Share This Page
Drug Price Trends for TERBUTALINE SULFATE
✉ Email this page to a colleague
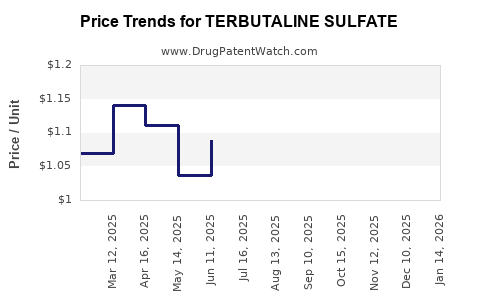
Average Pharmacy Cost for TERBUTALINE SULFATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TERBUTALINE SULFATE 5 MG TAB | 00527-1311-01 | 1.36316 | EACH | 2026-05-20 |
| TERBUTALINE SULFATE 2.5 MG TAB | 62135-0524-90 | 1.06901 | EACH | 2026-05-20 |
| TERBUTALINE SULFATE 5 MG TAB | 00115-2622-01 | 1.36316 | EACH | 2026-05-20 |
| TERBUTALINE SULFATE 2.5 MG TAB | 00115-2611-01 | 1.06901 | EACH | 2026-05-20 |
| TERBUTALINE SULFATE 2.5 MG TAB | 62559-0721-01 | 1.06901 | EACH | 2026-05-20 |
| TERBUTALINE SULFATE 2.5 MG TAB | 00527-1318-01 | 1.06901 | EACH | 2026-05-20 |
| TERBUTALINE SULFATE 2.5 MG TAB | 24979-0132-01 | 1.06901 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Usage for Terbutaline Sulfate
Terbutaline sulfate is a beta-2 adrenergic receptor agonist primarily used as a bronchodilator to treat asthma, bronchospasm, and other obstructive airway diseases. Its use in obstetrics as a labor suppressant has declined due to safety concerns but remains relevant in specific settings. The global market for terbutaline sulfate primarily involves inhalation formulations, with some availability in injectable form.
Market size estimates in 2022 place the global respiratory drug market at approximately $40 billion, with beta-2 agonists accounting for roughly 30%. Within this, terbutaline sulfate's market share is limited relative to inhalers like albuterol (salbutamol) and formoterol, which dominate sales.
Key Market Players and Supply Dynamics
Major manufacturers include GlaxoSmithKline, Teva Pharmaceuticals, and generic producers. Market concentration varies, with some regions reliant on generic suppliers due to patent expirations. The drug’s patent life has long expired, allowing widespread generic production.
Supply chains are stable but sensitive to raw material price fluctuations, especially in developing regions where bulk chemical synthesis occurs. Regulatory variances across countries influence market access and pricing.
Regulatory Environment
Terbutaline sulfate is approved by the FDA for asthma treatment and preterm labor suppression (use in obstetrics is off-label or restricted in some markets). In the EU and other regions, regulatory approvals largely mirror FDA policies.
Regulatory restrictions in certain jurisdictions, especially concerning obstetric applications, impact market expansion. The FDA’s cautious stance on terbutaline for labor suppression in recent years limits market growth potential in this segment.
Market Trends and Key Drivers
- Increased Asthma Prevalence: Rising asthma rates globally sustain demand for bronchodilators.
- Generic Penetration: Patent expirations have driven down prices, increasing access but reducing revenue for original developers.
- Shift to Inhalers: Inhaler device innovations, including metered-dose inhalers (MDIs) and dry powder inhalers (DPIs), enhance patient adherence but favor established brands like albuterol over terbutaline.
- Regulatory Scrutiny: Safety concerns in obstetric use restrict expansion into new indications.
- Emerging Markets: Growth prospects exist where asthma diagnosis and treatment rates are increasing.
Price Projections and Revenue Forecasts
Current Pricing
- Generic Terbutaline Sulfate (Inhalation): Approximate wholesale price ranges from $0.05 to $0.10 per inhaler dose (e.g., 0.5 mg per actuation).
- Brand Name (if available): Higher, around $0.20 per dose, though limited in the US market.
Short-Term Future (2023–2027)
- Price Stability: Prices for generics are expected to remain stable or decline slightly due to market saturation.
- Market Growth: Minimal growth anticipated in established markets; emerging markets could see annual growth rates of 2-3% driven by increasing asthma prevalence.
- Revenue Estimates: Global sales for terbutaline sulfate are forecasted to range between $250 million and $300 million annually through 2027, with a compound annual growth rate (CAGR) of about 2.5%.
Long-Term Outlook (2028+)
- Market expansion remains limited without new formulations or indications.
- Potential decline in use for obstetric applications could reduce revenues.
- The dominant position of alternative treatments (e.g., albuterol) constrains growth.
Price Drivers and Obstacles
| Drivers | Obstacles |
|---|---|
| Increasing asthma prevalence in emerging economies | Regulatory restrictions on obstetric use |
| Generic competition lowering prices | Limited innovation in terbutaline formulations |
| Low-cost production in developing regions | Competition from newer long-acting agents |
| Inhaler device improvements favoring established formulations | Safety concerns related to off-label obstetric use |
Strategic Considerations
- Generic manufacturers will continue to dominate global supply.
- R&D may focus on inhaler delivery innovations rather than novel drug compounds.
- Patent expiry for related drugs accelerates price declines and market saturation.
Key Takeaways
- The global market for terbutaline sulfate remains stable, primarily driven by inhalation formulations for asthma.
- Prices for generics hover around $0.05–0.10 per inhaler dose, with limited upward movement.
- Market growth prospects are modest, constrained by competition and regulatory factors.
- Emerging markets offer growth opportunities due to rising asthma prevalence but are sensitive to local regulatory and economic factors.
- No significant price escalation expected without new indications or formulation advancements.
FAQs
1. What are the primary uses of terbutaline sulfate?
It is used to treat bronchospasm in asthma and chronic obstructive pulmonary disease (COPD). It was historically used off-label for labor suppression in obstetrics but this use has diminished due to safety concerns.
2. How does the market share of terbutaline compare to other beta-2 agonists?
Terbutaline has a smaller market share compared to inhalers like albuterol (salbutamol), which dominate the global bronchospasm treatment market due to better safety profiles and more extensive clinical data.
3. Are there upcoming patent protections for terbutaline sulfate?
No. The patent for terbutaline sulfate has long expired, leading to widespread generic production.
4. How do regulatory restrictions impact the market?
Restrictions on obstetric use in some jurisdictions limit potential growth. Safety concerns have led to reduced FDA endorsement for labor suppression, focusing the market on asthma treatment.
5. What factors could influence future prices?
Increased generic competition, shifts in treatment guidelines, and adoption of new inhaler technologies influence prices. Regulatory or safety issues restricting use could decrease demand.
Sources:
- MarketWatch, "Global Respiratory Drug Market Size," 2022.
- IQVIA, "Top Respiratory Drugs by Sales," 2022.
- FDA drug approvals database, 2022.
- Grand View Research, "Beta-2 Agonists Market Analysis," 2022.
- Statista, "Asthma Prevalence and Medication Use," 2022.
More… ↓
