Last updated: April 24, 2026
How large is the testosterone drug market and where is demand coming from?
Testosterone is a mature, globally traded androgen therapy with demand driven by:
- Aging demographics in key markets (US, EU, Japan), increasing prevalence of male hypogonadism and clinician use of replacement therapy.
- Indication expansion in practice patterns, including men with late-onset hypogonadism (LOH) and off-label use in select settings.
- Shift between formulations as payer coverage and safety/tolerability profiles influence prescribing (injectables, gels, patches, buccal/topical systems, and longer-acting depot products).
Key market structure facts
- Testosterone is a high-volume, price-regulated-to-competitive category in many geographies.
- Generics and authorized generics are common, especially for older depot and topical molecules, compressing price and margins.
- Brand differentiation typically comes from device/formulation, dosing convenience, and clinical/safety positioning rather than new molecular entities.
What are the major sub-segments by formulation and why do they matter economically?
Formulation mix strongly influences revenue because it drives:
- Net price (topicals often face more aggressive generic competition than certain branded devices depending on geography and coverage)
- Adherence and persistence (long-acting injectables can reduce clinic-administered dosing friction, but can face substitution and contracting barriers)
- Distribution economics (topicals require consistent retail pharmacy supply and payer prior authorization patterns; injectables depend on medical channel reimbursement cadence)
Formulation landscape (commercially relevant buckets)
| Sub-segment |
Typical route |
Revenue drivers |
Cost pressures |
| Testosterone gels/creams |
Topical |
Ease of home use; ongoing refills |
Generic substitution and payer step edits |
| Long-acting injectables |
IM depot |
Dosing interval convenience; clinic-administered |
Contracting; biosimilar-style competition is less relevant but generics exist |
| Short-acting injectables |
IM |
Low acquisition cost in many markets |
Margin compression from generic pricing |
| Patches or oral/buccal systems |
Transdermal/oral |
Convenience and steadier PK positioning in some settings |
Lower scale if patient preference shifts |
What market dynamics are currently shaping pricing and volumes?
Testosterone’s financial trajectory is shaped less by scientific novelty and more by competitive pricing, payer behavior, and brand-to-generic churn.
1) Patent expiry and generic entry cycles
- Testosterone has multiple legacy products across multiple dosage forms, creating staggered competitive entry waves.
- When a widely used formulation enters generic competition, the result is typically a mix shift toward lower-cost options and a fast erosion of brand net price.
2) Payer restrictions and prior authorization
- Many payers manage spend via:
- Step therapy or formulary tiers
- Documentation requirements for hypogonadism diagnosis
- Quantity limits by dosing regimen
- These controls reduce incremental volume growth and force manufacturers to compete for formulary placement.
3) Safety and monitoring narratives
- Clinician adoption depends on ongoing guideline-driven monitoring (hematocrit, prostate surveillance practices where applicable).
- Any label refinement or safety communication can affect initiation rates or persistence, but the dominant economic impact still comes from competitive pricing and coverage.
4) Supply and distribution constraints
- Like other widely used generics and brand products, production capacity, controlled-substance style handling, and distribution availability can move near-term unit sales.
- Economic effect: short-term volume disruptions, followed by catch-up sales that may not fully restore lost net price.
How does testosterone’s competitive landscape affect financial trajectory?
Testosterone generally trades as a defensive, high-volume category with thin-to-moderate margins once generics gain share. The key determinant of financial trajectory for any specific product is:
- Whether it is exposed to generic substitution
- Whether it has device/formulation characteristics that retain payer and prescriber preference
- Contracting strength in major purchasing channels
- Residual exclusivity for a given dosage form (if any)
Revenue math that governs outcomes
In practice, testosterone revenue outcomes follow this pattern:
- Unit growth is possible when awareness and diagnosis rates rise, but
- Net revenue growth usually slows or turns negative when generic penetration accelerates,
- unless a branded formulation maintains formulary access and resists substitution long enough to defend unit share through contracts.
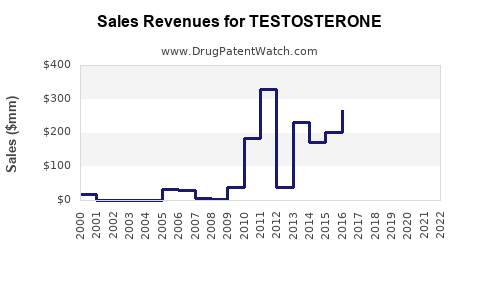
What do historical financial patterns typically look like for testosterone products?
Across mature hormone replacement categories, companies generally experience:
- Peak brand phase before generic entry
- Gradual share erosion tied to increasing formulary adoption of generics/authorized generics
- Price compression once multiple substitutes compete
- Consolidation by formulation where the lowest-cost options become preferred unless clinical differentiators dominate
For business planning, the forecast question becomes:
- Do new or defended formulation cycles offset generic headwinds?
- Or do share and price both trend down over the medium term?
What financial trajectory signals matter for investors and R&D planners?
For testosterone exposure, the most actionable signals are:
- Share vs. price decomposition
- If revenue falls, check whether the decline is mostly unit loss or net price erosion.
- Formulary and contract outcomes
- A single large payer contract can change net price and persistence.
- Launch timing of competing formulations
- Multiple overlapping dosage-form contests increase rapid substitution risk.
- Lifecycle strategy
- Companies that can extend competitiveness by improving adherence and minimizing switching typically see flatter revenue curves.
How do regulatory and reimbursement forces intersect with commercial performance?
- Regulatory alignment drives what can be marketed and how physicians perceive appropriateness of prescribing.
- Reimbursement and coverage policy determines what gets prescribed at scale.
- The economic impact is often immediate: payer edits can reduce volume within months, independent of clinical demand.
What is the likely medium-term outlook for testosterone revenues?
The medium-term outlook remains mature:
- Demand continues to be supported by baseline population need and diagnosis awareness.
- Revenue growth is constrained by structural price competition from generic and authorized generic availability.
- Products that defend formulary position and minimize switching can maintain more stable revenues, but category-wide growth will lag inflation.
From a financial trajectory standpoint, the category tends to show:
- Volatility at formulation level around competition entry,
- Gradual normalization of net prices after substitution stabilizes.
Where can value concentrate: branded durability vs. volume defense
Testosterone’s economics can concentrate in two ways:
- Branded durability through entrenched contracts and patient adherence benefits that reduce switching.
- Volume defense in markets where payer policies are less restrictive or where clinicians prefer specific dosing modalities.
Value map by business model
| Strategy |
What it does to revenue |
What it does to margin |
| Defend formulary position for a branded formulation |
Slows share loss |
Keeps net price higher for longer |
| Compete with authorized generics or low-cost equivalents |
Stabilizes units |
Compresses net price but can preserve cash flow |
| Shift toward longer-acting or adherence-optimized devices |
Improves persistence |
Can protect price if payers accept differentiators |
Key Takeaways
- Testosterone demand is steady but revenue growth is structurally constrained by generic and authorized generic competition across multiple dosage forms.
- The category’s financial trajectory is driven mainly by formulation mix, payer coverage rules, and timing of competitive entry, not by fundamental new clinical breakthroughs.
- For any testosterone exposure, the most predictive performance drivers are share vs. price decomposition and formulary/contract durability.
- Medium-term category revenues are likely to be mature with episodic volatility, with branded products holding up only where they sustain payer access and limit switching.
FAQs
1) Why does testosterone pricing compress even when demand is stable?
Generic and authorized generic entry increases substitute availability, forcing net price declines through payer contracting and pharmacy switching.
2) Which testosterone formulation tends to face the most rapid net price erosion?
Topical regimens and legacy formulations that have multiple generic alternatives and broad substitution pathways typically face faster price compression.
3) What payer policies most affect testosterone revenue?
Prior authorization, step therapy, documentation requirements, and quantity limits reduce initiation and persistence for patients without qualifying diagnoses.
4) What is the biggest determinant of a branded testosterone product’s revenue trajectory?
The product’s ability to maintain formulary placement and reduce switching after competing products enter.
5) Is testosterone a good example of a mature pharma market?
Yes: it shows high-volume demand, layered formulation competition, and revenue patterns dominated by lifecycle and reimbursement rather than breakthrough science.
References
[1] FDA. Drug Approval Reports and labeling database for testosterone-containing products. U.S. Food & Drug Administration. https://www.fda.gov/drugs/drug-approvals-and-databases
[2] National Library of Medicine (NLM). PubMed search results and reviews related to testosterone therapies and hypogonadism. https://pubmed.ncbi.nlm.nih.gov/
[3] Endocrine Society. Clinical practice guidance on testosterone therapy and monitoring (hypogonadism). https://www.endocrine.org/clinical-practice-guidelines