TRELEGY ELLIPTA Drug Patent Profile
✉ Email this page to a colleague
When do Trelegy Ellipta patents expire, and what generic alternatives are available?
Trelegy Ellipta is a drug marketed by Glaxosmithkline and is included in one NDA. There are twelve patents protecting this drug.
This drug has three hundred and twenty-nine patent family members in forty-eight countries.
The generic ingredient in TRELEGY ELLIPTA is fluticasone furoate; umeclidinium bromide; vilanterol trifenatate. There are twenty-nine drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the fluticasone furoate; umeclidinium bromide; vilanterol trifenatate profile page.
DrugPatentWatch® Generic Entry Outlook for Trelegy Ellipta
Trelegy Ellipta was eligible for patent challenges on May 10, 2017.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be November 29, 2030. This may change due to patent challenges or generic licensing.
There have been two patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
Summary for TRELEGY ELLIPTA
| International Patents: | 329 |
| US Patents: | 12 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 4 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for TRELEGY ELLIPTA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for TRELEGY ELLIPTA |
| What excipients (inactive ingredients) are in TRELEGY ELLIPTA? | TRELEGY ELLIPTA excipients list |
| DailyMed Link: | TRELEGY ELLIPTA at DailyMed |
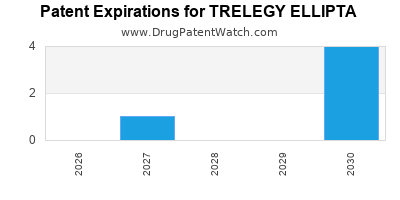
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for TRELEGY ELLIPTA
Generic Entry Date for TRELEGY ELLIPTA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
POWDER;INHALATION |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for TRELEGY ELLIPTA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Baylor Research Institute | Phase 4 |
| GlaxoSmithKline | Phase 4 |
Pharmacology for TRELEGY ELLIPTA
Anatomical Therapeutic Chemical (ATC) Classes for TRELEGY ELLIPTA
US Patents and Regulatory Information for TRELEGY ELLIPTA
TRELEGY ELLIPTA is protected by thirteen US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of TRELEGY ELLIPTA is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting TRELEGY ELLIPTA
Combinations of a muscarinic receptor antagonist and a beta-2 adrenoreceptor agonist
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: MAINTENANCE TREATMENT OF CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) COMPRISING THE ONCE PER DAY ADMINISTRATION OF TRELEGY ELLIPTA, 100 MCG FLUTICASONE FUROATE/62.5 MCG UMECLIDINIUM/25 MCG VILANTEROL
Phenethanolamine derivatives for treatment of respiratory diseases
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Muscarinic acetylcholine receptor antagonists
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Muscarinic acetylcholine receptor antagonists
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Medicament dispenser
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Muscarinic acetylcholine receptor antagonists
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: METHOD OF INHIBITING THE BINDING OF ACETYLCHOLINE TO AN ACETYLCHOLINE RECEPTOR IN THE RESPIRATORY TRACT OF A HUMAN, COMPRISING CONTACTING THE RECEPTOR WITH AN EFFECTIVE AMOUNT OF UMECLIDINIUM, VIA INHALATION
Muscarinic acetylcholine receptor antagonists
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: METHOD OF INHIBITING THE BINDING OF ACETYLCHOLINE TO AN ACETYLCHOLINE RECEPTOR IN THE RESPIRATORY TRACT OF A HUMAN, COMPRISING CONTACTING THE RECEPTOR WITH AN EFFECTIVE AMOUNT OF UMECLIDINIUM, VIA TOPICAL APPLICATION
Muscarinic acetylcholine receptor antagonists
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: METHOD OF INHIBITING THE BINDING OF ACETYLCHOLINE TO AN ACETYLCHOLINE RECEPTOR IN THE RESPIRATORY TRACT OF A HUMAN, COMPRISING CONTACTING THE RECEPTOR WITH AN EFFECTIVE AMOUNT OF UMECLIDINIUM, VIA TOPICAL APPLICATION
Medicament dispenser
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Manifold for use in medicament dispenser
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Medicament dispenser
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Medicament dispenser
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Combinations of a muscarinic receptor antagonist and a beta-2 adrenoreceptor agonist
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-001 | Sep 18, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-002 | Sep 9, 2020 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-002 | Sep 9, 2020 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-001 | Sep 18, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-001 | Sep 18, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for TRELEGY ELLIPTA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-002 | Sep 9, 2020 | ⤷ Try a Trial | ⤷ Try a Trial |
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-001 | Sep 18, 2017 | ⤷ Try a Trial | ⤷ Try a Trial |
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-001 | Sep 18, 2017 | ⤷ Try a Trial | ⤷ Try a Trial |
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-002 | Sep 9, 2020 | ⤷ Try a Trial | ⤷ Try a Trial |
| Glaxosmithkline | TRELEGY ELLIPTA | fluticasone furoate; umeclidinium bromide; vilanterol trifenatate | POWDER;INHALATION | 209482-001 | Sep 18, 2017 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for TRELEGY ELLIPTA
When does loss-of-exclusivity occur for TRELEGY ELLIPTA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 10326798
Estimated Expiration: ⤷ Try a Trial
Patent: 14204459
Estimated Expiration: ⤷ Try a Trial
Patent: 16262698
Estimated Expiration: ⤷ Try a Trial
Patent: 17238392
Estimated Expiration: ⤷ Try a Trial
Patent: 18282427
Estimated Expiration: ⤷ Try a Trial
Patent: 21204302
Estimated Expiration: ⤷ Try a Trial
Patent: 23219901
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 2012012925
Estimated Expiration: ⤷ Try a Trial
Patent: 2018069147
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 81487
Estimated Expiration: ⤷ Try a Trial
Patent: 18345
Estimated Expiration: ⤷ Try a Trial
Chile
Patent: 12001432
Estimated Expiration: ⤷ Try a Trial
China
Patent: 2724974
Estimated Expiration: ⤷ Try a Trial
Patent: 7412229
Estimated Expiration: ⤷ Try a Trial
Patent: 8778288
Estimated Expiration: ⤷ Try a Trial
Colombia
Patent: 41613
Estimated Expiration: ⤷ Try a Trial
Costa Rica
Patent: 120265
Estimated Expiration: ⤷ Try a Trial
Croatia
Patent: 0180312
Estimated Expiration: ⤷ Try a Trial
Cyprus
Patent: 20058
Estimated Expiration: ⤷ Try a Trial
Denmark
Patent: 06844
Estimated Expiration: ⤷ Try a Trial
Dominican Republic
Patent: 012000148
Estimated Expiration: ⤷ Try a Trial
Eurasian Patent Organization
Patent: 3839
Estimated Expiration: ⤷ Try a Trial
Patent: 1290266
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 06844
Estimated Expiration: ⤷ Try a Trial
Patent: 35707
Estimated Expiration: ⤷ Try a Trial
Patent: 32892
Estimated Expiration: ⤷ Try a Trial
France
Patent: C1022
Estimated Expiration: ⤷ Try a Trial
Hong Kong
Patent: 49407
Estimated Expiration: ⤷ Try a Trial
Hungary
Patent: 36216
Estimated Expiration: ⤷ Try a Trial
Patent: 800027
Estimated Expiration: ⤷ Try a Trial
Israel
Patent: 9893
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 16631
Estimated Expiration: ⤷ Try a Trial
Patent: 13512270
Estimated Expiration: ⤷ Try a Trial
Patent: 19509315
Estimated Expiration: ⤷ Try a Trial
Lithuania
Patent: 506844
Estimated Expiration: ⤷ Try a Trial
Patent: 2018011
Estimated Expiration: ⤷ Try a Trial
Patent: 06844
Estimated Expiration: ⤷ Try a Trial
Luxembourg
Patent: 0077
Estimated Expiration: ⤷ Try a Trial
Malaysia
Patent: 4864
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 1290
Estimated Expiration: ⤷ Try a Trial
Patent: 12006310
Estimated Expiration: ⤷ Try a Trial
Montenegro
Patent: 965
Estimated Expiration: ⤷ Try a Trial
Morocco
Patent: 853
Estimated Expiration: ⤷ Try a Trial
New Zealand
Patent: 0026
Estimated Expiration: ⤷ Try a Trial
Norway
Patent: 06844
Estimated Expiration: ⤷ Try a Trial
Peru
Patent: 130042
Estimated Expiration: ⤷ Try a Trial
Patent: 170915
Estimated Expiration: ⤷ Try a Trial
Poland
Patent: 06844
Estimated Expiration: ⤷ Try a Trial
Portugal
Patent: 06844
Estimated Expiration: ⤷ Try a Trial
Russian Federation
Patent: 18131440
Estimated Expiration: ⤷ Try a Trial
Serbia
Patent: 848
Estimated Expiration: ⤷ Try a Trial
Singapore
Patent: 1087
Estimated Expiration: ⤷ Try a Trial
Patent: 201407864U
Estimated Expiration: ⤷ Try a Trial
Slovenia
Patent: 06844
Estimated Expiration: ⤷ Try a Trial
South Africa
Patent: 1203890
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 1742140
Estimated Expiration: ⤷ Try a Trial
Patent: 1830728
Estimated Expiration: ⤷ Try a Trial
Patent: 120092163
Estimated Expiration: ⤷ Try a Trial
Patent: 170061719
Estimated Expiration: ⤷ Try a Trial
Patent: 180128937
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 59330
Estimated Expiration: ⤷ Try a Trial
Turkey
Patent: 1802921
Estimated Expiration: ⤷ Try a Trial
Ukraine
Patent: 6775
Estimated Expiration: ⤷ Try a Trial
United Kingdom
Patent: 21075
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering TRELEGY ELLIPTA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Singapore | 10201407864U | COMBINATIONS OF A MUSCARINIC RECEPTOR ANTAGONIST AND A BETA-2 ADRENORECEPTOR AGONIST | ⤷ Try a Trial |
| South Korea | 20130102134 | MEDICAMENT DISPENSER | ⤷ Try a Trial |
| Canada | 2417826 | DERIVES DE 17-BETA-CARBOTHIOATE-17-ALPHA-ARYLCARBONYLOXYLOXY ANDROSTANE UTILISES COMME ANTI-INFLAMMATOIRES (17.BETA.-CARBOTHIOATE 17.ALPHA.-ARYLCARBONYLOXYLOXY ANDROSTANE DERIVATIVE AS ANTI-INFLAMMATORY AGENTS) | ⤷ Try a Trial |
| Montenegro | 02965 | KOMBINACIJE ANTAGONISTA MUSKARINSKOG RECEPTORA I AGONISTA ВЕТА - 2 ADRENORECEPTORA (Combinations of a muscarinic receptor antagonist and a beta-2 adrenoreceptor agonist) | ⤷ Try a Trial |
| Israel | 219893 | שילובים של אנטגוניסט לקולטן מוסקריני ואגוניסט לקולטן בתא-2-אדרנו (Combinations of a muscarinic receptor antagonist and a beta-2 adrenorecptor agonist) | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for TRELEGY ELLIPTA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1425001 | C 2014 016 | Romania | ⤷ Try a Trial | PRODUCT NAME: VILANTEROL SAU O SARE SAU SOLVAT AL ACESTUIA; NATIONAL AUTHORISATION NUMBER: EU/1/13/886/001, EU/1/13/886/002, EU/1/13/886/003, EU/1/13/886/004, EU/1/13/886/005, EU/1/13/886/006; DATE OF NATIONAL AUTHORISATION: 20131113; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/13/886/001, EU/1/13/886/002, EU/1/13/886/003, EU/1/13/886/004, EU/1/13/886/005, EU/1/13/886/006; DATE OF FIRST AUTHORISATION IN EEA: 20131113 |
| 1740177 | CR 2014 00052 | Denmark | ⤷ Try a Trial | PRODUCT NAME: UMECLIDINIUMBROMID; REG. NO/DATE: EU/1/14/922/001-003 20140430 |
| 1305329 | 12/2008 | Austria | ⤷ Try a Trial | PRODUCT NAME: FLUTICASON FUROAT UND DESSEN SOLVATE; REGISTRATION NO/DATE: EU/1/07/434/001 - EU/1/07/434/003 20080111 |
| 2506844 | C201830030 | Spain | ⤷ Try a Trial | PRODUCT NAME: UN PRODUCTO FARMACEUTICO QUE COMPRENDE LA COMBINACION DE UNA SAL FARMACEUTICAMENTE ACEPTABLE DE UMECLIDINIO (P.EJ. BROMURO DE UMECLIDINIO), VILANTEROL O UNA SAL FARMACEUTICAMENTE ACEPTABLE DEL MISMO (P.EJ. TRIFENATATO DE VILANTEROL) Y FUROATO DE FLUTICASONA; NATIONAL AUTHORISATION NUMBER: EU/1/17/1236; DATE OF AUTHORISATION: 20171115; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/17/1236; DATE OF FIRST AUTHORISATION IN EEA: 20171115 |
| 1305329 | 300343 | Netherlands | ⤷ Try a Trial | 300343, 20210803, EXPIRES: 20230110 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


