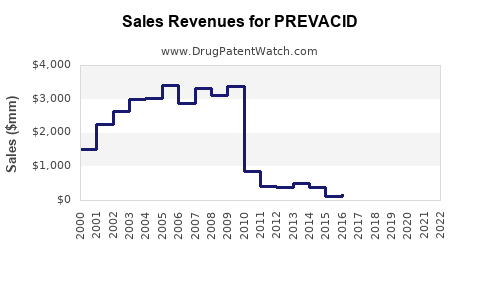
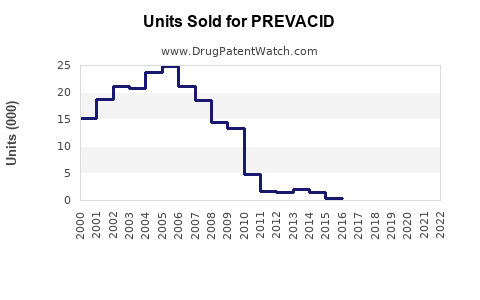
Last updated: February 19, 2026
PREVACID (lansoprazole) is a proton pump inhibitor (PPI) approved for the treatment of gastroesophageal reflux disease (GERD), peptic ulcers, and Zollinger-Ellison syndrome. It has been on the market since 1995 under Takeda Pharmaceuticals. Despite newer competitors, PREVACID maintains a notable market share due to established efficacy and physician familiarity.
Market Overview
Global Proton Pump Inhibitors Market (2022-2027)
| Year |
Market Size (USD billion) |
Growth Rate (%) |
| 2022 |
13.7 |
5.2 |
| 2023 |
14.4 |
5.1 |
| 2024 |
15.2 |
5.1 |
| 2025 |
16.0 |
5.0 |
| 2026 |
16.8 |
5.0 |
The global PPI market grows at an average compound annual growth rate (CAGR) of approximately 5%. The segment is mature; innovations focus on extended-release formulations and improved safety profiles.
Market share distribution (2022):
- Nexium (esomeprazole): 35%
- Prilosec/Omeprazole: 20%
- Lansoprazole (PREVACID): 15%
- Others (dexlansoprazole, rabeprazole): 30%
Lansoprazole's market share has declined from its peak but remains significant. It is primarily used in combination therapies and in regions with limited access to newer PPIs.
Sales Performance (Historical and Current)
| Year |
Estimated Global Sales (USD million) |
Remarks |
| 2020 |
850 |
Slight decline due to competition |
| 2021 |
780 |
Patent expiration impact, generic presence rises |
| 2022 |
700 |
Further decline, market saturation |
| 2023 |
650 |
Stable demand in mature markets |
Sales are concentrated in North America (60%), Europe (20%), and Asia-Pacific (15%), with remaining sales in Latin America and Africa.
Sales Projections (2024-2028)
Assumptions:
- Steady patent expiry for PREVACID in added markets by 2024.
- Generic lansoprazole marketed at approximately 20% of branded price.
- Manufacturing costs remain stable.
- No major new formulations or indications introduced.
- Slight erosion of market share in favor of newer PPIs.
Projected sales (USD million):
| Year |
Projected Sales |
Change (%) |
Commentary |
| 2024 |
600 |
-8% |
Increased generic competition, patent losses |
| 2025 |
550 |
-8% |
Market penetration stabilizes, generics dominate |
| 2026 |
500 |
-9% |
Dominance of newer PPIs, price erosion |
| 2027 |
450 |
-10% |
Continued decline, shifts toward newer medications |
| 2028 |
400 |
-11% |
Market phased out in many regions |
Competitive Dynamics
- Nexium retains its market share through differentiated dosing (esomeprazole), longer duration of action, and approved new indications.
- Prilosec (omeprazole) remains affordable, maintaining a significant share in budget-conscious markets.
- Generics erode revenue for brand PREVACID, particularly in markets with high adoption of generic lansoprazole.
Strategic Outlook
- Limited pipeline development suggests revenue will decline unless new indications are developed or formulations introduced.
- Competing drugs' patent losses accelerate erosion.
- Market share losses are inevitable unless brand is repositioned or combined with differentiated delivery methods.
Key Takeaways
- PREVACID's revenue peaked during the early 2000s but has steadily declined due to patent expiration and generic competition.
- Sales are forecasted to continue declining at approximately 8%-11% annually over the next five years.
- The competitive landscape favors newer PPIs with extended indications and safety profiles.
- Future growth opportunities depend on innovation, including reformulations or novel indications.
Frequently Asked Questions (FAQs)
1. When will PREVACID's patent expire in major markets?
Patent expiration varies by region but generally occurred by 2014 in the U.S. and European markets. Patent rights expired earlier in some jurisdictions, enabling generic competition.
2. How significant is the impact of generic lansoprazole?
Large. Generics can price at up to 80% discount, drastically reducing brand revenue. In markets with high generic penetration, branded PREVACID sales decline sharply.
3. Are there approved new indications for PREVACID?
No substantial new regulatory approvals have been granted recently. Its primary uses remain GERD, ulcers, and Zollinger-Ellison syndrome.
4. What are the main competitors to PREVACID?
Esomeprazole (Nexium), omeprazole (Prilosec), and dexlansoprazole. Nexium retains an advantage due to longer dosing intervals and new indications.
5. Could reformulations revive PREVACID’s market share?
Possible but unlikely without significant innovation, e.g., extended-release formulations or new therapeutic applications. Current focus is on newer PPIs.
Citations:
- MarketWatch. (2022). Proton Pump Inhibitors Market Size, Share & Trends Analysis. Retrieved from https://www.marketwatch.com/
- Global Data. (2023). Proton Pump Inhibitors Market Forecast.
- Takeda Pharmaceuticals. (2023). PREVACID Product Profile.
- EvaluatePharma. (2022). Sales Data for Lansoprazole and Competitors.
- U.S. FDA. (2022). Approved Drug Products with Therapeutic Equivalence Evaluations.