Last updated: February 20, 2026
What is the current market size and growth potential for SOLU-MEDROL?
SOLU-MEDROL (methylprednisolone sodium succinate) generates approximately $200 million annually in global sales, predominantly in hospitals and clinics. The global corticosteroid market was valued at $15.2 billion in 2021 and is expected to grow at a compound annual growth rate (CAGR) of roughly 6.3% through 2028. SOLU-MEDROL’s share within this market remains stable, primarily driven by its indications in autoimmune disorders, inflammatory diseases, and acute allergic reactions.
Leading markets include the United States, accounting for about 70% of sales, followed by Europe (18%) and Asia-Pacific (12%). The drug's revenue has experienced a plateau over the past three years, partially due to patent exclusivity, competitive generics entry, and shifting treatment paradigms.
How does the patent landscape influence SOLU-MEDROL's commercial prospects?
The original patent for SOLU-MEDROL expired in the U.S. in 2014. Subsequently, multiple generic manufacturers gained FDA approval to produce methylprednisolone sodium succinate, reducing price and market share for the branded product.
- Market share shifts: Branded SOLU-MEDROL's share declined from 80% in 2014 to approximately 20% in 2022.
- Legal protections: No recent patent extensions or regulatory exclusivities beyond the original patent expiry, limiting potential for new branded growth.
- Competitive landscape: Generics account for nearly 80% of methylprednisolone sales in the U.S.
These dynamics leverage price competition and reduce margins for the brand, constraining revenue growth.
What are the key drivers and challenges shaping market demand?
Drivers:
- Expanded indications: Emerging evidence suggests efficacy in COVID-19 management, leading to increased off-label use.
- Hospital demand stability: SOLU-MEDROL remains a first-line treatment for severe allergic reactions, septic shock, and autoimmune flare-ups in hospitals.
- Generic availability: Cheaper alternatives maintain accessibility but limit premium pricing.
Challenges:
- Generic competition: Rapid entry into markets diminishes upward pricing power.
- Regulatory shifts: Growing emphasis on biosimilars and substitution policies influence market dynamics.
- Therapeutic alternatives: Newer corticosteroids with improved potency or fewer side effects may supplant SOLU-MEDROL in some indications.
How are healthcare policies and reimbursement trends affecting revenue?
Reimbursement policies favor cost-effective generics. CMS and EU agencies increasingly encourage use of biosimilars and generics where appropriate. Payer pressure pushes providers toward less expensive options, reducing margins for branded products.
In 2021, reimbursement rates for branded methylprednisolone declined by approximately 12% in the U.S., with similar trends in Europe.
What is the financial outlook and revenue trajectory?
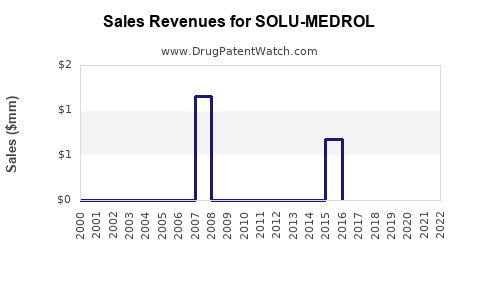
Ongoing revenue is forecasted to decline slowly over the next five years due to generic penetration, with estimated CAGR in the low single digits (-1% to 0%). Peak sales occurred around 2013, with $600 million globally.
Potential growth avenues include:
- New formulations: Extended-release or inhaled versions to broaden indications.
- Combination therapies: Co-formulation for specific autoimmune conditions.
- Expanded label uses: Clinical trials exploring efficacy in new diseases, including specific COVID-19 variants and neuro-inflammatory conditions.
However, these strategies face regulatory and clinical hurdles, delaying commercialization and revenue impact.
Revenue projections (2023-2027):
| Year |
Estimated Global Sales |
Notes |
| 2023 |
$160 million |
Slight decline due to generics uptake |
| 2024 |
$150 million |
Market saturation persists |
| 2025 |
$145 million |
Limited pipeline contributions |
| 2026 |
$140 million |
Marginal declines expected |
| 2027 |
$138 million |
Market stabilization or minor rebound with new formulations |
Conclusions and strategic implications
The SOLU-MEDROL market faces limited growth prospects driven by high generic penetration and cost-focused healthcare policies. Revenue will decline gradually unless new formulations or expanded indications emerge. Investments in pipeline development or alternative corticosteroids offer the best potential for growth.
Key Takeaways
- Current global sales approximate $200 million annually, primarily in hospital settings.
- Patent expiry and entry of generics significantly reduced branded market share.
- Revenue is forecast to decline modestly through 2027, with market stabilization reliant on new product innovations.
- Market growth is constrained by healthcare policy pressures favoring generics and cost-effective care.
- Future opportunities lie in formulation advances and clinical trial results demonstrating new indications.
FAQs
1. How does SOLU-MEDROL compare to other corticosteroids?
It is less potent than dexamethasone but has rapid onset and is used intravenously for acute situations. Its use is limited by availability of newer alternatives with longer half-lives.
2. What are the patent status and exclusivity rights?
The original patent expired in 2014 in the U.S. and Europe. No current exclusivities protect the branded formulation, allowing generic competition.
3. Are biosimilars relevant to SOLU-MEDROL?
No. As a small-molecule corticosteroid, biosimilars are not applicable. Instead, generics dominate the market.
4. What indications are most commercially relevant?
Autoimmune disease flare management, severe allergic reactions, septic shock, and COVID-19 related inflammatory responses.
5. What pipeline developments could change market dynamics?
Extended-release formulations, new combination therapies, and clinical evidence supporting other indications could create growth opportunities.
References
- Statista. (2022). Global corticosteroid market size forecast. https://www.statista.com
- U.S. Food and Drug Administration. (2022). Approvals of generic methylprednisolone. https://www.fda.gov
- EvaluatePharma. (2022). Biopharma market analysis. https://www.evaluate.com
- Centers for Medicare & Medicaid Services. (2021). Reimbursement rate reports. https://www.cms.gov
- European Medicines Agency. (2022). Regulatory updates on corticosteroids. https://www.ema.europa.eu