SELZENTRY Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Selzentry, and when can generic versions of Selzentry launch?
Selzentry is a drug marketed by Viiv Hlthcare and is included in two NDAs.
The generic ingredient in SELZENTRY is maraviroc. There are two drug master file entries for this compound. Five suppliers are listed for this compound. Additional details are available on the maraviroc profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Selzentry
A generic version of SELZENTRY was approved as maraviroc by HETERO LABS LTD III on February 7th, 2022.
Summary for SELZENTRY
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 91 |
| Clinical Trials: | 43 |
| Patent Applications: | 5,576 |
| Formulation / Manufacturing: | see details |
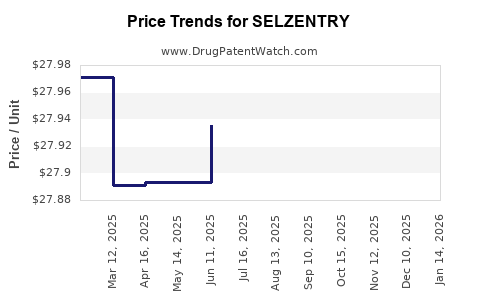
| Drug Prices: | Drug price information for SELZENTRY |
| What excipients (inactive ingredients) are in SELZENTRY? | SELZENTRY excipients list |
| DailyMed Link: | SELZENTRY at DailyMed |
Recent Clinical Trials for SELZENTRY
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Hospital Clínic de Barcelona | Phase 2 |
| Hospital Universitario Infanta Leonor | Phase 2 |
| Hospital Clinic of Barcelona | Phase 2 |
Pharmacology for SELZENTRY
| Drug Class | CCR5 Co-receptor Antagonist |
| Mechanism of Action | Chemokine Co-receptor 5 Antagonists |
Anatomical Therapeutic Chemical (ATC) Classes for SELZENTRY
US Patents and Regulatory Information for SELZENTRY
SELZENTRY is protected by zero US patents and two FDA Regulatory Exclusivities.
FDA Regulatory Exclusivity protecting SELZENTRY
NEW PATIENT POPULATION
Exclusivity Expiration: ⤷ Try a Trial
PEDIATRIC EXCLUSIVITY
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Viiv Hlthcare | SELZENTRY | maraviroc | SOLUTION;ORAL | 208984-001 | Nov 4, 2016 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Viiv Hlthcare | SELZENTRY | maraviroc | TABLET;ORAL | 022128-004 | Nov 4, 2016 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Viiv Hlthcare | SELZENTRY | maraviroc | SOLUTION;ORAL | 208984-001 | Nov 4, 2016 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Viiv Hlthcare | SELZENTRY | maraviroc | TABLET;ORAL | 022128-003 | Nov 4, 2016 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Viiv Hlthcare | SELZENTRY | maraviroc | TABLET;ORAL | 022128-002 | Aug 6, 2007 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Viiv Hlthcare | SELZENTRY | maraviroc | TABLET;ORAL | 022128-001 | Aug 6, 2007 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for SELZENTRY
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Viiv Hlthcare | SELZENTRY | maraviroc | TABLET;ORAL | 022128-003 | Nov 4, 2016 | ⤷ Try a Trial | ⤷ Try a Trial |
| Viiv Hlthcare | SELZENTRY | maraviroc | SOLUTION;ORAL | 208984-001 | Nov 4, 2016 | ⤷ Try a Trial | ⤷ Try a Trial |
| Viiv Hlthcare | SELZENTRY | maraviroc | TABLET;ORAL | 022128-002 | Aug 6, 2007 | ⤷ Try a Trial | ⤷ Try a Trial |
| Viiv Hlthcare | SELZENTRY | maraviroc | TABLET;ORAL | 022128-004 | Nov 4, 2016 | ⤷ Try a Trial | ⤷ Try a Trial |
| Viiv Hlthcare | SELZENTRY | maraviroc | TABLET;ORAL | 022128-001 | Aug 6, 2007 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for SELZENTRY
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| ViiV Healthcare B.V. | Celsentri | maraviroc | EMEA/H/C/000811 Celsentri, in combination with other antiretroviral medicinal products, is indicated for treatment experienced adults, adolescents and children of 2 years of age and older and weighing at least 10 kg infected with only CCR5-tropic HIV-1 detectable, |
Authorised | no | no | no | 2007-09-18 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for SELZENTRY
See the table below for patents covering SELZENTRY around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Eurasian Patent Organization | 200100516 | ⤷ Try a Trial | |
| Israel | 143512 | אזאביציקלואלקאנים, תכשירים רוקחיים הכוללים אותם ושימושם בייצור תרופות למניעה או טיפול בהדבקה על ידי נגיף הכשל החיסוני (Azabicycloalkanes, pharmaceutical compositions comprising same and use thereof for the manufacture of medicaments for the prevention or treatment of infection by immunodeficiency virus) | ⤷ Try a Trial |
| South Korea | 20010089701 | ⤷ Try a Trial | |
| Brazil | 9905977 | ⤷ Try a Trial | |
| Serbia | 51436 | DERIVATI TRIAZOLIL TROPANA KAO MODULATORI CCR5 (TRIAZOL TROPANE DERIVATIVES AS CCR5 MOUDLATORS) | ⤷ Try a Trial |
| European Patent Office | 1013276 | ⤷ Try a Trial | |
| Mexico | PA02011631 | DERIVADOS DE TROPANO UTILES EN TERAPIA. (TRYASOLYL TROPANE DERIVATIVES AS CCR5 MODULATORS.) | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for SELZENTRY
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1284974 | 6/2008 | Austria | ⤷ Try a Trial | PRODUCT NAME: MARAVIROC, GEGEBENENFALLS IN DER FORM EINES PHARMAZEUTISCH VERTRAEGLICHEN SALZES; REGISTRATION NO/DATE: EU/1/07/418/001-010 20070918 |
| 1284974 | PA2008004 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: MARAVIROCUM; REGISTRATION NO/DATE: EU/1/07/418/001 - EU/1/07/418/010 20070918 |
| 1284974 | PA2008004,C1284974 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: MARAVIROCUM; REGISTRATION NO/DATE: EU/1/07/418/001 - EU/1/07/418/010, 0070918 |
| 1284974 | 91417 | Luxembourg | ⤷ Try a Trial | 91417, EXPIRES: 20220918 |
| 1284974 | C01284974/01 | Switzerland | ⤷ Try a Trial | FORMER OWNER: PFIZER INC., US |
| 1284974 | 314 | Finland | ⤷ Try a Trial | |
| 1284974 | SPC/GB08/014 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: MARAVIROC, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REGISTERED: UK EU/1/07/418/001 20070918; UK EU/1/04/418/002 20070918; UK EU/1/04/418/003 20070918; UK EU/1/04/418/004 20070918; UK EU/1/04/418/005 20070918; UK EU/1/04/418/006 20070918; UK EU/1/04/418/007 20070918; UK EU/1/04/418/008 20070918; UK EU/1/04/418/009 20070918; UK EU/1/04/418/010 20070918 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


