | ⤷ Try a Trial |
| Novo Nordisk Inc | VAGIFEM | estradiol | TABLET;VAGINAL | 020908-002 | Nov 25, 2009 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for VAGIFEM
See the table below for patents covering VAGIFEM around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Germany | 20120246 | ⤷ Try a Trial | |
| Japan | 4851057 | ⤷ Try a Trial | |
| Canada | 2258310 | INSTRUMENT POUR L'INTRODUCTION D'UN SUPPOSITOIRE (INSTRUMENT FOR INSERTING A SUPPOSITORY) | ⤷ Try a Trial |
| Japan | 2010189409 | USE OF OESTROGEN IN MANUFACTURE OF COMPOSITION CONTAINING OESTROGEN FOR TREATMENT OF ATROPHIC VAGINITIS | ⤷ Try a Trial |
| Germany | 69722203 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for VAGIFEM
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0770388 | 2009/012 | Ireland | ⤷ Try a Trial | PRODUCT NAME: QLAIRA-ESTRADIOL VALERATE/DIENOGEST; NAT REGISTRATION NO/DATE: PA1410/58/1 20090109; FIRST REGISTRATION NO/DATE: BE327792 20081103 |
| 0770388 | PA2009004,C0770388 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: ESTRADIOLI VALERAS + DIENOGESTUM; NAT. REGISTRATION NO/DATE: LT/1/09/1512/001, 2009 04 06 LT/1/09/1512/002, 2009 04 06 LT/1/09/1512/003 20090406; FIRST REGISTRATION: BE 327792 20081103 |
| 1380301 | 2009C/007 | Belgium | ⤷ Try a Trial | PRODUCT NAME: DROSPIRENONE-ETHINYLESTRADIOL; AUTHORISATION NUMBER AND DATE: BE321386 20080811 |
| 1453521 | 300814 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: LEVONORGESTREL EN ETHINYLESTRADIOL; NATIONAL REGISTRATION NO/DATE: RVG 117453 20151211; FIRST REGISTRATION: SK 17/0017/15-S 20150211 |
| 0136011 | 99C0003 | Belgium | ⤷ Try a Trial | PRODUCT NAME: ESTRADIOL AND NORETHISTERONE; FIRST REGISTRATION NO/DATE: 403 IS 106 F3 19980928; FIRST REGISTRATION: SE 14007 19980306 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |


