PROMETHAZINE Drug Patent Profile
✉ Email this page to a colleague
When do Promethazine patents expire, and what generic alternatives are available?
Promethazine is a drug marketed by Cenci, Pai Holdings, Abbott, Am Regent, Bedford Labs, Epic Pharma Llc, Hikma, Hospira, Marsam Pharms Llc, Mylan Institutional, Sandoz, Teva Pharms Usa, Watson Labs, Wockhardt Bio Ag, Xgen Pharms, Able, Annora Pharma, Cosette, Padagis Israel, Sun Pharma Canada, Watson Labs Inc, Amneal Pharms, Chartwell Rx, Kv Pharm, Pharm Assoc, Quagen, Strides Pharma, Whiteworth Town Plsn, Amneal Pharms Ny, Aurobindo Pharma Usa, Chartwell Molecular, Impax Labs, Ivax Sub Teva Pharms, Kvk Tech, Prinston Inc, Pvt Form, Rising, Sun Pharm Inds Inc, Sun Pharm Industries, Tablicaps, Teva, Zydus Pharms Usa, Actavis Mid Atlantic, Pharmobedient, Ani Pharms, Hetero Labs Ltd Iii, Xttrium Labs Inc, and Wockhardt. and is included in one hundred and one NDAs.
The generic ingredient in PROMETHAZINE is codeine phosphate; phenylephrine hydrochloride; promethazine hydrochloride. There are nineteen drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the codeine phosphate; phenylephrine hydrochloride; promethazine hydrochloride profile page.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for PROMETHAZINE?
- What are the global sales for PROMETHAZINE?
- What is Average Wholesale Price for PROMETHAZINE?
Summary for PROMETHAZINE
| US Patents: | 0 |
| Applicants: | 48 |
| NDAs: | 101 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 67 |
| Patent Applications: | 2,764 |
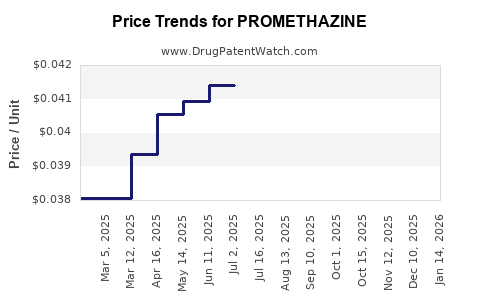
| Drug Prices: | Drug price information for PROMETHAZINE |
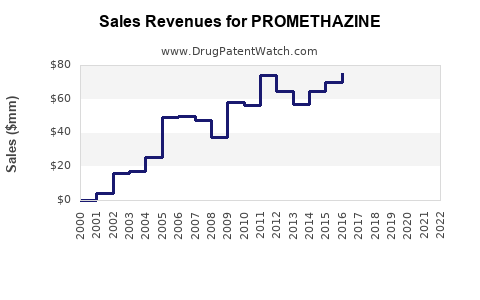
| Drug Sales Revenues: | Drug sales revenues for PROMETHAZINE |
| DailyMed Link: | PROMETHAZINE at DailyMed |
Recent Clinical Trials for PROMETHAZINE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Montefiore Medical Center | PHASE4 |
| Ji Xunming,MD,PhD | NA |
| Mercy Bon Secours Saint Vincent Medical Center | PHASE3 |
US Patents and Regulatory Information for PROMETHAZINE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Impax Labs | PROMETHAZINE HYDROCHLORIDE | promethazine hydrochloride | TABLET;ORAL | 040791-001 | May 20, 2008 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Sun Pharm Industries | PROMETHAZINE HYDROCHLORIDE | promethazine hydrochloride | TABLET;ORAL | 084557-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Watson Labs Inc | PROMETHAZINE HYDROCHLORIDE | promethazine hydrochloride | SUPPOSITORY;RECTAL | 040479-001 | Jun 24, 2003 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Able | PROMETHAZINE HYDROCHLORIDE | promethazine hydrochloride | SUPPOSITORY;RECTAL | 040449-001 | Feb 27, 2003 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Market Dynamics and Financial Trajectory for Promethazine
More… ↓