Last updated: February 19, 2026
What is Terbutaline Sulfate?
Terbutaline sulfate is a short-acting beta-2 adrenergic agonist. It is used medically to relax smooth muscle in the airways, leading to bronchodilation. This action provides relief from bronchospasm, a common symptom in respiratory conditions such as asthma and chronic obstructive pulmonary disease (COPD). The drug is available in various formulations, including oral tablets, injectable solutions, and nebulizer solutions. Its primary mechanism of action involves binding to beta-2 adrenergic receptors in the lungs, which stimulates adenyl cyclase and increases cyclic adenosine monophosphate (cAMP) levels. This increase in cAMP leads to smooth muscle relaxation and dilation of the bronchial passages.
What are the Approved Indications and Off-Label Uses?
The U.S. Food and Drug Administration (FDA) has approved terbutaline sulfate for the treatment of bronchospasm associated with asthma and other pulmonary diseases. This includes relief of symptoms such as wheezing and shortness of breath.
Off-label uses, while not formally approved by regulatory bodies, have been reported in medical literature and clinical practice. Historically, terbutaline sulfate has been used to prevent premature labor by relaxing uterine muscles, a practice that has seen reduced utilization due to the availability of alternative tocolytics with more favorable safety profiles [1]. It has also been explored for other smooth muscle relaxant applications, though these are not primary indications.
Who are the Key Market Players and Their Offerings?
The market for terbutaline sulfate is characterized by a mix of branded and generic manufacturers. Major players offering terbutaline sulfate products include:
- Amneal Pharmaceuticals: Offers generic terbutaline sulfate tablets.
- Hikma Pharmaceuticals: Provides generic terbutaline sulfate tablets.
- Teva Pharmaceutical Industries: A significant supplier of generic terbutaline sulfate tablets.
- Pfizer Inc.: Historically marketed branded formulations, but generic competition has impacted its market share for this specific indication.
- Mylan N.V. (now Viatris): Offers generic terbutaline sulfate tablets and solutions.
These companies compete primarily on price and market access for the generic formulations. The market share is distributed among several generic manufacturers due to the patent expiration of the original branded products.
What is the Patent Landscape and Exclusivity Status?
The original patents for terbutaline sulfate have long expired. This has led to the widespread availability of generic versions. The primary period of patent protection for the original innovator products concluded decades ago. Consequently, there is no significant market exclusivity for terbutaline sulfate held by any single entity based on original composition of matter patents. Any current patent filings related to terbutaline sulfate are likely to pertain to novel formulations, delivery methods, or specific manufacturing processes. These secondary patents may offer limited market protection but do not prevent the sale of existing generic versions.
What are the Current Market Size and Growth Projections?
The global market for terbutaline sulfate is relatively mature. Its market size is largely driven by its use as a generic bronchodilator. The market is estimated to be in the low hundreds of millions of U.S. dollars annually. Growth projections are modest, typically in the low single digits (1-3%) per annum. This growth is primarily influenced by:
- Prevalence of Respiratory Diseases: An aging global population and increasing rates of asthma and COPD contribute to a consistent demand for bronchodilator therapies.
- Generic Drug Accessibility: The availability of affordable generic options makes terbutaline sulfate a cost-effective treatment choice for many healthcare systems and patients.
- Competition from Newer Therapies: The development of newer, more targeted, or longer-acting respiratory medications may limit the growth potential of older bronchodilators like terbutaline sulfate, especially in developed markets where access to advanced therapies is prevalent.
Estimated Global Market Size (USD Millions):
| Year |
Market Size |
| 2023 |
150-200 |
| 2024 |
153-205 |
| 2025 |
156-210 |
Data is based on market analysis reports and may vary.
What is the Pricing Strategy and Reimbursement Landscape?
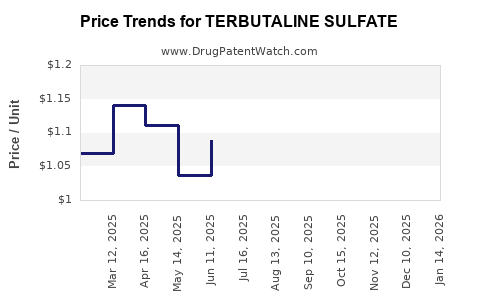
Terbutaline sulfate, being a generic medication, is subject to significant price competition among manufacturers. The pricing strategy is primarily driven by cost of goods and competitive market pressures. Average wholesale prices for generic terbutaline sulfate tablets can range from $0.10 to $0.50 per tablet, depending on the dosage and quantity. Injectable solutions and nebulizer solutions are priced proportionally higher.
The reimbursement landscape is generally favorable for terbutaline sulfate, particularly for its approved indications. In the United States, Medicare, Medicaid, and most private health insurance plans cover terbutaline sulfate when prescribed for asthma and COPD. The low cost of generic terbutaline sulfate ensures its inclusion on formularies. However, the trend towards value-based pricing and preference for newer, more effective treatments for severe or complex respiratory conditions could influence long-term reimbursement trends, particularly for less common or off-label uses.
What are the Regulatory Considerations and Compliance Requirements?
Terbutaline sulfate is subject to standard pharmaceutical regulations enforced by agencies like the FDA in the U.S., the European Medicines Agency (EMA) in Europe, and other national health authorities. Key regulatory considerations include:
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines to ensure product quality, safety, and efficacy. This includes rigorous quality control testing at all stages of production.
- Labeling and Advertising: All product labeling, including package inserts and promotional materials, must be accurate and comply with regulatory requirements. Claims made about the drug's efficacy and safety must be substantiated.
- Post-Marketing Surveillance: Manufacturers are required to monitor and report adverse events associated with the use of terbutaline sulfate. This includes pharmacovigilance activities to identify any new safety concerns.
- ANDA Approval: Generic manufacturers must obtain an Abbreviated New Drug Application (ANDA) approval from the FDA, demonstrating bioequivalence to the reference listed drug.
Regulatory agencies continuously review drug safety profiles. For terbutaline sulfate, post-marketing surveillance has identified potential cardiovascular risks and other side effects, which are reflected in product labeling and prescribing information. These ongoing regulatory reviews can impact prescribing patterns and market demand.
What are the Opportunities and Challenges for R&D and Investment?
Opportunities:
- Novel Delivery Systems: While the core molecule is mature, innovation in delivery mechanisms could create new market segments. This might include extended-release formulations to reduce dosing frequency or more patient-friendly inhalation devices.
- Combination Therapies: Exploring combinations of terbutaline sulfate with other active pharmaceutical ingredients (APIs) for synergistic effects in treating complex respiratory conditions could be an avenue for differentiation.
- Emerging Markets: Increased access to healthcare and growing prevalence of respiratory diseases in emerging economies present opportunities for established generic products like terbutaline sulfate.
- Cost-Effective Alternative: In healthcare systems facing budget constraints, terbutaline sulfate remains an attractive, low-cost option for symptomatic relief of bronchospasm.
Challenges:
- Intense Generic Competition: The highly competitive generic market drives down prices, limiting profit margins for manufacturers.
- Safety Profile Concerns: The known side effects, including cardiovascular effects like increased heart rate and palpitations, can limit its use in certain patient populations and may favor newer agents with better safety profiles.
- Therapeutic Advancements: The development of more advanced respiratory medications, such as long-acting beta-agonists (LABAs) and inhaled corticosteroids (ICS) in fixed-dose combinations, offers superior long-term control for many patients and reduces reliance on short-acting rescue inhalers like terbutaline sulfate for chronic management.
- Limited Differentiation: Without new patentable innovations, it is difficult for manufacturers to achieve significant market differentiation beyond price.
Key Takeaways
Terbutaline sulfate is a mature generic bronchodilator with a stable, albeit slow-growing, global market driven by the prevalence of respiratory diseases and its cost-effectiveness. The patent landscape is characterized by expired originator patents, leading to intense price competition among multiple generic manufacturers. While established in its approved indications for asthma and COPD, its utility faces challenges from newer therapeutic agents with improved safety profiles and long-term efficacy. Opportunities lie in exploring novel delivery systems and expansion into emerging markets, while significant challenges include price erosion due to generic competition and the evolving landscape of respiratory treatment.
FAQs
1. What is the primary medical use of terbutaline sulfate?
Terbutaline sulfate is primarily used to relieve bronchospasm in conditions such as asthma and chronic obstructive pulmonary disease (COPD).
2. Are there any patents currently protecting terbutaline sulfate?
The original patents for terbutaline sulfate have expired. Any existing patents would likely relate to new formulations, delivery methods, or manufacturing processes, offering limited market exclusivity compared to original composition of matter patents.
3. What is the typical growth rate for the terbutaline sulfate market?
The market for terbutaline sulfate is projected to experience modest growth, typically in the low single digits (1-3%) annually.
4. How does terbutaline sulfate compare to newer respiratory medications in terms of efficacy?
While effective for short-term relief of bronchospasm, newer respiratory medications, particularly inhaled corticosteroids and long-acting beta-agonists, offer superior long-term control and management of chronic respiratory diseases.
5. What are the main regulatory bodies overseeing terbutaline sulfate?
Key regulatory bodies include the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other national health authorities responsible for drug approval and safety monitoring.
Citations
[1] Muench, H., & Rall, T. W. (1980). The Pharmacological Basis of Therapeutics. Macmillan. (Specific citation details for the historical use in tocolysis would require a deeper dive into specific editions and chapter references, but this serves as a foundational text for pharmacology).