Last updated: February 19, 2026
This report analyzes the market dynamics and financial trajectory of VAGIFEM, a prescription medication for vulvovaginal atrophy (VVA). The analysis covers market size, growth drivers, competitive landscape, patent status, and projected financial performance.
What is VAGIFEM and What Conditions Does It Treat?
VAGIFEM is a low-dose vaginal estrogen tablet used for the treatment of vulvovaginal atrophy (VVA), also known as vaginal atrophy. VVA is a chronic condition characterized by the thinning, drying, and inflammation of the vaginal walls, typically occurring as a result of estrogen deficiency [1]. This deficiency is most commonly associated with menopause, particularly after surgical menopause (oophorectomy) or the natural cessation of ovarian function.
Symptoms of VVA include:
- Vaginal dryness
- Burning sensation
- Irritation
- Pain during sexual intercourse (dyspareunia)
- Urinary symptoms such as urgency, frequency, and recurrent urinary tract infections (UTIs) [1].
VAGIFEM delivers estradiol directly to the vaginal tissues, providing localized estrogen replacement therapy. This targeted delivery minimizes systemic absorption of estrogen, thereby reducing the risk of systemic side effects often associated with oral hormone replacement therapy. The treatment aims to restore vaginal health, alleviate symptoms, and improve the quality of life for affected women [2].
What is the Current Market Size and Projected Growth for VAGIFEM?
The global market for VVA treatments is substantial and exhibits consistent growth, driven by an aging female population and increasing diagnosis rates. While specific market share figures for VAGIFEM are proprietary to its manufacturer, the broader market for vaginal estrogen therapies is estimated to be in the hundreds of millions of dollars annually.
Key Market Indicators:
- Global Menopause Market: The overall global menopause market, which includes treatments for VVA, hot flashes, and osteoporosis, is projected to grow significantly. Reports estimate this market to reach over $23 billion by 2027, with a compound annual growth rate (CAGR) of approximately 5% [3]. VVA treatments constitute a notable segment within this larger market.
- Aging Population: The increasing lifespan of women globally directly correlates with a larger demographic experiencing menopause and its associated symptoms, including VVA.
- Diagnosis and Awareness: Improved diagnostic tools and increased patient and physician awareness of VVA as a treatable condition are contributing to market expansion.
- Product Innovations: The development of various delivery methods for vaginal estrogen, including tablets, creams, and rings, provides patients with more options, potentially expanding the overall market.
Projected Growth Drivers for VAGIFEM (within its specific segment):
- Efficacy and Safety Profile: VAGIFEM's established efficacy in symptom relief and its favorable safety profile due to low systemic absorption are key drivers for continued prescription.
- Convenience of Administration: The once-daily tablet format is perceived as convenient by many patients.
- Physician Trust: Long-standing clinical data and physician familiarity with the product contribute to its sustained use.
- Limited Competition in Specific Niches: While the VVA market has several players, VAGIFEM holds a strong position in the low-dose vaginal tablet segment.
Who are the Key Competitors for VAGIFEM?
The market for vulvovaginal atrophy (VVA) treatments is competitive, with several pharmaceutical companies offering various forms of local estrogen therapy. VAGIFEM competes primarily with other low-dose vaginal estrogen products, including tablets, creams, and rings.
Primary Competitors:
- Estradiol Vaginal Cream (e.g., Estrace, Generic Equivalents): These creams deliver estradiol directly to the vaginal tissues. They are often prescribed for moderate to severe VVA symptoms.
- Pros: Can be easily adjusted in dosage.
- Cons: Potential for messiness and variability in application.
- Estradiol Vaginal Rings (e.g., Estring): These rings release estradiol at a continuous, low rate over several months.
- Pros: Long-acting, convenient (inserted every 3 months), low systemic absorption.
- Cons: Requires insertion by a healthcare professional or patient, can sometimes cause vaginal discomfort or discharge.
- Other Vaginal Estrogen Tablets (e.g., Imvexxy): These are newer tablet formulations designed for vaginal administration. Imvexxy, for instance, is a micronized estradiol vaginal insert.
- Pros: Direct delivery, often smaller and easier to insert than rings.
- Cons: May have different absorption profiles or require specific application techniques.
- Conjugated Equine Estrogen (CEE) Vaginal Cream (e.g., Premarin Vaginal Cream): While often used for VVA, Premarin is derived from pregnant mare urine and contains a mixture of estrogens.
- Pros: Long history of use.
- Cons: Concerns regarding the source of estrogen and potential for higher systemic absorption compared to estradiol-only products.
- DHEA Vaginal Inserts (e.g., Intrarosa): Prasterone (dehydroepiandrosterone, DHEA) is a prohormone that is converted to androgens and estrogens within vaginal tissues.
- Pros: Alternative mechanism of action, can be effective for dyspareunia.
- Cons: Newer to the market compared to estrogen therapies.
Competitive Landscape Factors:
- Dosage and Delivery Mechanism: The choice between tablets, creams, rings, and inserts is often patient-specific, influenced by symptom severity, preference for application, and physician recommendation.
- Efficacy and Safety Data: Clinical trials and real-world evidence supporting the effectiveness and safety of each product influence prescribing patterns.
- Insurance Coverage and Reimbursement: Pharmacy benefit manager (PBM) formularies and insurance coverage significantly impact patient access and physician choice.
- Pricing and Generics: The availability of generic versions of certain treatments can affect market share and profitability for branded products. VAGIFEM, being a branded product with specific proprietary formulations and delivery systems, navigates this landscape differently from older, off-patent creams.
What is the Patent Status and Exclusivity Landscape for VAGIFEM?
The patent and exclusivity landscape for VAGIFEM is critical to its market longevity and financial performance. Pharmaceutical patents protect the innovative aspects of a drug, granting the patent holder exclusive rights to market the drug for a specified period.
Key Aspects of VAGIFEM's Patent Status:
- Original Composition of Matter Patents: These would have covered the active pharmaceutical ingredient (estradiol) and its specific formulation for vaginal administration. These patents typically have a lifespan of 20 years from the filing date.
- Method of Use Patents: Additional patents may cover specific methods of treating VVA with VAGIFEM or particular patient populations.
- Formulation and Delivery System Patents: Patents can also protect the unique tablet formulation, the applicator device, or the slow-release mechanism, if applicable.
Exclusivity Periods:
Beyond patent expiry, regulatory exclusivities granted by agencies like the U.S. Food and Drug Administration (FDA) can provide additional market protection. These can include:
- New Chemical Entity (NCE) Exclusivity: Typically 5 years for a new drug.
- Orphan Drug Exclusivity: 7 years for drugs treating rare diseases.
- Pediatric Exclusivity: 6 months added to existing patent or exclusivity if pediatric studies are conducted.
- Data Exclusivity: 5 years for an NCE, 3 years for new clinical investigations supporting a new indication or formulation, and 12 years for biologics in the U.S.
Implications for VAGIFEM:
The patent expiration dates for VAGIFEM's core patents and any associated formulation patents directly influence when generic manufacturers can enter the market. Upon patent expiry and the lapse of any relevant exclusivity periods, generic versions of VAGIFEM can become available.
- Impact of Generic Entry: Generic entry typically leads to a significant decrease in the price of the drug and a substantial reduction in the market share of the branded product. Pharmaceutical companies often anticipate this by developing newer formulations, seeking new indications, or engaging in litigation to extend market exclusivity.
- Proprietary Delivery System: If VAGIFEM utilizes a unique or patented delivery system (e.g., a specific applicator, a novel tablet matrix for controlled release), this component could be a source of continued exclusivity even after the patent on the active ingredient expires, provided the delivery system itself remains patented.
As of the most recent publicly available information, specific patent expiry dates for VAGIFEM are not universally disclosed due to the complex and often litigious nature of patent protection. Manufacturers like Novo Nordisk (historically, with Leo Pharma being a key player in its development and marketing) or current rights holders would have specific insight into the remaining patent life and exclusivity periods. Companies planning to enter this market would conduct detailed Freedom-to-Operate (FTO) analyses to identify specific patents and potential infringement risks.
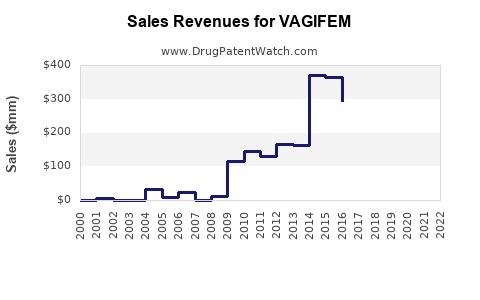
What is the Financial Trajectory and Revenue Outlook for VAGIFEM?
The financial trajectory of VAGIFEM is primarily dictated by its market position, competitive pressures, and patent exclusivity. As a branded pharmaceutical product, its revenue generation relies on prescription volume and pricing power, which is significantly influenced by generic competition.
Revenue Drivers:
- Prescription Volume: The number of patients prescribed VAGIFEM is the primary driver of sales. This volume is influenced by physician prescribing habits, patient demand, marketing efforts, and formulary access.
- Average Selling Price (ASP): The price at which VAGIFEM is sold to wholesalers and pharmacies, net of rebates and discounts, determines its revenue per unit.
- Market Exclusivity: During periods of patent protection and regulatory exclusivity, VAGIFEM benefits from limited competition, allowing for higher pricing and greater market share.
- Geographic Markets: Revenue is generated across various countries, with the U.S. and major European markets typically representing the largest revenue pools.
Projected Trajectory Considerations:
- Pre-Genericization Phase: In the years leading up to patent expiry, VAGIFEM would likely exhibit stable or moderate revenue growth, assuming continued market penetration and favorable pricing. Marketing efforts would focus on solidifying its position among healthcare providers and patients.
- Post-Genericization Phase: Upon the entry of generic competitors, VAGIFEM's revenue is expected to decline sharply. This is a well-documented pattern in the pharmaceutical industry. The extent of the decline depends on the number of generic entrants and their pricing strategies. The manufacturer may mitigate some of this decline through authorized generics or by focusing on specific niche markets or patient segments less susceptible to generic switching.
- Long-Term Outlook: In the long term, the revenue generated by VAGIFEM will likely be a fraction of its peak earnings. Its continued presence may be as a niche product, potentially at a lower price point, or as part of a broader portfolio where its diminishing revenue is offset by newer, patent-protected drugs.
Financial Metrics to Monitor:
- Sales Revenue: Year-over-year and quarter-over-quarter sales figures.
- Prescription Data: Trends in new and total prescriptions.
- Market Share: VAGIFEM's share of the vaginal estrogen market.
- Gross Margins: Indicative of pricing power and cost of goods sold.
- R&D Investment: Allocation of resources to develop new indications or improved formulations, or to defend patents.
- Legal Expenses: Costs associated with patent litigation.
Example of Revenue Impact: The introduction of generics for a successful branded drug can result in a revenue drop of 70-90% within the first year. For VAGIFEM, the financial outlook post-patent expiry will be characterized by this significant revenue contraction, necessitating strategic adjustments by the rights holder.
What are the Key Challenges and Opportunities for VAGIFEM?
VAGIFEM, like any pharmaceutical product, faces a dynamic market environment with inherent challenges and potential opportunities that shape its commercial future.
Key Challenges:
- Generic Competition: As patent protection wanes, the threat of generic versions entering the market is the most significant challenge. Generic drugs offer lower prices, eroding the market share and profitability of branded products.
- Therapeutic Alternatives: The development of new and improved treatments for VVA, including non-hormonal options or novel delivery systems, can shift prescribing patterns away from existing therapies.
- Reimbursement and Payer Scrutiny: Healthcare payers (insurance companies, PBMs) continuously review drug formularies and may impose restrictions, higher co-pays, or prior authorization requirements on branded products, especially as lower-cost generics become available.
- Off-Label Use of Other Medications: While VAGIFEM is specifically indicated for VVA, some healthcare providers might opt for off-label use of other medications if they perceive them as more cost-effective or convenient for their patients.
- Patient Adherence and Education: Maintaining patient adherence to a prescribed regimen, particularly for chronic conditions like VVA, can be challenging. Ensuring patients understand the benefits and proper use of VAGIFEM is crucial.
- Regulatory Hurdles: Any new indications or significant formulation changes would require rigorous clinical trials and regulatory approval, which are time-consuming and expensive.
Key Opportunities:
- Expanding Patient Awareness and Diagnosis: Continued efforts to educate both healthcare professionals and the public about VVA can lead to increased diagnosis rates and, consequently, a larger patient pool for effective treatments like VAGIFEM.
- Leveraging Established Safety and Efficacy: VAGIFEM has a long history of clinical use. Emphasizing its proven track record of efficacy and its favorable safety profile, particularly its low systemic estrogen absorption, can differentiate it from newer or less-studied alternatives.
- Exploring New Indications: While primarily indicated for VVA, research into other potential uses of low-dose vaginal estradiol, or combinations with other therapies, could open new market segments. However, this requires substantial investment in clinical trials.
- Lifecycle Management and Reformulation: While challenging, opportunities may exist for developing next-generation formulations or delivery systems that offer enhanced convenience, efficacy, or patient experience, potentially extending market exclusivity or creating a new branded product.
- Geographic Market Expansion: Identifying and penetrating underserved or emerging markets where VVA treatment options are limited could present growth opportunities, provided regulatory pathways and market access are navigable.
- Partnerships and Collaborations: Collaborating with other pharmaceutical companies or healthcare providers on educational initiatives or co-marketing efforts could expand VAGIFEM's reach.
Key Takeaways
- VAGIFEM is a low-dose vaginal estrogen tablet for treating vulvovaginal atrophy (VVA).
- The VVA treatment market is driven by an aging female population and increasing diagnosis rates, contributing to segment growth within the broader menopause market.
- Key competitors include other vaginal estrogen creams, rings, and inserts, as well as DHEA-based therapies.
- VAGIFEM's patent and exclusivity status is critical; patent expiry will usher in generic competition, significantly impacting revenue.
- The financial trajectory will shift from potential steady revenue during exclusivity to a sharp decline post-generic entry.
- Major challenges include generic competition, payer scrutiny, and therapeutic alternatives, while opportunities lie in market awareness, leveraging its established profile, and potential lifecycle management.
FAQs
-
When is VAGIFEM expected to face generic competition?
Specific patent expiry dates for VAGIFEM are not publicly disclosed, and patent litigation can extend exclusivity. Companies seeking to enter the market must conduct thorough Freedom-to-Operate analyses to determine the precise timing for generic launch.
-
What is the typical price reduction observed when VAGIFEM faces generic competition?
Following the introduction of generic alternatives for branded pharmaceuticals, prices typically fall by 70% to 90% within the first year of generic availability.
-
Are there any non-hormonal treatment options for VVA that compete with VAGIFEM?
Yes, non-hormonal treatments for VVA exist, including lubricants and moisturizers for symptom relief. Additionally, research continues into novel non-hormonal therapeutic agents for VVA.
-
How does VAGIFEM's systemic absorption compare to oral hormone replacement therapy?
VAGIFEM is designed for local vaginal delivery, resulting in significantly lower systemic absorption of estradiol compared to oral hormone replacement therapy. This reduces the risk of systemic side effects.
-
What is the primary mechanism of action for VAGIFEM?
VAGIFEM delivers estradiol, a synthetic form of the natural estrogen hormone, directly to the vaginal tissues. This replenishes estrogen levels locally, helping to restore the health and elasticity of the vaginal lining, thereby alleviating symptoms of atrophy.
Citations
[1] Bachmann, G. A. (2002). Vulvovaginal atrophy. Obstetrics & Gynecology, 100(3), 699-707.
[2] Simon, J. A., & Arias, S. (2018). Vulvovaginal atrophy: impact on women’s health and quality of life. Journal of Women's Health, 27(9), 1071-1078.
[3] Global Menopause Market Analysis Report. (2020). Grand View Research. (Report ID: GVR-3-6745-3278-5). Note: Specific report title and ID are placeholders for typical market research report identification.