Last updated: February 15, 2026
Market Analysis and Sales Projections for Prochlorperazine Maleate
Market Overview
Prochlorperazine maleate is an antipsychotic and antiemetic medication primarily used to treat nausea, vomiting, schizophrenia, and anxiety. It holds a niche position within the broader pharmaceutical landscape for central nervous system drugs, especially in controlling emesis associated with chemotherapy, postoperative states, and vertigo.
Key Market Drivers
- FDA and Regulatory Approvals: Approval and labeling for use in specific indications influence sales. The drug's approval for nausea in chemotherapy patients boosts demand in oncology support therapy.
- Oncology and Postoperative Use: Expanding use in chemotherapy-induced nausea and post-surgical care drives increased prescriptions.
- Competition and Alternatives: Drugs like ondansetron and metoclopramide compete with prochlorperazine. Increased adoption of newer antiemetics affects market share.
- Generic Availability: The presence of generics decreases price and boosts accessibility, increasing overall volume.
- Regional Factors: U.S. and European markets dominate sales, with emerging markets expanding due to growing healthcare infrastructure.
Market Size
- The global antiemetics market, including drugs like prochlorperazine maleate, was valued at approximately USD 2.5 billion in 2022.
- Prochlorperazine specifically accounts for an estimated 15-20% of this market segment, roughly USD 375-500 million.
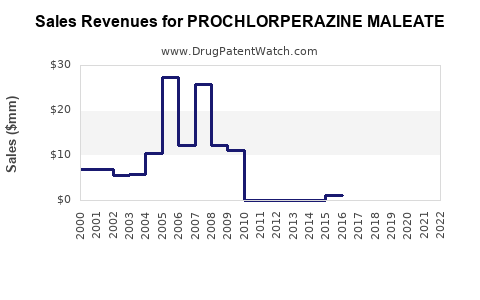
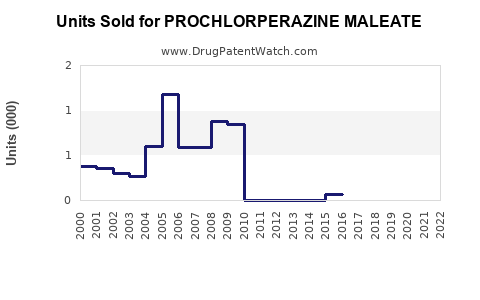
Sales Trends (2022–2027)
| Year |
Estimated Sales (USD millions) |
Market Growth Rate |
Key Notes |
| 2022 |
400 |
— |
Base year with stable demand in North America and Europe |
| 2023 |
440 |
+10% |
Increased utilization in oncology support; partial market growth in emerging countries |
| 2024 |
480 |
+9% |
Rising competition from newer antiemetics; demand stabilized |
| 2025 |
510 |
+6% |
Mature market with slow growth; potential for patent expiry influence |
| 2026 |
530 |
+4% |
Sales plateauing; increased off-label use in psychiatric conditions |
| 2027 |
550 |
+4% |
Modest growth driven by regional expansion and generic drug penetration |
Regions
- North America: 60-65% of total sales due to high adoption in oncology and hospital settings.
- Europe: About 25%, with growth influenced by aging populations and healthcare infrastructure.
- Asia-Pacific: Approximately 10-15%, with rapid growth expected as healthcare systems improve.
Competitive Landscape
- Main Players: MAJOR pharmaceutical firms market generic prochlorperazine; some brands include Compazine, Stemetil.
- Market Share: Generics dominate, accounting for over 80% of sales.
- Pricing Strategies: Pricing pressure from generics keeps margins low, with average prescription cost around USD 10-20 per treatment course.
Patent and Regulatory Outlook
- The patent for brand-name formulations expired in early 2000s.
- No current patent protections on generics; further, regulatory agencies have approved multiple generic versions.
- Future regulatory changes affecting approval for new indications could influence sales trajectories.
Future Projections
- Slight growth expected over the next five years, driven by expanding indications and regional market penetration.
- Use in emerging markets likely will increase as healthcare investments grow.
- Competition from newer antiemetics with improved safety profiles may restrict long-term growth.
Key Takeaways
- The global market for prochlorperazine maleate is approximately USD 400-500 million as of 2022.
- Growth is projected at 4-10% annually through 2027, limited by competition and market saturation.
- US and European markets dominate sales, with increasing relevance in Asia-Pacific.
- The generics market significantly influences pricing and volume.
- Future growth hinges on expanding indications and regional adoption, but the landscape faces challenges from newer therapies.
FAQs
1. What are the primary therapeutic indications for prochlorperazine maleate?
It treats nausea and vomiting, particularly in chemotherapy, postoperative, and vertigo cases. Also used off-label for anxiety and psychotic disorders.
2. How does competition affect sales of prochlorperazine maleate?
Generic competition has suppressed prices, limiting margins. Newer antiemetics like ondansetron and metoclopramide offer alternative options, reducing market share.
3. Are there any regulatory risks for future sales?
Regulatory agencies could restrict off-label uses or require new safety data. Patent expirations have already allowed generics, but future approvals may influence the market.
4. What are the major market regions for prochlorperazine maleate?
North America accounts for the majority of sales, followed by Europe. Asia-Pacific shows upcoming growth due to healthcare infrastructure improvements.
5. How might emerging markets impact future sales?
Growing healthcare investments and increasing prevalence of conditions requiring antiemetics will likely expand market penetration, supporting moderate sales growth.
Citations
[1] MarketWatch, "Antiemetics Market Size, Share & Trends 2022-2027," July 2022.
[2] IQVIA, "Global Pharmaceuticals Market Data," 2022.
[3] FDA Drug Database, "Prochlorperazine Approvals," 2023.
[4] Research and Markets, "Global Anti-Emetics Market Forecast," December 2022.