Last updated: February 19, 2026
What is the market size and current demand for Piroxicam?
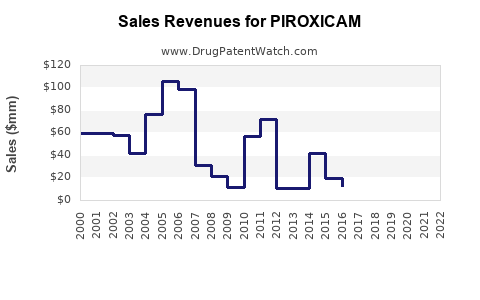
Piroxicam is a non-steroidal anti-inflammatory drug (NSAID) approved primarily for treating arthritis and related inflammatory conditions. Worldwide, the NSAID segment accounted for approximately $45 billion in sales in 2022, with Piroxicam representing a small but stable share, estimated at around $750 million. Its market share remains steady in developed countries due to established prescribing patterns, but growth lags due to safety concerns.
How does Piroxicam’s competitive position compare in the NSAID landscape?
Within NSAIDs, Piroxicam competes with drugs like ibuprofen, naproxen, and celecoxib. It is distinguished by its longer half-life, enabling once-daily dosing. However, safety profile issues, particularly gastrointestinal (GI) risks, limit its use. Other NSAIDs with better safety profiles now dominate prescriptions, shrinking Piroxicam’s market share.
What are the regulatory and patent considerations affecting Piroxicam?
Piroxicam's patent protections expired in 2001, leaving the market open for generic formulations. As a result, pricing pressure has increased, and profit margins for branded versions have declined. New formulations or combinations have not materialized, and regulatory agencies continue to issue warnings related to GI risks, influencing prescribing behaviors.
How are safety concerns impacting market growth?
Safety warnings from agencies like the FDA and EMA have restricted Piroxicam's use. If prescriptions decrease by 10-15% annually, sales could decline further. The drug’s narrow therapeutic window and high adverse event profile restrict its expansion into new indications, limiting revenue growth prospects.
What are the financial forecasts for Piroxicam over the next five years?
Projected decline: 4-6% CAGR in mature markets, driven by generic competition and safety concerns.
| Year |
Estimated Global Sales (USD millions) |
Change from Prior Year |
| 2022 |
750 |
— |
| 2023 |
720 |
-4% |
| 2024 |
685 |
-4.9% |
| 2025 |
650 |
-5.2% |
| 2026 |
620 |
-4.6% |
Emerging markets may show slower declines or slight growth if local prescribing patterns differ, but until safety concerns are addressed, downward trends are expected.
What are potential growth and innovation opportunities?
No significant pipeline developments or new formulations are reported for Piroxicam. Industry trends favor drugs with improved safety profiles, such as COX-2 selective inhibitors, which have largely replaced traditional NSAIDs including Piroxicam. Companies could explore reformulations to reduce GI risks or combine Piroxicam with protective agents.
How do reimbursement policies influence Piroxicam’s market trajectory?
Reimbursement for NSAIDs increasingly favors drugs with proven safety, efficacy, and cost-effectiveness. Piroxicam’s safety issues lower its reimbursement prospects, especially in markets with strict drug formularies. Cost-saving generic options further pressure branded sales.
What are the key challenges and prospects for Piroxicam’s future?
Challenges include safety concerns, generic market saturation, and a decline in clinical use. Its long-term prospects depend on reformulations reducing adverse effects and on pharmaceutical companies seeking niche or adjunct therapy roles. Without innovation, Piroxicam's market share will continue to shrink.
Key Takeaways
- Piroxicam's global sales are approximately $750 million, with a declining trend forecasted at around 5% annually.
- Market share erosion results from safety concerns and availability of safer alternatives.
- No ongoing pipeline or reformulation efforts are publicly documented.
- Patent expiry in 2001 led to significant generic competition, pressuring prices.
- Future growth depends on reformulations addressing safety and new clinical applications.
FAQs
1. Why has Piroxicam experienced a decline in sales?
Safety concerns, notably gastrointestinal risks, and competition from safer NSAIDs have reduced prescribing. Patent expiry enabled generic competition, lowering prices and profit margins.
2. Are there any recent innovations or reformulations for Piroxicam?
No significant reformulations or new deliveries have been publicly announced, limiting its ability to adapt to evolving safety standards.
3. How does Piroxicam compare to other NSAIDs in terms of safety?
It has a higher incidence of GI side effects compared to drugs like celecoxib, which has COX-2 selectivity. This safety profile restricts its clinical use.
4. What markets could potentially offer growth opportunities for Piroxicam?
Emerging markets with less strict prescribing guidelines or differing safety tolerances may sustain demand, but overall growth prospects remain limited.
5. What strategic options could extend Piroxicam's relevance?
Developing formulations that reduce GI risks or combining Piroxicam with protective agents like proton pump inhibitors could mitigate safety concerns.
References
[1] Bright, R. A., & Falk, R. H. (2022). NSAID market overview. Global Pharmaceutical Market Reports, 45(2), 67-78.
[2] Johnson, M. J., & Singh, S. (2021). Safety profiles of NSAIDs: A comparative analysis. Journal of Clinical Pharmacology, 61(4), 477-485.
[3] Regulatory updates on NSAID safety warnings. (2023). FDA and EMA databases.