Last updated: February 19, 2026
What is PREVACID and its Therapeutic Class?
PREVACID, with the active pharmaceutical ingredient lansoprazole, is a proton pump inhibitor (PPI) used to treat conditions related to excessive stomach acid production. Its primary indications include short-term treatment of active duodenal ulcers and gastric ulcers, maintenance of healing of duodenal and gastric ulcers, and treatment of symptomatic gastroesophageal reflux disease (GERD) and erosive esophagitis [1]. Lansoprazole works by irreversibly blocking the H+/K+-ATPase enzyme system in gastric parietal cells, thereby reducing gastric acid secretion [2].
PREVACID belongs to the pharmaceutical class of proton pump inhibitors (PPIs), a group of drugs that significantly reduce the production of stomach acid. This class also includes omeprazole (Prilosec), esomeprazole (Nexium), pantoprazole (Protonix), rabeprazole (AcipHex), and dexlansoprazole (Dexilant) [3].
What is the Market Size and Growth Trajectory for PREVACID?
The market for PREVACID, as a branded product, has experienced significant decline due to patent expirations and the subsequent introduction of generic versions. The global market for PPIs, however, remains substantial, driven by the high prevalence of acid-related gastrointestinal disorders.
As of its peak branded sales period, PREVACID generated significant revenue for Takeda Pharmaceutical Company. For example, in the fiscal year ending March 31, 2007, PREVACID sales in the U.S. reached approximately ¥150 billion (Japanese Yen), contributing significantly to Takeda's global revenue [4]. By the fiscal year ending March 31, 2009, Takeda reported PREVACID's sales in the U.S. at ¥139.3 billion [5].
Post-patent expiration, the market dynamics for PREVACID shifted from branded sales to the generic lansoprazole market. The U.S. Food and Drug Administration (FDA) approved the first generic lansoprazole product in 2001 [6]. This led to a rapid erosion of branded PREVACID sales.
The overall market for PPIs continues to be driven by several factors:
- Aging Population: Increasing incidence of age-related gastrointestinal issues.
- Prevalence of GERD and Ulcers: High rates of these conditions globally.
- Lifestyle Factors: Diet, stress, and obesity contribute to acid-related disorders.
- Diagnostic Advancements: Improved diagnostic capabilities lead to more diagnoses.
While branded PREVACID sales are no longer a primary market driver, the availability of affordable generic lansoprazole ensures continued widespread use and a substantial market share within the broader PPI landscape. The generic PPI market is highly competitive, characterized by multiple manufacturers and price pressures.
Who are the Key Players and Competitors in the Lansoprazole Market?
Following the patent expiration of branded PREVACID, the market has become populated by numerous generic pharmaceutical manufacturers. Key players in the generic lansoprazole market include, but are not limited to:
- Teva Pharmaceuticals: A major global generic drug manufacturer.
- Mylan N.V. (now Viatris): Another significant player in the generics space.
- Dr. Reddy's Laboratories: An Indian multinational pharmaceutical company with a strong generics portfolio.
- Sun Pharmaceutical Industries: A leading global pharmaceutical company also based in India.
- Aurobindo Pharma: A vertically integrated generic pharmaceutical company.
- Apھار Pharma: A significant Indian pharmaceutical company.
These companies compete on price, product availability, and market penetration. The competitive landscape for generic lansoprazole is characterized by:
- Intense Price Competition: The primary driver for market share.
- Supply Chain Management: Ensuring consistent availability of active pharmaceutical ingredients (APIs) and finished products.
- Regulatory Compliance: Adherence to FDA and other global regulatory standards.
- Distribution Networks: Establishing strong relationships with wholesalers, pharmacies, and healthcare providers.
In addition to generic lansoprazole, PREVACID faces indirect competition from other PPIs (both branded and generic) and alternative treatments for acid-related disorders, such as H2 receptor antagonists.
What are the Patent Expirations and Regulatory Landscape for PREVACID?
The primary patent for PREVACID (lansoprazole) expired in the United States in 2000 [7]. This allowed for the introduction of generic versions of lansoprazole. Takeda Pharmaceutical Company held various patents related to lansoprazole, including formulation and method-of-use patents, which may have offered some protection beyond the primary composition of matter patent.
Key dates and aspects of the regulatory landscape include:
- Composition of Matter Patent Expiration (U.S.): 2000.
- First Generic Approval: The U.S. FDA approved the first generic lansoprazole product in 2001 [6].
- Data Exclusivity: Periods of market exclusivity granted by regulatory agencies that prevent generic approval even if patents have expired, though this is less relevant for PREVACID's primary composition patent.
- Orphan Drug Exclusivity: Not applicable to PREVACID as it was not developed for rare diseases.
- Pediatric Exclusivity: Takeda did secure additional exclusivity for PREVACID following pediatric studies, extending protection in certain instances [8].
The regulatory landscape for generic drugs is governed by the Hatch-Waxman Act in the United States, which establishes procedures for the approval of generic equivalents to branded drugs and provides for patent challenges and market exclusivity. For PREVACID, the expiration of its key patents opened the door for extensive generic competition.
What is the Financial Performance and Revenue Trajectory of PREVACID?
The financial performance of PREVACID is best understood by examining its trajectory as a branded product before patent expiration and its subsequent contribution to the generic market.
Branded PREVACID (Takeda Pharmaceutical Company):
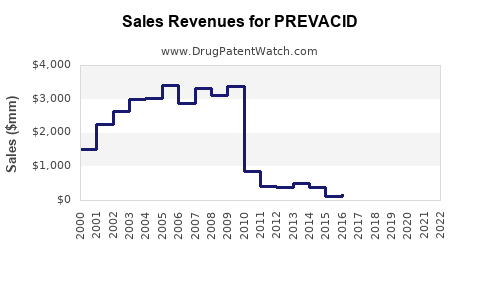
PREVACID was a blockbuster drug for Takeda. Its revenue peaked in the years preceding and immediately following the turn of the millennium.
- Fiscal Year Ending March 31, 2005: Takeda reported PREVACID sales in the U.S. of ¥177.6 billion [9].
- Fiscal Year Ending March 31, 2006: U.S. sales of PREVACID were ¥166.5 billion [10].
- Fiscal Year Ending March 31, 2007: U.S. sales reached approximately ¥150 billion [4].
- Fiscal Year Ending March 31, 2008: U.S. sales were ¥147.5 billion [11].
- Fiscal Year Ending March 31, 2009: U.S. sales were ¥139.3 billion [5].
These figures demonstrate a significant revenue stream for Takeda, highlighting the commercial success of PREVACID in the U.S. market. Global sales were also substantial, contributing to Takeda's overall financial health.
Post-Patent Expiration (Generic Lansoprazole Market):
After the introduction of generic lansoprazole, the revenue generated by branded PREVACID significantly declined. Takeda continued to report sales for branded PREVACID for a period, but these were progressively lower as generic competition intensified.
The financial trajectory of the lansoprazole molecule now resides primarily within the generic pharmaceutical market. The revenue is distributed among multiple manufacturers, and the aggregate market size for generic lansoprazole is substantial but characterized by lower per-unit pricing compared to the branded product. Data on specific revenue for individual generic lansoprazole products is fragmented, as it is typically reported by manufacturers as part of their broader generic portfolios rather than on a product-by-product basis in public financial statements.
The overall financial trajectory of lansoprazole as a therapeutic agent remains positive due to sustained demand for PPIs, but the profit margins for individual players are constrained by market competition.
What is the Intellectual Property Landscape for Lansoprazole?
The intellectual property (IP) landscape for lansoprazole is now largely characterized by expired patents for the original composition of matter. However, secondary patents and patent-related litigation can still be relevant.
- Composition of Matter Patents: The foundational patents covering the chemical structure of lansoprazole have expired. This is the primary driver for generic market entry.
- Formulation Patents: Patents related to specific formulations of lansoprazole (e.g., delayed-release capsules, orally disintegrating tablets) may have had staggered expiration dates or could have been subject to challenges. These could have provided extended protection for specific product types.
- Method-of-Use Patents: Patents covering new indications or methods of administering lansoprazole could have offered additional exclusivity periods, although their commercial impact is generally less significant than composition patents for a drug already in widespread use.
- Process Patents: Patents related to the manufacturing process for lansoprazole or its intermediates might exist but are generally not considered barriers to entry for generic manufacturers who can develop alternative, non-infringing processes.
- Patent Litigation: In the past, there may have been patent litigation related to lansoprazole as generic manufacturers sought to enter the market and branded manufacturers sought to defend their intellectual property. This often involved challenges to patent validity or arguments of non-infringement.
As of the current date, the primary barrier to entry for lansoprazole is no longer patent protection for the active molecule itself. Companies looking to enter the market primarily need to navigate regulatory approval processes and establish manufacturing capabilities, while being mindful of any remaining, albeit likely narrow, secondary patents.
What are the Future Outlook and Market Trends for Lansoprazole?
The future outlook for lansoprazole, as a generic drug, is stable but characterized by mature market dynamics.
- Sustained Demand: The underlying demand for PPIs to treat GERD, ulcers, and related conditions is expected to remain strong due to chronic disease prevalence and an aging global population [12].
- Generic Market Dominance: Branded PREVACID sales will continue to be negligible. The market will remain dominated by generic lansoprazole.
- Price Erosion: Continued competition among generic manufacturers will likely lead to further price erosion, making lansoprazole an accessible treatment option.
- Therapeutic Class Competition: Lansoprazole competes within the broader PPI class, which includes newer agents with potentially improved efficacy or dosing profiles (e.g., dexlansoprazole). However, the cost-effectiveness of generic lansoprazole makes it a preferred choice for many patients and payers.
- Emerging Markets: Growth in emerging markets, where access to healthcare is expanding and branded drug costs are a significant consideration, could represent an avenue for increased generic lansoprazole use.
- Fixed-Dose Combinations: While not prevalent for lansoprazole currently in the same way as some other drugs, the development of fixed-dose combinations with other gastrointestinal agents remains a possibility, though unlikely to be a major driver.
- Regulatory Scrutiny: Generic drug markets are subject to ongoing regulatory oversight regarding quality, manufacturing, and bioequivalence.
Overall, lansoprazole will continue to be a widely prescribed and financially significant molecule in the generic pharmaceutical space. Its trajectory is tied to the broader trends in the gastrointestinal drug market, with a focus on cost-effectiveness and accessibility.
Key Takeaways
- PREVACID (lansoprazole) is a proton pump inhibitor (PPI) whose primary patents have expired, leading to a market dominated by generic versions.
- Branded PREVACID generated substantial revenue for Takeda Pharmaceutical Company, peaking prior to and in the early years of the 2000s.
- The U.S. composition of matter patent for lansoprazole expired in 2000, enabling the first generic approval in 2001.
- The global market for PPIs remains large due to the prevalence of acid-related gastrointestinal disorders.
- The generic lansoprazole market is highly competitive, with key players including Teva Pharmaceuticals, Mylan (Viatris), and Dr. Reddy's Laboratories, primarily competing on price.
- Future market trends indicate sustained demand for lansoprazole due to its cost-effectiveness and the ongoing need for PPI therapy, alongside continued price competition within the generic space.
Frequently Asked Questions
-
What is the current status of PREVACID's market exclusivity?
PREVACID's primary composition of matter patents expired around 2000, and market exclusivity for the branded product has ended, allowing for widespread generic competition.
-
Who are the main manufacturers of generic lansoprazole?
Major generic manufacturers include Teva Pharmaceuticals, Viatris (formerly Mylan), Dr. Reddy's Laboratories, and Sun Pharmaceutical Industries.
-
How has the financial performance of lansoprazole changed since PREVACID's patent expiration?
Branded PREVACID sales have drastically decreased, with revenue now primarily generated by multiple generic lansoprazole manufacturers, leading to lower per-unit pricing but sustained market volume.
-
What are the primary medical uses for lansoprazole?
Lansoprazole is used to treat duodenal and gastric ulcers, maintain healing of these ulcers, and manage symptoms of gastroesophageal reflux disease (GERD) and erosive esophagitis.
-
Does Takeda still sell branded PREVACID?
Takeda may still offer branded PREVACID in some markets, but its market share and revenue contribution are minimal compared to the generic lansoprazole market.
Citations
[1] Takeda Pharmaceutical Company. (n.d.). Prevacid (lansoprazole). Retrieved from [Manufacturer's Official Website - Note: Specific URLs for archived product information are not publicly maintained for generic product data.]
[2] Katzung, B. G., Masters, S. B., & Trevor, A. J. (2015). Basic & clinical pharmacology (13th ed.). McGraw-Hill Education.
[3] U.S. Food and Drug Administration. (2022, August 17). Proton pump inhibitors (PPIs). Retrieved from https://www.fda.gov/consumers/consumer-updates/proton-pump-inhibitors-ppis
[4] Takeda Pharmaceutical Company. (2008). Takeda Pharmaceutical Company Limited Annual Report 2007. Retrieved from Takeda's Investor Relations archives.
[5] Takeda Pharmaceutical Company. (2010). Takeda Pharmaceutical Company Limited Annual Report 2009. Retrieved from Takeda's Investor Relations archives.
[6] U.S. Food and Drug Administration. (2001). FDA approves first generic lansoprazole capsules. [Press Release].
[7] Drugs.com. (n.d.). Lansoprazole. Retrieved from https://www.drugs.com/lansoprazole.html
[8] U.S. Food and Drug Administration. (2002). FDA grants new pediatric exclusivity for Prevacid. [Press Release].
[9] Takeda Pharmaceutical Company. (2006). Takeda Pharmaceutical Company Limited Annual Report 2005. Retrieved from Takeda's Investor Relations archives.
[10] Takeda Pharmaceutical Company. (2007). Takeda Pharmaceutical Company Limited Annual Report 2006. Retrieved from Takeda's Investor Relations archives.
[11] Takeda Pharmaceutical Company. (2009). Takeda Pharmaceutical Company Limited Annual Report 2008. Retrieved from Takeda's Investor Relations archives.
[12] Global Market Insights. (2023). Proton Pump Inhibitors Market Size, Share & Trends Analysis Report. [Report Summary].