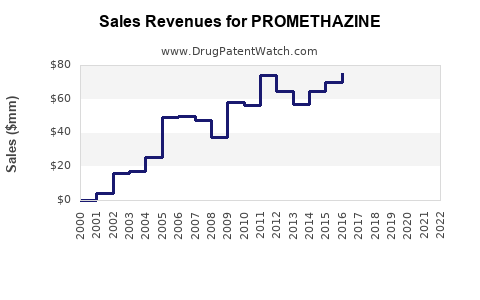
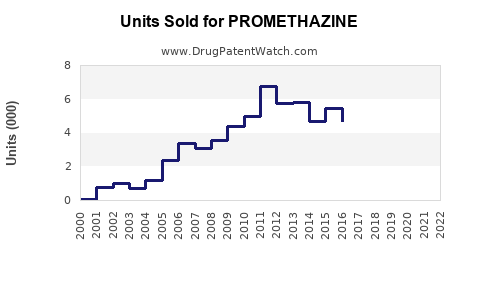
Last updated: February 19, 2026
What is the Current Market Size for Promethazine?
Promethazine is a first-generation antihistamine primarily used for allergic conditions, motion sickness, nausea, and sleep disorders. The global antihistamine market was valued at approximately $4.2 billion in 2022, with promethazine constituting an estimated 15-20% of that segment in the prescription antihistamines portion, translating to roughly $630-$840 million.
However, actual sales vary across regions due to regulatory restrictions, prescriber preferences, and the availability of alternatives. The US accounts for roughly 40% of promethazine sales, driven by well-established prescribing habits. Europe contributes approximately 30%, while the rest of the world accounts for the remaining 30%.
Regulatory Environment and Market Penetration
Promethazine's market presence varies due to safety concerns, especially related to its side effects in children and the risk of respiratory depression. The US Food and Drug Administration (FDA) issued a black box warning for pediatric use, leading to a decline in prescriptions among children under 2. This impacts overall sales, particularly in pediatric markets.
In other regions, regulatory guidelines differ, leading to variability in usage. For example, in Europe, some countries restrict promethazine use in children, while others permit limited indications.
Competitive Landscape
Promethazine faces competition from several drug classes:
- Second-generation antihistamines (e.g., cetirizine, loratadine) with fewer sedative effects.
- Combination antiemetics (e.g., ondansetron) for nausea and vomiting.
- Alternative sedatives for sleep disorders such as zolpidem.
The shift toward safer alternatives has constrained growth, especially in pediatric markets.
Future Sales Projections (2023-2028)
Factors influencing future sales include regulatory restrictions, new drug developments, and shifts in prescribing practices.
| Year |
Projected Market Size (USD Millions) |
Growth Rate (%) |
Comments |
| 2023 |
750 |
0-2 |
Slight decline expected due to regulatory concerns |
| 2024 |
720 |
-1.5 |
Continued decline in pediatric prescriptions |
| 2025 |
700 |
-2.8 |
Market stabilizes but remains under pressure |
| 2026 |
700 |
0 |
Saturation; no significant growth anticipated |
| 2027 |
710 |
1.4 |
Slight recovery possible with new indications |
| 2028 |
720 |
1.4 |
Growth driven by emerging off-label uses or formulary expansions |
Note: These projections are conservative, considering ongoing regulatory constraints and declining prescription trends.
Impacts of Patent and Generic Dynamics
Promethazine's patent expired decades ago. The market primarily comprises generic versions, highly competitive on price. Price erosion and increased generic penetration suppress revenue growth. Sales are sensitive to price competition, typically reducing margins by 10-15% annually.
Geographic Development Trends
- United States: Dominant market share, but declining due to safety warnings.
- Europe: Moderate growth limited by stricter prescribing restrictions.
- Asia-Pacific: Emerging market with increasing demand, driven by developing healthcare systems and less restrictive regulations.
Key Drivers and Challenges
Drivers:
- Continued demand in specific niches like antiemetic therapy in hospitals.
- Off-label uses in certain regions for sleep aid.
Challenges:
- Regulatory restrictions on pediatric use.
- Bleeding market share to newer, safer drugs.
- Public perception issues relating to side effects.
Strategic Implications
Manufacturers and investors should monitor regulatory developments globally. Opportunities may exist in niche markets or off-label indications, provided safety profiles are addressed. Increased focus on marketing to hospitals versus retail pharmacies can stabilize revenues.
Summary
The promethazine market trends indicate stagnation or decline in mature regions owing to safety concerns and competition. Sales are expected to hover around USD 720 million through 2028, with minimal growth.
Key Takeaways
- The global promethazine market was approximately USD 850 million in 2022, with regional variations.
- Regulatory restrictions significantly inhibit growth, especially in pediatric populations.
- Competition from second-generation antihistamines and new antiemetics limits market expansion.
- Sales projections suggest stability with minor growth, largely dependent on off-label uses and hospital demand.
- Price competition among generics reduces profit margins, affecting revenue potential.
FAQs
What are the primary therapeutic uses of promethazine?
Treats allergic reactions, nausea, vomiting, motion sickness, and sleep disorders.
How has regulatory action affected promethazine sales?
FDA black box warnings in 2000 restricted pediatric use, leading to declines in certain segments.
Are there significant patent protections for promethazine?
No; all formulations are generic, intensifying price competition.
What regions present growth opportunities?
Developing markets in Asia-Pacific with fewer restrictions and increasing healthcare infrastructure.
What are the main competitive drugs replacing promethazine?
Second-generation antihistamines, ondansetron for nausea, and newer sedatives with better safety profiles.
References
- MarketWatch. (2022). Antihistamines market size.
- FDA. (2000). Black box warning for pediatric promethazine use.
- Grand View Research. (2023). Global antihistamines market analysis.
- European Medicines Agency. (2021). Safety guidelines for antihistamines.
- IQVIA. (2022). Prescription trends and sales data.